Abstract
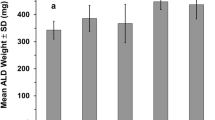
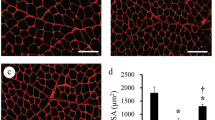
Cellular responses in the compensatory hypertrophied (plantaris) muscle induced by surgical ablation of synergistic muscles (soleus and gastrocnemius) were determined during 10-week anabolic androgenic steroid (AAS) treatment. Adult Wistar male rats were divided randomly into the Control and Steroid groups, and contralateral surgery was performed. Nandrolone decanoate was administered to the Steroid group. [3H]thymidine and [14C]leucine labeling were used to determine the serial changes in cellular mitotic activity and amino acid uptake. Myogenic cells and cellular responses in blood vessels and nerve fibers were analyzed by immunohistochemistry. Significantly lower cellular mitotic activity associated with lower volume of muscle fiber necrosis was observed in the Steroid group during the first week. However, amino acid uptake and final muscle wet weight gain did not differ between the groups. Marked activation/proliferation of muscular, vascular, and peripheral nerve-related cells was seen with the inflammatory responses in both groups. However, this activation was dependent on the volume of muscle fiber damage and was not preferentially accelerated by AAS loading. These results indicated that AAS loading significantly diminished muscle fiber damages, but they did not accelerate final muscle wet weight gain and activation of myogenic, vascular, and peripheral nerve related cells in the compensatory enlarged muscles.







Similar content being viewed by others
References
Allbrook D (1981) Skeletal muscle regeneration. Muscle Nerve 4:234–245
Armstrong RB, Marum P, Tullson P, Saubert CW (1979) Acute hypertrophic response of skeletal muscle to removal of synergists. J Appl Physiol 46:835–842
Boissonneault G, Gagnon J, Ho-Kim MA, Tremblay RR (1987) Lack of effect of anabolic steroids on specific mRNAs of skeletal muscle undergoing compensatory hypertrophy. Mol Cell Endocrinol 51:19–24
D’Andrea A, Caso P, Salerno G, Scarafile R, De Corato G, Mita C, Di Salvo G, Severino S, Cuomo S, Liccardo B, Esposito N, Calabro R (2007) Left ventricular early myocardial dysfunction after chronic misuse of anabolic androgenic steroids: a Doppler myocardial and strain imaging analysis. Br J Sports Med 41:149–155
Griggs RC, Kingston W, Jozefowicz RF, Herr BE, Forbes G, Halliday D (1989) Effect of testosterone on muscle mass and muscle protein synthesis. J Appl Physiol 66:498–503
Haupt H, Rovere G (1984) Anabolic steroids: a review of the literature. Am J Sports Med 12:469–484
Hausmann R, Hammer S, Betz P (1998) Performance enhancing drugs (doping agents) and sudden death—a case report and review of the literature. Int J Legal Med 111:261–264
Johansson P, Ray A, Zhou Q, Huang W, Karlsson K, Nyberg F (1997) Anabolic androgenic steroids increase beta-endorphin levels in the ventral tegmental area in the male rat brain. Neurosci Res 27:185–189
Johansson P, Hallberg M, Kindlundh A, Nyberg F (2000) The effect on opioid peptides in the rat brain, after chronic treatment with the anabolic androgenic steroid, nandrolone decanoate. Brain Res Bull 51:413–418
Le Greves P, Huang W, Johansson P, Thonwall M, Zhou Q, Nyberg F (1997) Effects of an anabolic-androgenic steroid on the regulation of the NMDA receptor NR1, NR2A and NR2B subunit mRNAs in brain regions of the male rat. Neurosci Lett 226:61–64
McCall GE, Allen DL, Linderman JK, Grindeland RE, Roy RR, Mukku VR, Edgerton VR (1998) Maintenance of myonuclear domain size in rat soleus after overload and growth hormone/IGF-I treatment. J Appl Physiol 84:1407–1412
Overbeek GA, de Visser J (1961) A comparison of the myotrophic and androgenic activities of the phenylpropionates and decanoates of testosterone and nandrolone. Acta Endocrinol (Copenh) 38:285–292
Pope HJ, Katz D (1994) Psychiatric and medical effects of anabolic-androgenic steroid use. A controlled study of 160 athletes. Arch Gen Psychiatry 51:375–382
Roy RR, Monke SR, Allen DL, Edgerton VR (1999) Modulation of myonuclear number in functionally overloaded and exercised rat plantaris fibers. J Appl Physiol 87:634–642
Spath JA, Lefer A (1975) Effects of dexamethasone on myocardial cells in the early phase of acute myocardial infarction. Am Heart J 90:50–55
Spath JJ, Lane D, Lefer A (1974) Protective action of methylprednisolone on the myocardium during experimental myocardial ischemia in the cat. Circ Res 35:44–51
Swanson LW (1987) The hypothalamus. In: Bjorklund A, Hokfelt T, Swanson LW (eds) Handbook of chemical neuroanatomy, integrated systems of the CNS, part 1: hypothalamus, hippocampus, amygdala, retina, vol 5. Elsevier, Amsterdam, pp 1–24
Tamaki T, Shiraishi T (1996) Characteristics of compensatory hypertrophied muscle in the rat: II. Comparison of histochemical and functional properties. Anat Rec 246:335–342
Tamaki T, Akatsuka A, Tokunaga M, Uchiyama S, Shiraishi T (1996) Characteristics of compensatory hypertrophied muscle in the rat: I. Electron microscopic and immunohistochemical studies. Anat Rec 246:325–334
Tamaki T, Akatsuka A, Tokunaga M, Ishige K, Uchiyama S, Shiraishi T (1997) Morphological and biochemical evidence of muscle hyperplasia following weight-lifting exercise in rats. Am J Physiol 273:C246–C256
Tamaki T, Uchiyama S, Uchiyama Y, Akatsuka A, Roy RR, Edgerton VR (2001) Anabolic steroids increase exercise tolerance. Am J Physiol Endocrinol Metab 280:E973–E981
Tamaki T, Akatsuka A, Ando K, Nakamura Y, Matsuzawa H, Hotta T, Roy RR, Edgerton VR (2002a) Identification of myogenic-endothelial progenitor cells in the interstitial spaces of skeletal muscle. J Cell Biol 157:571–577
Tamaki T, Akatsuka A, Yoshimura S, Roy RR, Edgerton VR (2002b) New fiber formation in the interstitial spaces of rat skeletal muscle during postnatal growth. J Histochem Cytochem 50:1097–1111
Tamaki T, Shiraishi T, Takeda H, Matsumiya T, Roy RR, Edgerton VR (2003a) Nandrolone decanoate enhances hypothalamic biogenic amines in rats. Med Sci Sports Exerc 35:32–38
Tamaki T, Akatsuka A, Okada Y, Matsuzaki Y, Okano H, Kimura M (2003b) Growth and differentiation potential of main- and side-population cells derived from murine skeletal muscle. Exp Cell Res 291:83–90
Tamaki T, Uchiyama Y, Okada Y, Ishikawa T, Sato M, Akatsuka A, Asahara T (2005) Functional recovery of damaged skeletal muscle through synchronized vasculogenesis, myogenesis, and neurogenesis by muscle-derived stem cells. Circulation 112:2857–2866
Tamaki T, Okada Y, Uchiyama Y, Tono K, Masuda M, Wada M, Hoshi A, Ishikawa T, Akatsuka A (2007a) Clonal multipotency of skeletal muscle-derived stem cells between mesodermal and ectodermal lineage. Stem Cells 25:2283–2290
Tamaki T, Okada Y, Uchiyama Y, Tono K, Masuda M, Wada M, Hoshi A, Akatsuka A (2007b) Synchronized reconstitution of muscle fibers, peripheral nerves and blood vessels by murine skeletal muscle-derived CD34(−)/45(−) cells. Histochem Cell Biol 128:349–360
Tamaki T, Akatsuka A, Okada Y, Uchiyama Y, Tono K, Wada M, Hoshi A, Iwaguro H, Iwasaki H, Oyamada A, Asahara T (2008a) Cardiomyocyte formation by skeletal muscle-derived multi-myogenic stem cells after transplantation into infarcted myocardium. PLoS ONE 3:e1789
Tamaki T, Okada Y, Uchiyama Y, Tono K, Masuda M, Nitta M, Hoshi A, Akatsuka A (2008b) Skeletal muscle-derived CD34+/45− and CD34−/45− stem cells are situated hierarchically upstream of Pax7+ cells. Stem Cells Dev 17:653–667
Timson BF (1990) Evaluation of animal models for the study of exercise-induced muscle enlargement. J Appl Physiol 69:1935–1945
Wilson JD (1988) Androgen abuse by athletes. Endocr Rev 9:181–199
Acknowledgment
This work was supported by a Grant-in-Aid for Scientific Research (B-12480012) from the Ministry of Education, Science and Culture of Japan.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Tamaki, T., Uchiyama, Y., Okada, Y. et al. Anabolic-androgenic steroid does not enhance compensatory muscle hypertrophy but significantly diminish muscle damages in the rat surgical ablation model. Histochem Cell Biol 132, 71–81 (2009). https://doi.org/10.1007/s00418-009-0584-2
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00418-009-0584-2