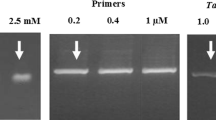
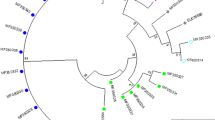
Abstract
Trichomonas vaginalis is a human urogenital pathogen that causes trichomoniasis, the most common nonviral parasitic sexually transmitted infection in the world. Presently, there are no reports on comparative sequence analysis as well as on the identification of phylogenetic positions of T. vaginalis isolates from the Philippines relative to known trichomonads. In this study, 5.8S rDNA and the flanking internal transcribed spacer (ITS) regions of 57 T. vaginalis isolates were sequenced. The phylogenetic positions of the isolates relative to known trichomonads were determined using the model-based (GTR+Γ+I) neighbor-joining, maximum likelihood, and Bayesian-inference analyses and the nonmodel-based maximum parsimony analysis. Construction of a phylogenetic tree showed the clustering of all the sequences in one branch together with other T. vaginalis strains obtained through basic local alignment search tool search. Sequencing of the 5.8S rDNA gene and the flanking ITS1and ITS2 regions of T. vaginalis isolates from the Philippines demonstrated low genetic polymorphism. However, comparison of the ribosomal DNA sequences may have implications on some phenotypic characteristics of T. vaginalis.


Similar content being viewed by others
References
Diamond LS, Harlow DH, Cunnick CC (1978) A new medium for the axenic cultivation of Entamoeba histolytica and other Entamoeba. T Roy Soc Trop Med H 72:431–432
Felleisen RS (1997) Comparative sequence analysis of 5.8S rRNA genes and internal transcribed spacer (ITS) regions of trichomonadid protozoa. Parasitology 115:111–119
Guindon S, Lethiec F, Duroux P, Gascuel O (2005) PHYML Online – a web server for fast maximum likelihood-based phylogenetic inference. Nucleic Acids Res 33:W557–W559
Hampl V, Vaňáčová S, Kulda J, Flegr J (2001) Concordance between genetic relatedness and phenotypic similarities of Trichomonas vaginalis strains. BMC Evol Biol 1:11
Hobbs MM, Lapple DM, Lawing LF, Schwebke JR, Cohen MS, Swygard H, Atashili J, Leone PA, Miller WC, Seña AC (2006) Methods for detection of Trichomonas vaginalis in the male partners of infected women: implications for control of trichomoniasis. J Clin Microbiol 44:3994–3999
Kleina P, Bettim-Bandinelli J, Bonatto SL, Benchimol M, Bogo MR (2004) Molecular phylogeny of Trichomonadidae family inferred from ITS-1, 5.8S rRNA and ITS-2 sequences. Int J Parasitol 34:963–970
Kulda J (1989) Employment of experimental animals in studies of Trichomonas vaginalis infection. In: Hongiberg BM (ed) Trichomonads parasitic in humans. Springer, New York, pp 205–277
Morgulis A, Coulouris G, Raytselis Y, Madden TL, Agarwala R, Shäffer AA (2008) Database indexing for production MegaBLAST searches. Bioinformatics 24:1757–1764
Page RD (1996) TreeView: an application to display phylogenetic trees on personal computers. Comput Appl Biosci 12:357–358
Petrin D, Delgaty K, Bhatt R, Garber G (1998) Clinical and microbiological aspects of Trichomonas vaginalis. Clin Microbiol Rev 11:300–317
Rein MF, Müller M (1990) Trichomonas vaginalis and trichomoniasis. In: Holmes KK (ed) Sexually transmitted diseases. McGraw-Hill, New York, pp 481–492
Rivera WL, Tachibana H, Silva-Tahat MR, Uemura H, Kanbara H (1996) Differentiation of Entamoeba histolytica and E. dispar DNA from cysts present in stool specimens by polymerase chain reaction: its field application in the Philippines. Parasitol Res 82:585–589
Ronquist F, Huelsenbeck JP (2003) MrBayes 3: Bayesian phylogenetic inference under mixed models. Bioinformatics 19:1572–1574
Shafir SC, Sorvillo FJ (2006) Viability of Trichomonas vaginalis in urine: epidemiologic and clinical implications. J Clin Microbiol 44:3787–3789
Snipes LJ, Gamard PM, Narcisi EM, Ben Beard C, Lehmann T, Evan Secor W (2000) Molecular epidemiology of metronidazole resistance in a population of Trichomonas vaginalis clinical isolates. J Clin Microbiol 38:3004–3009
Staden R, Beal KF, Bonfield JK (2000) The Staden package, 1998. Methods Mol Biol 132:115–130
Swofford DL (2000) PAUP*. Phylogenetic analysis using parsimony (*and other methods). V. 4.0b10. Sinauer, Sunderland
Walker RL, Hayes DC, Sawyer SJ, Nordhausen RW, Van Hoosear KA, Bon Durant RH (2003) Comparison of the 5.8S rRNA gene and internal transcribed spacer regions of trichomonadid protozoa recovered from the bovine preputial cavity. J Vet Diagn Invest 15:14–20
World Health Organization (2001) Global prevalence and incidence of selected curable sexually transmitted infections. www.who.int/docstore/hiv/GRSTI/006.htm
Zhang Z, Schwartz S, Wagner L, Miller W (2000) A greedy algorithm for aligning DNA sequences. J Comput Biol 7:203–214
Acknowledgements
We thank M.I.P. Queza, M.V. Villareal, D.E.V. Adao, and I.K.C. Fontanilla for their technical support. This work was supported by a research grant from the Office of the Vice Chancellor for Research and Development of the University of the Philippines-Diliman (Grant No. 080809 PNSE) to W.L.R.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Rivera, W.L., Ong, V.A. & Masalunga, M.C. Molecular characterization of Trichomonas vaginalis isolates from the Philippines. Parasitol Res 106, 105–110 (2009). https://doi.org/10.1007/s00436-009-1635-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00436-009-1635-2