Abstract
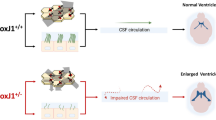
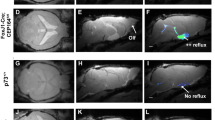
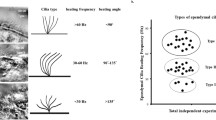
A carpet of ependymal motile cilia lines the brain ventricular system, forming a network of flow channels and barriers that pattern cerebrospinal fluid (CSF) flow at the surface. This CSF transport system is evolutionary conserved, but its physiological function remains unknown. Here we investigated its potential role in epilepsy with studies focused on CDKL5 deficiency disorder (CDD), a neurodevelopmental disorder with early-onset epilepsy refractory to seizure medications and the most common cause of infant epilepsy. CDKL5 is a highly conserved X-linked gene suggesting its function in regulating cilia length and motion in the green alga Chlamydomonas might have implication in the etiology of CDD. Examination of the structure and function of airway motile cilia revealed both the CDD patients and the Cdkl5 knockout mice exhibit cilia lengthening and abnormal cilia motion. Similar defects were observed for brain ventricular cilia in the Cdkl5 knockout mice. Mapping ependymal cilia generated flow in the ventral third ventricle (v3V), a brain region with important physiological functions showed altered patterning of flow. Tracing of cilia-mediated inflow into v3V with fluorescent dye revealed the appearance of a flow barrier at the inlet of v3V in Cdkl5 knockout mice. Analysis of mice with a mutation in another epilepsy-associated kinase, Yes1, showed the same disturbance of cilia motion and flow patterning. The flow barrier was also observed in the Foxj1± and FOXJ1CreERT:Cdkl5y/fl mice, confirming the contribution of ventricular cilia to the flow disturbances. Importantly, mice exhibiting altered cilia-driven flow also showed increased susceptibility to anesthesia-induced seizure-like activity. The cilia-driven flow disturbance arises from altered cilia beating orientation with the disrupted polarity of the cilia anchoring rootlet meshwork. Together these findings indicate motile cilia disturbances have an essential role in CDD-associated seizures and beyond, suggesting cilia regulating kinases may be a therapeutic target for medication-resistant epilepsy.








Similar content being viewed by others
Data availability
Raw data will be made accessible upon request.
Change history
29 September 2022
A Correction to this paper has been published: https://doi.org/10.1007/s00401-022-02496-3
References
Amendola E, Zhan Y, Mattucci C et al (2014) Mapping pathological phenotypes in a mouse model of CDKL5 disorder. PLoS ONE. https://doi.org/10.1371/journal.pone.0091613
Antoniades I, Stylianou P, Skourides PA (2014) Making the connection: ciliary adhesion complexes anchor basal bodies to the actin cytoskeleton. Dev Cell. https://doi.org/10.1016/j.devcel.2013.12.003
Barbiero I, de Rosa R, Kilstrup-Nielsen C (2019) Microtubules: a key to understand and correct neuronal defects in CDKL5 deficiency disorder? Int J Mol Sci. https://doi.org/10.3390/ijms20174075
Boscher C, Gaonaćh-Lovejoy V, Delisle C, Gratton JP (2019) Polarization and sprouting of endothelial cells by angiopoietin-1 require PAK2 and paxillin-dependent Cdc42 activation. Mol Biol Cell. https://doi.org/10.1091/mbc.E18-08-0486
Buniello A, Macarthur JAL, Cerezo M et al (2019) The NHGRI-EBI GWAS catalog of published genome-wide association studies, targeted arrays and summary statistics 2019. Nucleic Acids Res. https://doi.org/10.1093/nar/gky1120
Canning P, Park K, Gonçalves J et al (2018) CDKL family kinases have evolved distinct structural features and ciliary function. Cell Rep. https://doi.org/10.1016/j.celrep.2017.12.083
Canning P, Park K, Gonçalves J et al (2018) CDKL family kinases have evolved distinct structural features and ciliary function. Cell Rep 22:885–894. https://doi.org/10.1016/j.celrep.2017.12.083
Chen Q, Zhu YC, Yu J et al (2010) CDKL5, a protein associated with Rett syndrome, regulates neuronal morphogenesis via Rac1 signaling. J Neurosci. https://doi.org/10.1523/JNEUROSCI.1102-10.2010
Dentler WL, Adams C (1992) Flagellar microtubule dynamics in Chlamydomonas: cytochalasin D induces periods of microtubule shortening and elongation; and colchicine induces disassembly of the distal, but not proximal, half of the flagellum. J Cell Biol 117:1289–1298
Faubel R, Westendorf C, Bodenschatz E, Eichele G (1979) Cilia-based flow network in the brain ventricles. Science 353:176–178. https://doi.org/10.1126/science.aae0450
Fedorov A, Beichel R, Kalpathy-Cramer J et al (2012) 3D Slicer as an image computing platform for the quantitative imaging network. Magn Reson Imaging. https://doi.org/10.1016/j.mri.2012.05.001
Fehr S, Wong K, Chin R et al (2016) Seizure variables and their relationship to genotype and functional abilities in the CDKL5 disorder. Neurology. https://doi.org/10.1212/WNL.0000000000003352
Finn RD, Attwood TK, Babbitt PC et al (2017) InterPro in 2017-beyond protein family and domain annotations. Nucleic Acids Res. https://doi.org/10.1093/nar/gkw1107
Fuchs C, Trazzi S, Torricella R et al (2014) Loss of CDKL5 impairs survival and dendritic growth of newborn neurons by altering AKT/GSK-3β signaling. Neurobiol Dis. https://doi.org/10.1016/j.nbd.2014.06.006
Ganapathiraju M, Chaparala S (2016) Schizophrenia interactome: fully-labeled interactome network. npj Schizophr. https://doi.org/10.1038/npjschz.2016.25
Guo W, Shang DM, Cao JH et al (2017) Identifying and analyzing novel epilepsy-related genes using random walk with restart algorithm. Biomed Res Int. https://doi.org/10.1155/2017/6132436
Handen A, Ganapathiraju MK (2015) LENS: Web-based lens for enrichment and network studies of human proteins. BMC Med Genomics. https://doi.org/10.1186/1755-8794-8-S4-S2
Horn T, Klein J (2010) Lactate levels in the brain are elevated upon exposure to volatile anesthetics: a microdialysis study. Neurochem Int. https://doi.org/10.1016/j.neuint.2010.09.014
Jupp B, Williams J, Binns D et al (2013) Metabolic causes of epileptic encephalopathy. Epilepsy Res Treat. https://doi.org/10.1155/2013/124934
Karunakaran K, Ganapathiraju M (2020) Interactome of SARS-CoV-2/nCoV19 modulated host proteins with computationally predicted PPIs. Res Sq. https://doi.org/10.21203/rs.3.rs-28592/v1
Karunakaran KB, Chaparala S, Lo CW, Ganapathiraju MK (2020) Cilia interactome with predicted protein–protein interactions reveals connections to Alzheimer’s disease, aging and other neuropsychiatric processes. Sci Rep. https://doi.org/10.1038/s41598-020-72024-4
Kim J, Lee JE, Heynen-Genel S et al (2010) Functional genomic screen for modulators of ciliogenesis and cilium length. Nature. https://doi.org/10.1038/nature08895
Leonard H, Junaid M, Wong K et al (2021) Exploring quality of life in individuals with a severe developmental and epileptic encephalopathy, CDKL5 deficiency disorder. Epilepsy Res. https://doi.org/10.1016/j.eplepsyres.2020.106521
Li Y, Klena NT, Gabriel GC et al (2015) Global genetic analysis in mice unveils central role for cilia in congenital heart disease. Nature. https://doi.org/10.1038/nature14269
Liao Y, Wang J, Jaehnig EJ et al (2019) WebGestalt 2019: gene set analysis toolkit with revamped UIs and APIs. Nucleic Acids Res. https://doi.org/10.1093/nar/gkz401
Liao W, Lee K-Z, Chen J-C et al (2020) Deficiency of cyclin-dependent kinase-like 5 causes spontaneous seizures in neonatal mice. bioRxiv. https://doi.org/10.1101/2020.03.09.983981
Mirra V, Werner C, Santamaria F (2017) Primary ciliary dyskinesia: an update on clinical aspects, genetics, diagnosis, and future treatment strategies. Front Pediatr. https://doi.org/10.3389/fped.2017.00135
Mukherjee I, Roy S, Chakrabarti S (2019) Identification of important effector proteins in the FOXJ1 transcriptional network associated with ciliogenesis and ciliary function. Front Genet. https://doi.org/10.3389/fgene.2019.00023
Muthusamy N, Vijayakumar A, Cheng G, Ghashghaei HT (2014) A Knock-in Foxj1CreERT2: GFP mouse for recombination in epithelial cells with motile cilia. Genesis. https://doi.org/10.1002/dvg.22753
Negraes PD, Trujillo CA, Yu NK et al (2021) Altered network and rescue of human neurons derived from individuals with early-onset genetic epilepsy. Mol Psychiatry. https://doi.org/10.1038/s41380-021-01104-2
Noebels J (2015) Pathway-driven discovery of epilepsy genes. Nat Neurosci. https://doi.org/10.1038/nn.3933
Nuche-Berenguer B, Ramos-Álvarez I, Jensen RT (2016) The p21-activated kinase, PAK2, is important in the activation of numerous pancreatic acinar cell signaling cascades and in the onset of early pancreatitis events. Biochim Biophys Acta. https://doi.org/10.1016/j.bbadis.2016.02.008
Olson HE, Demarest ST, Pestana-Knight EM et al (2019) Cyclin-dependent kinase-like 5 deficiency disorder: clinical review. Pediatr Neurol. https://doi.org/10.1016/j.pediatrneurol.2019.02.015
Paz JT, Huguenard JR (2015) Microcircuits and their interactions in epilepsy: is the focus out of focus? Nat Neurosci. https://doi.org/10.1038/nn.3950
Pazour GJ, Agrin N, Leszyk J, Witman GB (2005) Proteomic analysis of a eukaryotic cilium. J Cell Biol. https://doi.org/10.1083/jcb.200504008
Rawlins EL, Ostrowski LE, Randell SH, Hogan BLM (2007) Lung development and repair: contribution of the ciliated lineage. Proc Natl Acad Sci USA. https://doi.org/10.1073/pnas.0610770104
Sattar S, Gleeson JG (2011) The ciliopathies in neuronal development: a clinical approach to investigation of Joubert syndrome and Joubert syndrome-related disorders. Dev Med Child Neurol. https://doi.org/10.1111/j.1469-8749.2011.04021.x
Schneider CA, Rasband WS, Eliceiri KW (2012) NIH image to imageJ: 25 years of image analysis HHS public access. Nat Methods. https://doi.org/10.1038/nmeth.2089
Shapiro AJ, Zariwala MA, Ferkol T et al (2016) Diagnosis, monitoring, and treatment of primary ciliary dyskinesia: PCD foundation consensus recommendations based on state of the art review. Pediatr Pulmonol. https://doi.org/10.1002/ppul.23304
Smith CL, Eppig JT (2012) The mammalian phenotype ontology as a unifying standard for experimental and high-throughput phenotyping data. Mamm Genome. https://doi.org/10.1007/s00335-012-9421-3
Smith CEL, Lake AVR, Johnson CA (2020) Primary cilia, ciliogenesis and the actin cytoskeleton: a little less resorption, a little more actin please. Front Cell Dev Biol. https://doi.org/10.3389/fcell.2020.622822
Staley K (2015) Molecular mechanisms of epilepsy. Nat Neurosci. https://doi.org/10.1038/nn.3947
Tam L-W, Ranum PT, Lefebvre PA (2013) CDKL5 regulates flagellar length and localizes to the base of the flagella in Chlamydomonas. Mol Biol Cell. https://doi.org/10.1091/mbc.e12-10-0718
Tang S, Wang I-TJ, Yue C et al (2017) Loss of CDKL5 in glutamatergic neurons disrupts hippocampal microcircuitry and leads to memory impairment in mice. J Neurosci. https://doi.org/10.1523/JNEUROSCI.0539-17.2017
Terzic B, Cui Y, Edmondson AC et al (2021) X-linked cellular mosaicism underlies age-dependent occurrence of seizure-like events in mouse models of CDKL5 deficiency disorder. Neurobiol Dis. https://doi.org/10.1016/j.nbd.2020.105176
Terzic B, Felicia Davatolhagh M, Ho Y et al (2021) Temporal manipulation of Cdkl5 reveals essential postdevelopmental functions and reversible CDKL5 deficiency disorder-related deficits. J Clin Investig. https://doi.org/10.1172/JCI143655
Tsukita S, Kunimoto K, Yamazaki Y et al (2012) Coordinated ciliary beating requires Odf2-mediated polarization of basal bodies via basal feet. Cell. https://doi.org/10.1016/j.cell.2011.10.052
Voss LJ, Sleigh JW, Barnard JPM, Kirsch HE (2008) The howling cortex: seizures and general anesthetic drugs. Anesth Analg. https://doi.org/10.1213/ane.0b013e3181852595
Wang ITJ, Allen M, Goffin D et al (2012) Loss of CDKL5 disrupts kinome profile and event-related potentials leading to autistic-like phenotypes in mice. Proc Natl Acad Sci USA. https://doi.org/10.1073/pnas.1216988110
Werner ME, Hwang P, Huisman F et al (2011) Actin and microtubules drive differential aspects of planar cell polarity in multiciliated cells. J Cell Biol. https://doi.org/10.1083/jcb.201106110
Yang J, Gao J, Adamian M et al (2005) The ciliary rootlet maintains long-term stability of sensory cilia. Mol Cell Biol. https://doi.org/10.1128/mcb.25.10.4129-4137.2005
Yoder SM, Dineen SL, Wang Z, Thurmond DC (2014) YES, a src family kinase, is a proximal glucose-specific activator of cell division cycle control protein 42 (Cdc42) in pancreatic islet β cells. J Biol Chem. https://doi.org/10.1074/jbc.M114.559328
Zuleta-Alarcon A, Castellon-Larios K, Moran K et al (2014) Anesthesia-related perioperative seizures: pathophysiology, predisposing factors and practical recommendations. Austin J Anesth Analg 2:1026
Acknowledgements
We thank Dr Pete Lefebvre for advising about the initial idea that CDD might be a motile ciliopathy. We thank Dr Joe Zhou and Dr Chay Kuo for generously providing Cdkl5fl/fl, Cdkl5y/fl and FOXJ1CreERT mice, and supporting with advice.
Funding
German Research Foundation grant FA 1457/1-1 (RJF); National Institutes of Health grant NIH HL142788 (CWL); National Institutes of Health grant NIH HL132024-01 (CWL); National Institutes of Health grant NIH GM051293 (SMK); MKG’s effort is supported by Department of Biomedical Informatics of University of Pittsburgh School of Medicine.
Author information
Authors and Affiliations
Contributions
Conceptualization: CWL, RJF; Methodology: CWL, RJF, SMK, YW, MKG; Data collection: RJF, MY; Recruitment of CDD patients: JG, NHB; Data analysis: RJF, VSC, MY; Computational Data analysis: RJF, YW, KBK; Visualization: RJF, TNF, YW, MKG, CWL; Funding acquisition: CWL, SMK, RJF; Project administration: CWL; Supervision: CWL, RJF; Writing—original draft: CWL, RJF; Writing—review and editing: CWL, RJF, SMK, YW, KBK, MKG.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Supplementary file2 (AVI 1914 KB)
Supplementary file3 (AVI 7974 KB)
Supplementary file4 (MOV 5459 KB)
Supplementary file5 (AVI 1701 KB)
Supplementary file6 (MP4 61788 KB)
Supplementary file7 (AVI 446 KB)
Supplementary file8 (MP4 76331 KB)
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Faubel, R.J., Santos Canellas, V.S., Gaesser, J. et al. Flow blockage disrupts cilia-driven fluid transport in the epileptic brain. Acta Neuropathol 144, 691–706 (2022). https://doi.org/10.1007/s00401-022-02463-y
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00401-022-02463-y