Abstract
Main conclusion
The wheat dehydrin (DHN-5) gives birth to salinity tolerance to transgenic Arabidopsis plants by the regulation of proline metabolism and the ROS scavenging system.
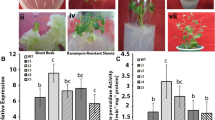
Dehydrins (DHNs) are involved in plant abiotic stress tolerance. In this study, we reported that salt tolerance of transgenic Arabidopsis plants overexpressing durum wheat dehydrin (DHN-5) was closely related to the activation of the proline metabolism enzyme (P5CS) and some antioxidant biocatalysts. Indeed, DHN-5 improved P5CS activity in the transgenic plants generating a significant proline accumulation. Moreover, salt tolerance of Arabidopsis transgenic plants was accompanied by an excellent activation of antioxidant enzymes like catalase (CAT), superoxide dismutase (SOD) and peroxide dismutase (POD) and generation of a lower level of hydrogen peroxide (H2O2) in leaves compared to the wild-type plants. The enzyme activities were enhanced in these transgenic plants in the presence of exogenous proline. Nevertheless, proline accumulation was slightly reduced in transgenic plants promoting chlorophyll levels. All these results suggest the crucial role of DHN-5 in response to salt stress through the activation of enzymes implicated in proline metabolism and in ROS scavenging enzymes.




Similar content being viewed by others
References
Arnold S, Curtiss A, Dean DH, Alzate O (2001) The role of a proline-induced broken-helix motif in alpha-helix 2 of Bacillus thuringiensis delta-endotoxins. FEBS Lett 490:70–74
Bowler C, Van Camp W, Van Montagu M, Inzé D, Asada K (1994) Superoxide dismutase in plants. Crit Rev Plant Sci 13:199–218
Bradford MM (1976) A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem 72:248–254
Brini F, Hanin M, Lumbreras V, Amara I, Khoudi H, Hassairi A, Pages M, Masmoudi K (2007a) Overexpression of wheat dehydrin DHN-5 enhances tolerance to salt and osmotic stress in Arabidopsis thaliana. Plant Cell Rep 26:2017–2026
Brini F, Hanin M, Mezghani I, Berkowitz GA, Masmoudi K (2007b) Overexpression of wheat Na+/H+ antiporter TNHX1 and H+-pyrophosphatase TVP1 improve salt- and drought-stress tolerance in Arabidopsis thaliana plants. J Exp Bot 58:301–308
Brini F, Saibi W, Amara I, Gargouri A, Masmoudi K, Hanin M (2010) Wheat dehydrin DHN-5 exerts a heat-protective effect on beta-glucosidase and glucose oxidase activities. Biosci Biotechnol Biochem 74:1050–1054
Brini F, Yamamoto A, Jlaiel L, Takeda S, Hobo T, Dinh HQ, Hattori T, Masmoudi K, Hanin M (2011) Pleiotropic effects of the wheat dehydrin DHN-5 on stress responses in Arabidopsis. Plant Cell Physiol 52:676–688
Chandler PM, Robertson M (1994) Gene expression regulated by abscisic acid and its relation to stress tolerance. Annu Rev Plant Biol 45:113–141
Dat JF, Lopez-Delgado H, Foyer CH, Scott IM (1998) Parallel changes in H2O2 and catalase during thermotolerance induced by salicylic acid or heat acclimation in mustard seedlings. Plant Physiol 116:1351–1357
Dimitrova MS, Tishinova V, Velcheva V (1994) Combined effect of zinc and lead on the hepatic superoxide dismutase-catalase system in carp (Cyprinus carpio). Comp Biochem Physiol C: Pharmacol Toxicol Endocrinol 108:43–46
Drira M, Saibi W, Brini F, Gargouri A, Masmoudi K, Hanin M (2013) The K-segments of the wheat dehydrin DHN-5 are essential for the protection of lactate dehydrogenase and β-glucosidase activities in vitro. Mol Biotechnol 54:643–650
Drira M, Saibi W, Amara I, Masmoudi K, Hanin M, Brini F (2015) Wheat dehydrin K-segments ensure bacterial stress tolerance, antiaggregation and antimicrobial effects. Appl Biochem Biotechnol 175:3310–3321
Ferrie A, Bethune T, Arganosa G, Waterer D (2011) Field evaluation of doubled haploid plants in the Apiaceae: dill (Anethum graveolens L.), caraway (Carum carvi L.), and fennel (Foeniculum vulgare Mill.). Plant Cell. Tissue Organ Culture (PCTOC) 104:407–413
Hanin M, Brini F, Ebel C, Toda Y, Takeda S, Masmoudi K (2011) Plant dehydrins and stress tolerance: versatile proteins for complex mechanisms. Plant Sign Behav 6:1503–1509
Hayzer D, Leisinger T (1980) The gene-enzyme relationships of proline biosynthesis in Escherichia coli. J Gen Microbiol 118:287–293
Ito Y, Katsura K, Maruyama K, Taji T, Kobayashi M, Seki M, Shinozaki K, Yamaguchi-Shinozaki K (2006) Functional analysis of rice DREB1/CBF-type transcription factors involved in cold-responsive gene expression in transgenic rice. Plant Cell Physiol 47:141–153
Khedr AHA, Abbas MA, Wahid AAA, Quick WP, Abogadallah GM (2003) Proline induces the expression of salt-stress-responsive proteins and may improve the adaptation of Pancratium maritimum L. to salt-stress. J Exp Bot 54:2553–2562
Maehly A, Chance B (1954) Catalases and peroxidases. Methods Biochem Anal 1:357–424
Marklund S, Marklund G (1974) Involvement of the superoxide anion radical in the autoxidation of pyrogallol and a convenient assay for superoxide dismutase. Eur J Biochem 47:469–474
Mhamdi A, Queval G, Chaouch S, Vanderauwera S, Van Breusegem F, Noctor G (2010) Catalase function in plants: a focus on Arabidopsis mutants as stress-mimic models. J Exp Botany: erq282
Mundy J, Chua NH (1988) Abscisic acid and water-stress induce the expression of a novel rice gene. EMBO J 7:2279–2286
Murashige T, Skoog F (1962) A revised medium for rapid growth and bio assays with tobacco tissue cultures. Physiol Plant 15:473–497
Rejeb KB, Abdelly C, Savouré A (2014) How reactive oxygen species and proline face stress together. Plant Physiol Biochem 80:278–284
Rorat T (2006) Plant dehydrins—tissue location, structure and function. Cell Mol Biol Lett 11:536–556
Saibi W, Abdeljalil S, Masmoudi K, Gargouri A (2012) Biocatalysts: beautiful creatures. Biochem Biophy Res Commun 426:289–293
Saibi W, Drira M, Yacoubi I, Feki K, Brini F (2015) Empiric, structural and in silico findings give birth to plausible explanations for the multifunctionality of the wheat dehydrin (DHN-5). Acta Physiol Plant 37
Sleimi N, Guerfali S, Bankaji I (2015) Biochemical indicators of salt stress in Plantago maritima: implications for environmental stress assessment. Ecol Ind 48:570–577
Szabados L, Savoure A (2010) Proline: a multifunctional amino acid. Trends Plant Sci 15:89–97
Thomashow MF (1999) Plant cold acclimation: freezing tolerance genes and regulatory mechanisms. Annu Rev Plant Biol 50:571–599
Velikova V, Yordanov I, Edreva A (2000) Oxidative stress and some antioxidant systems in acid rain-treated bean plants: protective role of exogenous polyamines. Plant Sci 151:59–66
Wahid A, Gelani S, Ashraf M, Foolad MR (2007) Heat tolerance in plants: an overview. Environ Exp Bot 61:199–223
Yoon JY, Hamayun M, Lee S-K, Lee I-J (2009) Methyl jasmonate alleviated salinity stress in soybean. J Crop Sci Biotechnol 12:63–68
Zhu J-K (2001) Plant salt tolerance. Trends Plant Sci 6:66–71
Acknowledgments
This work was supported by Grants from the Ministry of Higher Education and Scientific Research of Tunisia.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Saibi, W., Feki, K., Ben Mahmoud, R. et al. Durum wheat dehydrin (DHN-5) confers salinity tolerance to transgenic Arabidopsis plants through the regulation of proline metabolism and ROS scavenging system. Planta 242, 1187–1194 (2015). https://doi.org/10.1007/s00425-015-2351-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00425-015-2351-z