Abstract
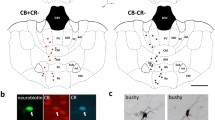
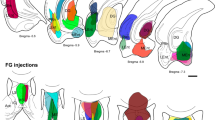
The reuniens nucleus in the midline thalamus projects to the medial prefrontal cortex (mPFC) and the hippocampus, and has been suggested to modulate interactions between these regions, such as spindle–ripple correlations during sleep and theta band coherence during exploratory behavior. Feedback from the hippocampus to the nucleus reuniens has received less attention but has the potential to influence thalamocortical networks as a function of hippocampal activation. We used the retrograde tracer cholera toxin B conjugated to two fluorophores to study thalamic projections to the dorsal and ventral hippocampus and to the prelimbic and infralimbic subregions of mPFC. We also examined the feedback connections from the hippocampus to reuniens. The goal was to evaluate the anatomical basis for direct coordination between reuniens, mPFC, and hippocampus by looking for double-labeled cells in reuniens and hippocampus. In confirmation of previous reports, the nucleus reuniens was the origin of most thalamic afferents to the dorsal hippocampus, whereas both reuniens and the lateral dorsal nucleus projected to ventral hippocampus. Feedback from hippocampus to reuniens originated primarily in the dorsal and ventral subiculum. Thalamic cells with collaterals to mPFC and hippocampus were found in reuniens, across its anteroposterior axis, and represented, on average, about 8 % of the labeled cells in reuniens. Hippocampal cells with collaterals to mPFC and reuniens were less common (~1 % of the labeled subicular cells), and located in the molecular layer of the subiculum. The results indicate that a subset of reuniens cells can directly coordinate activity in mPFC and hippocampus. Cells with collaterals in the hippocampus–reuniens–mPFC network may be important for the systems consolidation of memory traces and for theta synchronization during exploratory behavior.









Similar content being viewed by others
References
Berendse HW, Groenewegen HJ (1991) Restricted cortical termination fields of the midline and intralaminar thalamic nuclei in the rat. Neuroscience 42:73–102
Buzsáki G (2002) Theta oscillations in the hippocampus. Neuron 33:325–340
Cenquizca LA, Swanson LW (2006) An analysis of direct hippocampal cortical field CA1 axonal projections to diencephalon in the rat. J Comp Neurol 497:101–114. doi:10.1002/cne.20985
Condé F, Audinat E, Maire-Lepoivre E, Crépel F (1990) Afferent connections of the medial frontal cortex of the rat. A study using retrograde transport of fluorescent dyes. I. Thalamic afferents. Brain Res Bull 24:341–354
Conte WL, Kamishina H, Reep RL (2009) The efficacy of the fluorescent conjugates of cholera toxin subunit B for multiple retrograde tract tracing in the central nervous system. Brain Struct Funct 213:367–373. doi:10.1007/s00429-009-0212-x
Davidson TJ, Kloosterman F, Wilson MA (2009) Hippocampal replay of extended experience. Neuron 63:497–507. doi:10.1016/j.neuron.2009.07.027
Deng C, Rogers LJ (1999) Differential sensitivities of the two visual pathways of the chick to labelling by fluorescent retrograde tracers. J Neurosci Methods 89:75–86
Dolleman-Van Der Weel MJ, Witter MP (1996) Projections from the nucleus reuniens thalami to the entorhinal cortex, hippocampal field CA1, and the subiculum in the rat arise from different populations of neurons. J Comp Neurol 364:637–650. doi:10.1002/(SICI)1096-9861(19960122)364:4<637:AID-CNE3>3.0.CO;2-4
Ego-Stengel V, Wilson MA (2010) Disruption of ripple-associated hippocampal activity during rest impairs spatial learning in the rat. Hippocampus 20:1–10. doi:10.1002/hipo.20707
Fanselow MS, Dong H-W (2010) Are the dorsal and ventral hippocampus functionally distinct structures? Neuron 65:7–19. doi:10.1016/j.neuron.2009.11.031
Fenno L, Yizhar O, Deisseroth K (2011) The development and application of optogenetics. Annu Rev Neurosci 34:389–412. doi:10.1146/annurev-neuro-061010-113817
Foster DJ, Wilson MA (2006) Reverse replay of behavioural sequences in hippocampal place cells during the awake state. Nature 440:680–683. doi:10.1038/nature04587
Girardeau G, Benchenane K, Wiener SI et al (2009) Selective suppression of hippocampal ripples impairs spatial memory. Nat Neurosci 12:1222–1223. doi:10.1038/nn.2384
Goshen I, Brodsky M, Prakash R et al (2011) Dynamics of retrieval strategies for remote memories. Cell 147:678–689. doi:10.1016/j.cell.2011.09.033
Halassa MM, Siegle JH, Ritt JT et al (2011) Selective optical drive of thalamic reticular nucleus generates thalamic bursts and cortical spindles. Nat Neurosci 14:1118–1120. doi:10.1038/nn.2880
Herkenham M (1978) The connections of the nucleus reuniens thalami: evidence for a direct thalamo-hippocampal pathway in the rat. J Comp Neurol 177:589–610. doi:10.1002/cne.901770405
Hoover WB, Vertes RP (2007) Anatomical analysis of afferent projections to the medial prefrontal cortex in the rat. Brain Struct Funct 212:149–179. doi:10.1007/s00429-007-0150-4
Hoover WB, Vertes RP (2011) Collateral projections from nucleus reuniens of thalamus to hippocampus and medial prefrontal cortex in the rat: a single and double retrograde fluorescent labeling study. Brain Struct Funct. doi:10.1007/s00429-011-0345-6
Hyman JM, Zilli EA, Paley AM, Hasselmo ME (2005) Medial prefrontal cortex cells show dynamic modulation with the hippocampal theta rhythm dependent on behavior. Hippocampus 15:739–749. doi:10.1002/hipo.20106
Hyman JM, Zilli EA, Paley AM, Hasselmo ME (2010) Working memory performance correlates with prefrontal-hippocampal theta interactions but not with prefrontal neuron firing rates. Front Integr Neurosci 4:2. doi:10.3389/neuro.07.002.2010
Jay TM, Witter MP (1991) Distribution of hippocampal CA1 and subicular efferents in the prefrontal cortex of the rat studied by means of anterograde transport of phaseolus vulgaris-leucoagglutinin. J Comp Neurol 313:574–586. doi:10.1002/cne.903130404
Jay TM, Thierry A-M, Wiklund L, Glowinski J (1992) Excitatory amino acid pathway from the hippocampus to the prefrontal cortex. Contribution of AMPA receptors in hippocampo-prefrontal cortex transmission. Eur J Neurosci 4:1285–1295
Jones EG (2007) The thalamus. Cambridge University Press, London
Jones MW, Wilson MA (2005) Theta rhythms coordinate hippocampal-prefrontal interactions in a spatial memory task. PLoS Biol 3:e402. doi:10.1371/journal.pbio.0030402
Joseph KC, Kim SU, Stieber A, Gonatas NK (1978) Endocytosis of cholera toxin into neuronal GERL. Proc Natl Acad Sci USA 75:2815–2819
Kealy J, Commins S (2011) The rat perirhinal cortex: a review of anatomy, physiology, plasticity, and function. Prog Neurobiol 93:522–548. doi:10.1016/j.pneurobio.2011.03.002
Krettek JE, Price JL (1977) The cortical projections of the mediodorsal nucleus and adjacent thalamic nuclei in the rat. J Comp Neurol 171:157–191. doi:10.1002/cne.901710204
Lee MG, Chrobak JJ, Sik A et al (1994) Hippocampal theta activity following selective lesion of the septal cholinergic system. Neuroscience 62:1033–1047. doi:10.1016/0306-4522(94)90341-7
Louie K, Wilson MA (2001) Temporally structured replay of awake hippocampal ensemble activity during rapid eye movement sleep. Neuron 29:145–156
Loureiro M, Cholvin T, Lopez J, et al. (2012) The ventral midline thalamus (reuniens and rhomboid nuclei) contributes to the persistence of spatial memory in rats. J Neurosci 32:9947–9959. doi:10.1523/JNEUROSCI.0410-12.2012
Maviel T, Durkin TP, Menzaghi F, Bontempi B (2004) Sites of neocortical reorganization critical for remote spatial memory. Science 305:96–99. doi:10.1126/science.1098180
McKenna JT, Vertes RP (2004) Afferent projections to nucleus reuniens of the thalamus. J Comp Neurol 480:115–142. doi:10.1002/cne.20342
Moga MM, Weis RP, Moore RY (1995) Efferent projections of the paraventricular thalamic nucleus in the rat. J Comp Neurol 359:221–238. doi:10.1002/cne.903590204
Namura S, Takada M, Kikuchi H, Mizuno N (1994) Topographical organization of subicular neurons projecting to subcortical regions. Brain Res Bull 35:221–231
Nyakas C, Luiten PG, Spencer DG, Traber J (1987) Detailed projection patterns of septal and diagonal band efferents to the hippocampus in the rat with emphasis on innervation of CA1 and dentate gyrus. Brain Res Bull 18:533–545
Oka H, Yoshida K (1985) Septohippocampal connections to field CA1 of the rat identified with field potential analysis and retrograde labeling by horseradish peroxidase. Neurosci Lett 58:19–24
Otake K, Nakamura Y (1998) Single midline thalamic neurons projecting to both the ventral striatum and the prefrontal cortex in the rat. Neuroscience 86:635–649
Paxinos G, Watson C (1986) The rat brain in stereotaxic coordinates. Academic Press, London
Peyrache A, Battaglia FP, Destexhe A (2011) Inhibition recruitment in prefrontal cortex during sleep spindles and gating of hippocampal inputs. Proc Natl Acad Sci USA 108:17207–17212. doi:10.1073/pnas.1103612108
Quinn JJ, Ma QD, Tinsley MR et al (2008) Inverse temporal contributions of the dorsal hippocampus and medial prefrontal cortex to the expression of long-term fear memories. Learn Mem 15:368–372. doi:10.1101/lm.813608
Risold PY, Thompson RH, Swanson LW (1997) The structural organization of connections between hypothalamus and cerebral cortex. Brain Res Brain Res Rev 24:197–254
Rogan SC, Roth BL (2011) Remote control of neuronal signaling. Pharmacol Rev 63:291–315. doi:10.1124/pr.110.003020
Schofield BR, Schofield RM, Sorensen KA, Motts SD (2007) On the use of retrograde tracers for identification of axon collaterals with multiple fluorescent retrograde tracers. Neuroscience 146:773–783. doi:10.1016/j.neuroscience.2007.02.026
Shibata H (1993) Efferent projections from the anterior thalamic nuclei to the cingulate cortex in the rat. J Comp Neurol 330:533–542. doi:10.1002/cne.903300409
Siapas AG, Wilson MA (1998) Coordinated interactions between hippocampal ripples and cortical spindles during slow-wave sleep. Neuron 21:1123–1128
Siapas AG, Lubenov EV, Wilson MA (2005) Prefrontal phase locking to hippocampal theta oscillations. Neuron 46:141–151. doi:10.1016/j.neuron.2005.02.028
Sigurdsson T, Stark KL, Karayiorgou M et al (2010) Impaired hippocampal-prefrontal synchrony in a genetic mouse model of schizophrenia. Nature 464:763–767. doi:10.1038/nature08855
Sirota A, Csicsvari J, Buhl D, Buzsáki G (2003) Communication between neocortex and hippocampus during sleep in rodents. Proc Natl Acad Sci USA 100:2065–2069. doi:10.1073/pnas.0437938100
Squire LR, Clark RE, Knowlton BJ (2001) Retrograde amnesia. Hippocampus 11:50–55. doi:10.1002/1098-1063(2001)11:1<50::AID-HIPO1019>3.0.CO;2-G
Steffenach H-A, Witter M, Moser M-B, Moser EI (2005) Spatial memory in the rat requires the dorsolateral band of the entorhinal cortex. Neuron 45:301–313. doi:10.1016/j.neuron.2004.12.044
Steriade M, Domich L, Oakson G, Deschênes M (1987) The deafferented reticular thalamic nucleus generates spindle rhythmicity. J Neurophysiol 57:260–273
Stoeckel K, Schwab M, Thoenen H (1977) Role of gangliosides in the uptake and retrograde axonal transport of cholera and tetanus toxin as compared to nerve growth factor and wheat germ agglutinin. Brain Res 132:273–285
Su HS, Bentivoglio M (1990) Thalamic midline cell populations projecting to the nucleus accumbens, amygdala, and hippocampus in the rat. J Comp Neurol 297:582–593. doi:10.1002/cne.902970410
Suh J, Rivest AJ, Nakashiba T et al (2011) Entorhinal cortex layer III input to the hippocampus is crucial for temporal association memory. Science 334:1415–1420. doi:10.1126/science.1210125
Swanson LW (1981) A direct projection from Ammon’s horn to prefrontal cortex in the rat. Brain Res 217:150–154
Thompson SM, Robertson RT (1987) Organization of subcortical pathways for sensory projections to the limbic cortex. I. Subcortical projections to the medial limbic cortex in the rat. J Comp Neurol 265:175–188. doi:10.1002/cne.902650203
Van der Werf YD, Witter MP, Groenewegen HJ (2002) The intralaminar and midline nuclei of the thalamus. Anatomical and functional evidence for participation in processes of arousal and awareness. Brain Res Brain Res Rev 39:107–140
van Groen T, Wyss JM (1992) Projections from the lateral dorsal nucleus of the thalamus to the limbic and visual cortices in the rat. J Comp Neurol 324:427–448. doi:10.1002/cne.903240310
van Groen T, Wyss JM (1995) Projections from the anterodorsal and anteroventral nucleus of the thalamus to the limbic cortex in the rat. J Comp Neurol 358:584–604. doi:10.1002/cne.903580411
Vertes RP (2004) Differential projections of the infralimbic and prelimbic cortex in the rat. Synapse 51:32–58. doi:10.1002/syn.10279
Vertes RP, Hoover WB (2008) Projections of the paraventricular and parataenial nuclei of the dorsal midline thalamus in the rat. J Comp Neurol 508:212–237. doi:10.1002/cne.21679
Vertes RP, Hoover WB, Viana Di Prisco G (2004) Theta rhythm of the hippocampus: subcortical control and functional significance. Behav Cogn Neurosci Rev 3:173–200. doi:10.1177/1534582304273594
Vertes RP, Hoover WB, Do Valle AC et al (2006) Efferent projections of reuniens and rhomboid nuclei of the thalamus in the rat. J Comp Neurol 499:768–796. doi:10.1002/cne.21135
Vertes RP, Hoover WB, Szigeti-Buck K, Leranth C (2007) Nucleus reuniens of the midline thalamus: link between the medial prefrontal cortex and the hippocampus. Brain Res Bull 71:601–609. doi:10.1016/j.brainresbull.2006.12.002
Vidal-Gonzalez I, Vidal-Gonzalez B, Rauch SL, Quirk GJ (2006) Microstimulation reveals opposing influences of prelimbic and infralimbic cortex on the expression of conditioned fear. Learn Mem 13:728–733. doi:10.1101/lm.306106
Wilson MA, McNaughton BL (1994) Reactivation of hippocampal ensemble memories during sleep. Science 265:676–679
Wouterlood FG, Saldana E, Witter MP (1990) Projection from the nucleus reuniens thalami to the hippocampal region: light and electron microscopic tracing study in the rat with the anterograde tracer Phaseolus vulgaris-leucoagglutinin. J Comp Neurol 296:179–203. doi:10.1002/cne.902960202
Wyss JM, Swanson LW, Cowan WM (1979) A study of subcortical afferents to the hippocampal formation in the rat. Neuroscience 4:463–476
Yamano M, Luiten PG (1989) Direct synaptic contacts of medial septal efferents with somatostatin immunoreactive neurons in the rat hippocampus. Brain Res Bull 22:993–1001
Acknowledgments
We thank K. Rockland for advice and comments throughout the duration of the project and for feedback on the manuscript. We also thank MJ. Galazo and J. Castro for technical advice and the M. Sur and S. Tonegawa laboratories for access to confocal and epifluorescence microscopes. This work was supported by a postdoctoral fellowship from the Caja Madrid Foundation (C.V.) and US National Institutes of Health grant 5R01MH061976 (M.A.W.).
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Varela, C., Kumar, S., Yang, J.Y. et al. Anatomical substrates for direct interactions between hippocampus, medial prefrontal cortex, and the thalamic nucleus reuniens. Brain Struct Funct 219, 911–929 (2014). https://doi.org/10.1007/s00429-013-0543-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00429-013-0543-5