Abstract
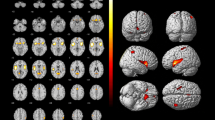
The aim of this study is to investigate regional cerebral blood flow (rCBF) in patients with syncope. We compared brain single photon emission computed tomography (SPECT) images of neurally mediated syncope patients with those of age/sex matched healthy volunteers. 99mTc-ethylcysteinate dimer (ECD) brain SPECT was performed in 35 patients (M/F = 17/18, mean 36.6 years) with syncope during the asymptomatic period, and in 35 healthy volunteers. For statistical parametric mapping (SPM) analysis, all SPECT images were spatially normalized to the standard SPECT template and then smoothed using a 14-mm full width at half maximum Gaussian kernel. The mean duration of syncope history was 4.9 years and the mean number of syncope episodes was 6.3. In all patients, syncope or presyncope episodes occurred during head-up tilt tests, and all were the vasodepressive type. SPM analysis of brain SPECT images showed significantly decreased rCBF in the right anterior insular cortex, left parahippocampal gyrus, bilateral fusiform gyri, bilateral middle and inferior temporal gyri, left lingual gyrus, bilateral precuneus and bilateral posterior lobes of the cerebellum in syncope patients at a false discovery rate corrected p < 0.05. There were no brain regions that showed increased rCBF in syncope patients. Furthermore, we found a negative correlation between the total number of syncopal episodes and the rCBF of the right prefrontal cortex, and between the duration of syncope history and the rCBF of the right cingulate gyrus at uncorrected p < 0.001. Decreases of rCBF in multiple brain regions may be responsible for autonomic dysregulation and improper processing of emotional stress in neurally mediated syncope patients, and frequent syncope episodes may lead to frontal dysfuction.


Similar content being viewed by others
References
Abboud FM (1993) Neurocardiogenic syncope. N Engl J Med 328:1117–1120
Amarenco P, Kase CS, Rosengart A, Pessin MS, Bousser MG, Caplan LR (1993) Very small (border zone) cerebellar infarcts. Distribution, causes, mechanisms and clinical features. Brain 116(Pt 1):161–186
Beebe DW, Gozal D (2002) Obstructive sleep apnea and the prefrontal cortex: towards a comprehensive model linking nocturnal upper airway obstruction to daytime cognitive and behavioral deficits. J Sleep Res 11:1–16
Buchel C, Morris J, Dolan RJ, Friston KJ (1998) Brain systems mediating aversive conditioning: an event-related fMRI study. Neuron 20:947–957
Cavanna AE (2007) The precuneus and consciousness. CNS Spectr 12:545–552
Chang LT (1978) A method for attenuation correction in radionuclide computed tomography. IEEE Trans Nucl Sci NS-25:638–643
Colivicchi F, Bassi A, Santini M, Caltagirone C (2004) Cardiac autonomic derangement and arrhythmias in right-sided stroke with insular involvement. Stroke 35:2094–2098
Critchley HD (2005) Neural mechanisms of autonomic, affective, and cognitive integration. J Comp Neurol 493:154–166
Dan D, Hoag JB, Ellenbogen KA, Wood MA, Eckberg DL, Gilligan DM (2002) Cerebral blood flow velocity declines before arterial pressure in patients with orthostatic vasovagal presyncope. J Am Coll Cardiol 39:1039–1045
Devinsky O, Morrell MJ, Vogt BA (1995) Contributions of anterior cingulate cortex to behaviour. Brain 118(Pt 1):279–306
Duvernoy HM (1991) The Human brain: surface, three-dimensional sectional anatomy with MRI, and blood supply. Springer-Verlag, New York
Franco Folino A (2007) Cerebral autoregulation and syncope. Prog Cardiovasc Dis 50:49–80
Friston KJ, Holmes AP, Worsley KJ, Poline JP, Frith CD, Frackowiak RSJ (1995) Statistical parametric maps in functional imaging: a general linear approach. Hum Brain Mapp 2:189–210
Han BI, Oh HJ, Bang OY, Lee JH (2006) Successful treatment of vasovagal syncope due to blood-injury phobia by physical maneuvering. J Clin Neurol 2:66–69
Hermann A, Schafer A, Walter B, Stark R, Vaitl D, Schienle A (2007) Diminished medial prefrontal cortex activity in blood-injection-injury phobia. Biol Psychol 75:124–130
Ignacak ML, Harbaugh SV, Dayyat E, Row BW, Gozal D, Czyzyk-Krzeska MF (2009) Intermittent hypoxia regulates RNA polymerase II in hippocampus and prefrontal cortex. Neuroscience 158:1436–1445
Ilgin N, Olgunturk R, Kula S, Turan L, Tunaoglu S, Temiz H, Gokcora N, Gucuyener K (2005) Brain perfusion assessed by 99 mTc-ECD SPECT imaging in pediatric patients with neurally mediated reflex syncope. Pacing Clin Electrophysiol 28:534–539
Insausti R, Amaral DG, Cowan WM (1987) The entorhinal cortex of the monkey: II. Cortical afferents. J Comp Neurol 264:356–395
Joo EY, Hong SB, Han HJ, Tae WS, Kim JH, Han SJ, Seo DW, Lee KH, Hong SC, Lee M, Kim S, Kim BT (2005) Postoperative alteration of cerebral glucose metabolism in mesial temporal lobe epilepsy. Brain 128:1802–1810
Joo EY, Tae WS, Kim JH, Kim BT, Hong SB (2004) Glucose hypometabolism of hypothalamus and thalamus in narcolepsy. Ann Neurol 56:437–440
Joo EY, Tae WS, Lee MJ, Kang JW, Park HS, Lee JY, Suh M, Hong SB (2010) Reduced brain gray matter concentration in patients with obstructive sleep apnea syndrome. Sleep 33:235–241
Lagi A, Tamburini C, Cipriani M, Fattorini L (1997) Vagal control of heart rate variability in vasovagal syncope: studies based on 24-h electrocardiogram recordings. Clin Auton Res 7:127–130
Lazzeri C, La Villa G, Barletta G, Franchi F (2000) 24-hour heart rate variability in patients with vasovagal syncope. Pacing Clin Electrophysiol 23:463–468
Lempert T, Bauer M, Schmidt D (1994) Syncope: a videometric analysis of 56 episodes of transient cerebral hypoxia. Ann Neurol 36:233–237
McNally KA, Paige AL, Varghese G, Zhang H, Novotny EJ Jr, Spencer SS, Zubal IG, Blumenfeld H (2005) Localizing value of ictal-interictal SPECT analyzed by SPM (ISAS). Epilepsia 46:1450–1464
Odano I, Takahashi N, Higuchi T, Ohkubo M, Hama S, Tsuda A, Otaki H, Noguchi E, Hatano M (1995) Evaluation of cerebral blood flow in patients with idiopathic orthostatic hypotension using Tc-99 m HMPAO brain SPECT during postural testing. Clin Nucl Med 20:807–809
Olgunturk R, Turan L, Tunaoglu FS, Kula S, Gokcora N, Karabacak NI, Azizoglu F (2003) Abnormality of the left ventricular sympathetic nervous function assessed by I-123 metaiodobenzylguanidine imaging in pediatric patients with neurocardiogenic syncope. Pacing Clin Electrophysiol 26:1926–1930
Oppenheimer SM, Gelb A, Girvin JP, Hachinski VC (1992) Cardiovascular effects of human insular cortex stimulation. Neurology 42:1727–1732
Oppenheimer SM, Kedem G, Martin WM (1996) Left-insular cortex lesions perturb cardiac autonomic tone in humans. Clin Auton Res 6:131–140
Phillips ML, Young AW, Senior C, Brammer M, Andrew C, Calder AJ, Bullmore ET, Perrett DI, Rowland D, Williams SC, Gray JA, David AS (1997) A specific neural substrate for perceiving facial expressions of disgust. Nature 389:495–498
Reinhard M, Waldkircher Z, Timmer J, Weiller C, Hetzel A (2008) Cerebellar autoregulation dynamics in humans. J Cereb Blood Flow Metab 28:1605–1612
Sealey B, Lui K (2004) Diagnosis and management of vasovagal syncope and dysautonomia. AACN Clin Issues. 15:462–477
Spence JS, Carmack PS, Gunst RF, Schucany WR, Woodward WA, Haley RW (2006) Using a white matter reference to remove the dependency of global signal on experimental conditions in SPECT analyses. Neuroimage 32:49–53
Toyry JP, Kuikka JT, Lansimies EA (1997) Regional cerebral perfusion in cardiovascular reflex syncope. Eur J Nucl Med 24:215–218
Vogt BA, Laureys S (2005) Posterior cingulate, precuneal and retrosplenial cortices: cytology and components of the neural network correlates of consciousness. Prog Brain Res 150:205–217
Zamrini EY, Meador KJ, Loring DW, Nichols FT, Lee GP, Figueroa RE, Thompson WO (1990) Unilateral cerebral inactivation produces differential left/right heart rate responses. Neurology 40:1408–1411
Zhang Z, Oppenheimer SM (1997) Characterization, distribution and lateralization of baroreceptor-related neurons in the rat insular cortex. Brain Res 760:243–250
Acknowledgments
This study was supported by a Grant (2010K000817) from Brain Research Center of the 21st Century Frontier Research Program funded by the Ministry of Education, Science and Technology, and by a grant of the Korean Health Technology R&D Project, Ministry for Health, Welfare & Family Affairs, Republic of Korea (No. A090579).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Joo, E.Y., Hong, S.B., Lee, M. et al. Cerebral blood flow abnormalities in patients with neurally mediated syncope. J Neurol 258, 366–372 (2011). https://doi.org/10.1007/s00415-010-5759-1
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00415-010-5759-1