Abstract
Purpose
The Na+, K+-ATPase (NKA) is important in regulating trans-membrane ion gradients, cellular excitability and muscle function. We investigated the effects of resistance training in healthy young adults on the adaptability of NKA content and of the specific α and β isoforms in human skeletal muscle.
Methods
Twenty-one healthy young males (22.9 ± 4.6 year; 1.80 ± 0.70 m, 85.1 ± 17.8 kg, mean ± SD) underwent 7 weeks of resistance training, training three times per week (RT, n = 16) or control (CON, n = 5). The training program was effective with a 39% gain in leg press muscle strength (p = 0.001). A resting vastus lateralis muscle biopsy was taken before and following RT or CON and assayed for NKA content ([3H]ouabain binding site content) and NKA isoform (α1, α2, β1, β2) abundances.
Results
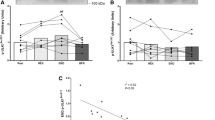
After RT, each of NKA content (12%, 311 ± 76 vs 349 ± 76 pmol g wet weight−1, p = 0.01), NKA α1 (32%, p = 0.01) and α2 (10%, p < 0.01) isoforms were increased, whereas β1 (p = 0.18) and β2 (p = 0.22) isoforms were unchanged. NKA content and isoform abundances were unchanged during CON.
Conclusions
Resistance training increased muscle NKA content through upregulation of both α1 and α2 isoforms, which were independent of β isoform changes. In animal models, modulations in α1 and α2 isoform abundances in skeletal muscle may affect fatigue resistance during exercise, muscle hypertrophy and strength. Whether similar in-vivo functional benefits of these NKA isoform adaptations occurs in human muscle with resistance training remains to be determined.




Similar content being viewed by others
Abbreviations
- 1RM:
-
One repetition maximum
- α:
-
Alpha subunit
- β:
-
Beta subunit
- CWI:
-
Coldwater immersion
- K+ :
-
Potassium ion
- [K+]:
-
Potassium ion concentration
- Na+ :
-
Sodium-ion
- [Na+]:
-
Sodium ion concentration
- NKA:
-
Na+, K+-ATPase
- RT:
-
Resistance training
- [3H]ouabain binding:
-
Tritiated ouabain binding
References
Aughey RJ, Murphy KT, Clark SA, Garnham AP, Snow RJ, Cameron-Smith D, Hawley JA, McKenna MJ (2007) Muscle Na+-K+-ATPase activity and isoform adaptations to intense interval exercise and training in well-trained athletes. J Appl Physiol 103(1):39–47
Benziane B, Widegren U, Pirkmajer S, Henriksson J, Stepto NK, Chibalin AV (2011) Effect of exercise and training on phospholemman phosphorylation in human skeletal muscle. Am J Physiol Endocrinol Metab 301(3):E456–E466
Candow DG, Burke DG (2007) Effect of short-term equal-volume resistance training with different workout frequency on muscle mass and strength in untrained men and women. J Strength Cond Res 21(1):204
Christiansen D, Bishop DJ, Broatch JR, Bangsbo J, McKenna MJ, Murphy RM (2018) Cold-water immersion after training sessions: effects on fiber type-specific adaptations in muscle K+ transport proteins to sprint-interval training in men. J Appl Physiol 125(2):429–444
Clausen T (2003) Na+-K+ pump regulation and skeletal muscle contractility. Physiol Rev 83(4):1269–1324
Clausen T (2010) Hormonal and pharmacological modification of plasma potassium homeostasis. Fundam Clin Pharmacol 24(5):595–605
Clausen T (2013) Quantification of Na+, K+ pumps and their transport rate in skeletal muscle: functional significance. J Gen Physiol 142(4):327–345. https://doi.org/10.1085/jgp.201310980
Cohen JW (1988) Statistical power analysis for the behavioral sciences, 2nd edn. Lawrence Erlbaum Associates, Hillsdale
Dela F, Holten M, Juel C (2004) Effect of resistance training on Na, K pump and Na+/H+ exchange protein densities in muscle from control and patients with type 2 diabetes. Pflügers Arch 447(6):928–933
DiFranco M, Hakimjavadi H, Lingrel JB, Heiny JA (2015) Na, K-ATPase α2 activity in mammalian skeletal muscle T-tubules is acutely stimulated by extracellular K+. J Gen Physiol JGP 201511407
Evertsen F, Medbo J, Jebens E, Nicolaysen K (1997) Hard training for 5 mo increases Na (+)-K+ pump concentration in skeletal muscle of cross-country skiers. Am J Physiol Regul Integr Comp Physiol 272(5):R1417–R1424
Fyfe JJ, Broatch JR, Trewin AJ, Hanson ED, Argus CK, Garnham AP, Petersen AC (2019) Cold water immersion attenuates anabolic signaling and skeletal muscle fiber hypertrophy, but not strength gain, following whole-body resistance training. J Appl Physiol 127(5):1403–1418
Green H, Dahly A, Shoemaker K, Goreham C, Bombardier E, Ball-Burnett M (1999a) Serial effects of high-resistance and prolonged endurance training on Na+-K+ pump concentration and enzymatic activities in human vastus lateralis. Acta Physiol Scand 165(2):177–184
Green H, Goreham C, Ouyang J, Ball-Burnett M, Ranney D (1999b) Regulation of fiber size, oxidative potential, and capillarization in human muscle by resistance exercise. Am J Physiol Regul Integr Comp Physiol 276(2):R591–R596
Green H, Barr D, Fowles J, Sandiford S, Ouyang J (2004) Malleability of human skeletal muscle Na+-K+-ATPase pump with short-term training. J Appl Physiol 97(1):143–148
Green H, Duhamel T, Stewart R, Tupling AR, Ouyang J (2008) Dissociation between changes in muscle Na+-K+-ATPase isoform abundance and activity with consecutive days of exercise and recovery. Am J Physiol Endocrinol Metab 294(4):E761–E767
Hansen O (2001) The α1 isoform of Na+, K+-ATPase in rat soleus and extensor digitorum longus. Acta Physiol Scand 173(3):335–341
Hansen O, Clausen T (1988) Quantitative determination of Na+-K+-ATPase and other sarcolemmal components in muscle cells. Am J Physiol Cell Physiol 254(1):C1–C7
Harmer AR, Ruell PA, McKenna MJ, Chisholm DJ, Hunter SK, Thom JM, Morris NR, Flack JR (2006) Effects of sprint training on extrarenal potassium regulation with intense exercise in type 1 diabetes. J Appl Physiol 100(1):26–34
He S, Shelly DA, Moseley AE, James PF, James JH, Paul RJ, Lingrel JB (2001) The α1-and α2-isoforms of Na-K-ATPase play different roles in skeletal muscle contractility. Am J Physiol Regul Integr Comp Physiol 281(3):R917–R925
Klitgaard H, Clausen T (1989) Increased total concentration of Na+, K+-pumps in vastus lateralis muscle of old trained human subjects. J Appl Physiol 67(6):2491–2494
Kutz LC, Mukherji ST, Wang X, Bryant A, Larre I, Heiny JA, Lingrel JB, Pierre SV, Xie Z (2018) Isoform-specific role of Na/K-ATPase α1 in skeletal muscle. Am J Physiol Endocrinol Metab 314(6):E620–E629. https://doi.org/10.1152/ajpendo.00275.2017
Lavoie L, Levenson R, Martin-Vasallo P, Klip A (1997) The molar ratios of α and β subunits of the Na+–K+-ATPase differ in distinct subcellular membranes from rat skeletal muscle. Biochemistry 36(25):7726–7732
Lingrel J, Moseley A, Dostanic I, Cougnon M, He S, James P, Woo A, O’Connor K, Neumann J (2003) Functional roles of the α isoforms of the Na, K-ATPase. Ann N Y Acad Sci 986(1):354–359
Madsen K, Franch J, Clausen T (1994) Effects of intensified endurance training on the concentration of Na, K-ATPase and Ca-ATPase in human skeletal muscle. Acta Physiol Scand 150(3):251–258
McKenna M, Schmidt T, Hargreaves M, Cameron L, Skinner S, Kjeldsen K (1993) Sprint training increases human skeletal muscle Na+-K+-ATPase concentration and improves K+ regulation. J Appl Physiol 75(1):173–180
Medbo JI, Jebens E, Vikne H, Refsnes PE, Gramvik P (2001) Effect of strenuous strength training on the Na+, K+-pump concentration in skeletal muscle of well-trained men. Eur J Appl Physiol 84(1–2):148–154
Murphy RM, Lamb GD (2013) Important considerations for protein analyses using antibody based techniques: down-sizing Western blotting up-sizes outcomes. J Physiol 591(23):5823–5831
Murphy KT, Snow RJ, Petersen AC, Murphy RM, Mollica J, Lee J-S, Garnham AP, Aughey RJ, Leppik JA, Medved I (2004) Intense exercise up-regulates Na+, K+-ATPase isoform mRNA, but not protein expression in human skeletal muscle. J Physiol 556(2):507–519
Nielsen JJ, Mohr M, Klarskov C, Kristensen M, Krustrup P, Juel C, Bangsbo J (2004) Effects of high-intensity intermittent training on potassium kinetics and performance in human skeletal muscle. J Physiol 554(3):857–870. https://doi.org/10.1113/jphysiol.2003.050658
Nørgaard A, Kjeldsen K, Clausen T (1984) A method for the determination of the total number of 3H-ouabain binding sites in biopsies of human skeletal muscle. Scand J Clin Lab Invest 44(6):509–518
Perry BD, Levinger P, Serpiello FR, Caldow MK, Cameron-Smith D, Bartlett JR, Feller JA, Bergman NR, Levinger I, McKenna MJ (2013) The effects of osteoarthritis and age on skeletal muscle strength, Na+-K+-ATPase content, gene and isoform expression. J Appl Physiol 115(10):1443–1449
Perry BD, Wyckelsma VL, Murphy RM, Steward CH, Anderson M, Levinger I, Petersen AC, McKenna MJ (2016) Dissociation between short-term unloading and resistance training effects on skeletal muscle Na+, K+-ATPase, muscle function, and fatigue in humans. J Appl Physiol 121(5):1074–1086. https://doi.org/10.1152/japplphysiol.00558.2016
Petersen AC, Murphy KT, Snow RJ, Leppik JA, Aughey RJ, Garnham AP, Cameron-Smith D, McKenna MJ (2005) Depressed Na+-K+-ATPase activity in skeletal muscle at fatigue is correlated with increased Na+-K+-ATPase mRNA expression following intense exercise. Am J Physiol Regul Integr Comp Physiol 289(1):266–274
Radzyukevich TL, Neumann JC, Rindler TN, Oshiro N, Goldhamer DJ, Lingrel JB, Heiny JA (2013) Tissue-specific role of the Na, K-ATPase α2 isozyme in skeletal muscle. J Biol Chem 288(2):1226–1237
Sejersted OM, Sjogaard G (2000) Dynamics and consequences of potassium shifts in skeletal muscle and heart during exercise. Physiol Rev 80(4):1411–1481
Skovgaard C, Christensen PM, Larsen S, Andersen TR, Thomassen M, Bangsbo J (2014) Concurrent speed endurance and resistance training improves performance, running economy, and muscle NHE1 in moderately trained runners. J Appl Physiol 117(10):1097–1109
Staron RS, Hagerman FC, Hikida RS, Murray TF, Hostler DP, Crill MT, Ragg KE, Toma K (2000) Fiber type composition of the vastus lateralis muscle of young men and women. J Histochem Cytochem 48(5):623–629
Vorup J, Tybirk J, Gunnarsson TP, Ravnholt T, Dalsgaard S, Bangsbo J (2016) Effect of speed endurance and strength training on performance, running economy and muscular adaptations in endurance-trained runners. Eur J Appl Physiol 116(7):1331–1341. https://doi.org/10.1007/s00421-016-3356-4
Wang J, Velotta JB, McDonough AA, Farley RA (2001) All human Na+-K+-ATPase α-subunit isoforms have a similar affinity for cardiac glycosides. Am J Physiol Cell Physiol 281(4):C1336–C1343
Wyckelsma VL, McKenna MJ, Serpiello FR, Lamboley CR, Aughey RJ, Stepto NK, Bishop DJ, Murphy RM (2015) Single-fiber expression and fiber-specific adaptability to short-term intense exercise training of Na+-K+-ATPase α-and β-isoforms in human skeletal muscle. J Appl Physiol 118(6):699–706
Wyckelsma VL, Levinger I, Murphy RM, Petersen AC, Perry BD, Hedges CP, Anderson MJ, McKenna MJ (2017) Intense interval training in healthy older adults increases skeletal muscle [3H]ouabain-binding site content and elevates Na+, K+-ATPase α2 isoform abundance in type II fibers. Physiol Rep. https://doi.org/10.14814/phy2.13219
Wyckelsma VL, Perry BD, Bangsbo J, McKenna MJ (2019) Inactivity and exercise training differentially regulate abundance of Na+-K+-ATPase in human skeletal muscle. J Appl Physiol 127(4):905–920
Author information
Authors and Affiliations
Contributions
MMA, ACP, TF, JRB, SLH, DJB, and MJM edited and conceived and designed research. ACP, TF, JRB, SLH, APG performed experiments. MMA analyzed data. MMA and MJM interpreted results of experiments. MA prepared figures. MMA and MJM drafted the manuscript. MMA, ACP, TF, JRB, SLH, DJB and MJM edited and revised manuscript. MMA, ACP, TF, JRB, SLH, BJB edited and MJM approved the final version of the manuscript.
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare that they have no conflict of interest.
Additional information
Communicated by Michael Lindinger.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Altarawneh, M.M., Petersen, A.C., Farr, T. et al. Resistance training upregulates skeletal muscle Na+, K+-ATPase content, with elevations in both α1 and α2, but not β isoforms. Eur J Appl Physiol 120, 1777–1785 (2020). https://doi.org/10.1007/s00421-020-04408-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00421-020-04408-3