Abstract
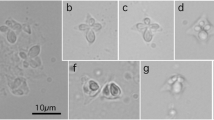
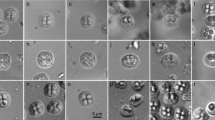
Kudoa neothunni is the first described Kudoa species having six shell valves and polar capsules, previously assigned to the genus Hexacapsula Arai and Matsumoto, 1953. Since its genetic analyses remain to be conducted, the present study characterizes the ribosomal RNA gene (rDNA) using two isolates from a yellowfin tuna (Thunnus albacares) with post-harvest myoliquefaction and a northern bluefin tuna (Thunnus thynnus) without tissue degradation. Spores of the two isolates localized in the myofiber of trunk muscles, forming pseudocysts, and showed typical morphology of K. neothunni with six equal-sized shell valves radially arranged in apical view: spores (n = 15) measuring 9.5–11.4 μm in width, 7.3–8.6 μm in suture width, 8.9–10.9 μm in thickness, and 7.3–7.7 μm in length; and polar capsules measuring 3.6–4.1 μm by 1.8–2.3 μm. In lateral view, the spores were pyramidal in shape without apical protrusions. Their 18S and 5.8S rDNA sequences were essentially identical, but variations in the ITS1 (62.4 % similarity across 757-bp length), ITS2 (66.9 % similarity across 599-bp length), and 28S (99.0 % similarity across 2,245-bp length) rDNA regions existed between the two isolates. On phylogenetic trees based on the 18S or 28S rDNA sequence, K. neothunni formed a clade with Kudoa spp. with more than four shell valves and polar capsules, particularly K. grammatorcyni and K. scomberomori. Semiquadrate spores of a kudoid species with four shell valves and polar capsules were detected from minute cysts (0.30–0.75 mm by 0.20–0.40 mm) embedded in the trunk muscle of a chub mackerel (Scomber japonicus) fished in the Sea of Japan. Morphologically, it resembled K. caudata described from a chub mackerel fished in the southeastern Pacific Ocean off Peru; however, it lacked filamentous projections on the shell valves of spores. Additionally, it morphologically resembled K. thunni described from a yellowfin tuna also fished in the Pacific Ocean; spores (n = 30) measuring 8.2–10.5 μm in width, 7.0–8.8 μm in thickness, and 6.1–6.8 μm in length; and polar capsule measuring 2.5–3.4 μm by 1.3–2.0 μm. The similarities of the 18S and 28S rDNA sequences between these two species were 98.5 % and 96.3 %, respectively. Simultaneously, the dimensions of cysts in the trunk muscle formed by K. thunni are clearly larger than those of the present species from a chub mackerel: 1.3–2.0 mm by 1.1–1.4 mm (n = 14) vs. 0.30–0.75 mm by 0.20–0.40 mm (n = 7), respectively. Thus, Kudoa scomberi n. sp. is proposed for this multivalvulid species found in the chub mackerel.







Similar content being viewed by others
References
Abollo E, Novoa B, Figueras A (2005) SSU rDNA analysis of Kudoa rosenbuschi (Myxosporea) from the Argentinean hake Merluccius hubbsi. Dis Aquat Org 64:135–139
Adlard RD, Bryant MS, Whipps CM, Kent ML (2005) Multivalvulid myxozoans from eastern Australia: three new species of Kudoa from scombrid and labrid fishes of the great barrier reef, Queensland, Australia. J Parasitol 91:1138–1142
Anisimova M, Gascuel O (2006) Approximate likelihood-ratio test for branches: a fast, accurate, and powerful alternative. Syst Biol 55:539–552
Arai Y, Matsumoto K (1953) On a new sporozoa, Hexacapsula neothunni gen. et sp. nov., from the muscle of yellowfin tuna, Neothunnus macropterus. Bull Jpn Soc Sci Fish 18:293–299
Bartosová P, Fiala I, Hypsa V (2009) Concatenated SSU and LSU rDNA data confirm the main evolutionary trends within myxosporeans (Myxozoa: Myxosporea) and provide an effective tool for their molecular phylogenetics. Mol Phylogenet Evol 53:81–93
Burger MAA, Adlard RD (2010a) Four new species of Kudoa Meglitsch, 1947 (Myxosporea: Multivalvulida) from Australia with recommendations for species descriptions in the Kudoidae. Parasitology 137:793–814
Burger MAA, Adlard RD (2010b) Phenotypic variation in a significant spore character in Kudoa (Myxosporea: Multivalvulida) species infecting brain tissue. Parasitology 137:1759–1772
Burger MAA, Adlard RD (2011) Low host specificity in the Kudoidae (Myxosporea: Multivalvulida) including seventeen new host records for Kudoa thalassomi. Folia Parasitol 58:1–16
Burger MAA, Cribb TH, Adlard RD (2007) Patterns of relatedness in the Kudoidae with descriptions of Kudoa chaetodoni n. sp. and K. lethrini n. sp. (Myxosporea: Multivalvulida). Parasitology 134:669–681
Casal G, Matos E, Matos P, Azevedo C (2008) Ultrastructural description of a new myxosporean parasite Kudoa aequidens sp. n. (Myxozpa, Myxosporea), found in the sub-opercular musculature of Aequidens plagiozonatus (Teleostei) from the Amazon river. Acta Protozool 47:135–141
Dawson-Coates JA, Chase JC, Funk V, Booy MH, Haines LR, Falkenberg CL, Whitaker DJ, Olafson RW, Pearson TW (2003) The relationship between flesh quality and numbers of Kudoa thyrsites plasmodia and spores in farmed Atlantic salmon, Salmo salar L. J Fish Dis 26:451–459
Dereeper A, Guignon V, Blanc G, Audic S, Buffet S, Chevenet F, Dufayard JF, Guindon S, Lefort V, Lescot M, Claverie J-M, Gascuel O (2008) Phylogeny.fr: robust phylogenetic analysis for the non-specialist. Nucleic Acids Res 36:465–469
Diamant A, Ucko M, Paperna I, Colorni A, Lipshitz A (2005) Kudoa iwatai (Myxosporea: Multivalvulida) in wild and cultured fish in the red sea: Redescription and molecular phylogeny. J Parasitol 91:1175–1189
Egusa S, Shiomitsu T (1983) Two new species of the genus Kudoa (Myxosporea: Multivalvulida) from marine cultured fishes in Japan. Fish Pathol 18:163–171 (in Japanese with English summary)
Funk VA, Raap M, Sojonky K, Jones S, Robinson J, Falkenberg C, Miller KM (2007) Development and validation of an RNA- and DNA-based quantitative PCR assay for deterimination of Kudoa thyrsites infection levels in Atlantic salmon Salmo salar. Dis Aquat Org 75:239–249
Funk VA, Olafson RW, Raap M, Smith D, Aitken L, Haddow JD, Wang D, Dawson-Coates JA, Burke RD, Miller KM (2008) Identification, characterization and deduced amino acid sequence of the dominant protease from Kudoa paniformis and K. thyrsites; a unique cytoplasmic cysterine protease. Comp Biochem Physiol B 149:477–489
Grabner DS, Yokoyama H, Shirakashi S, Kinami R (2012) Diagnostic PCR assays to detect and differentiate Kudoa septempunctata, K. thyrsites and K. lateolabracis (Myxozoa, Multivalvulida) in muscle tissue of olive flounder (Paralichthys olivaceus). Aquaculture 338–341:36–40
Grossel GW, Dykova I, Handlinger J, Munday BL (2003) Pentacapsula neurophila sp. n. (Multivalvulida) from the central nervous system of striped trumbeter, Latris lineata (Forster). J Fish Dis 26:315–320
Guindon S, Gascuel O (2003) A simple, fast, and accurate algorithm to estimate large phylogenies by maximum likelihood. Syst Biol 52:696–704
Gunter NL, Whipps CM, Cribb TH, Adlard RD (2006) Characterisation of Kudoa monodactyli n. sp. (Myxozoa: Multivalvulida) from Monodactylus argenterus (Teleostei: Monodactylidae) from Moreton Bay, Queensland, Australia. J Eukaryot Microbiol 53:374–378
Harada T, Kawai T, Sato H, Yokoyama H, Kumeda Y (2012) Development of a quantitative polymerase chain reaction assay for detection of Kudoa septempunctata in olive flounder (Paralichthys olivaceus). Int J Food Microbiol 155:161–167
Hsieh S, Chen C (1984) Septemcapsula yasunagai gen. et sp. nov., representative of a ne family of the class Myxosporea. Acta Zootax Sinica 9:225–227 (in Chinese with English summary)
Iversen ES, Van Meter NN (1967) A new myxosporidian (Sporozoa) infecting the Spanish mackerel. Bull Mar Sci 17:268–273
Kawai T, Sekizuka T, Yahata Y, Kuroda M, Kumeda Y, Iijima Y, Kamata Y, Sugita-Konishi Y, Ohnishi T (2012) Identification of Kudoa septempunctata as the causative agent of novel food poisoning outbreaks in Japan by consumption of Paralichthys olivaceus in raw fish. Clin Infect Dis 54:1046–1052
Kent ML, Andree KB, Bartholomew JL, El-Matbouli M, Desser SS, Devlin RH, Feist SW, Hedrick RP, Hoffmann RW, Khattra J, Hallett SL, Lester RJG, Longshaw M, Palenzuela O, Siddall ME, Xiao C (2001) Recent advances in our knowledge of the Myxozoa. J Eukaryot Microbiol 48:395–413
Konagaya S (1982) Histological observation of jellied yellowfin tuna meat. Bull Tokai Reg Fish Res Lab 106:55–75 (in Japanese with English summary)
Konagaya S (1984) Studies on the jellied meat of fish, with special reference to that of yellowfin tuna. Bull Tokai Reg Fish Res Lab 114:1–101 (in Japanese with English summary)
Konishi Y (2011) Overview of the study on new food-borne diseases associated with raw fish and flesh. Food Sanit Res 740:7–12 (in Japanese)
Kovaleva AA, Gaevskaya AV (1983) [First data on fish myxosporidia of the southeastern Pacific open waters.] Vestnik Zoologii 17:6-11. (In Russian)
Lom J, Arthur JR (1989) A guideline for the preparation of species descriptions in Myxosporea. J Fish Dis 12:151–156
Lom J, Noble ER (1984) Revised classification of the Myxosporea Bütschli, 1881. Folia Parasitol 31:193–205
Martínez de Velasco G, Rodero M, Cuéllar C, Chivato T, Mateos JM, Laguna R (2008) Skin prick test of Kudoa sp. antigens in patients with gastrointestinal and/or allergic symptoms related to fish ingestion. Parasitol Res 103:713–715
Matsukane Y, Sato H, Tanaka S, Kamata Y, Sugita-Konishi Y (2010) Kudoa septempunctata n. sp. (Myxosporea: Multivalvulida) from an aquacultured olive flounder (Paralichthys olivaceus) imported from Korea. Parasitol Res 107:865–872
Matsukane Y, Sato H, Tanaka S, Kamata Y, Sugita-Konishi Y (2011) Kudoa iwatai and two novel Kudoa spp., K. trachuri n. sp. and K. thunni n. sp. (Myxosporea: Multivalvulida), from daily consumed marine fish in western Japan. Parasitol Res 108:913–926
Meng F, Yokoyama H, Shirakashi S, Grabner D, Ogawa K, Ishimaru K, Sawada Y, Murata O (2011) Kudoa prunusi n. sp. (Myxozoa: Multivalvulida) from the brain of Pacific bluefin tuna Thunnus orientalis (Temminck & Schlegel, 1844) cultured in Japan. Parasitol Int 60:90–96
Moran JDW, Whitaker DJ, Kent ML (1999) A review of the myxosporean genus Kudoa Meglitsch, 1947, and its impact on the international aquaculture industry and commercial fisheries. Aquaculture 172:163–196
Naidenova NN, Zaika VE (1970) Three new genera of myxosporean parasites of fish from the Indian Ocean. Zool Zh 49:451–454 (in Russian with English summary)
Ohnishi T (2011) Food poisoning caused by Kudoa septempunctata. Food Sanit Res 740:13–20 (in Japanese)
Pampoulie C, Marques A, Rosecchi E, Crivelli AJ, Bouchereau J-L (1999) A new myxosporean parasite, Kudoa camarguensis n. sp., recorded on two goby species (Teleostei: Pisces) in the Rhône Delta (Mediterranean Sea, France). J Eukaryot Microbiol 46:304–310
Sato H (2011) Biology of the Myxozoa, a newly recognized parasitic pathogen causing food poisoning. Yamaguchi J Vet Med 38:1–26 (in Japanese with English summary)
Smythe AB, Sanderson MJ, Nadler SA (2006) Nematode small subunit phylogeny correlates with alignment parameters. Syst Biol 55:972–992
Thompson JD, Higgins DG, Gibson TJ (1994) CLUSTAL W: improving the sensitivity of progressive multiple sequence alignment through sequence weighting, position-specific gap penalties and weight matrix choice. Nucleic Acids Res 22:4673–4680
Whipps CM, Diggles BK (2006) Kudoa alliaria in flesh of Argentinian hoki Macruronus magellanicus (Gadiformes; Merlucciidae). Dis Aquat Organ 69:259–263
Whipps CM, Kent ML (2006) Phylogeography of the cosmopolitan marine parasite Kudoa thyrsites (Myxozoa: Myxosporea). J Eukaryot Microbiol 53:364–373
Whipps CM, Adlard RD, Bryant MS, Kent ML (2003) Two unusual myxozoans, Kudoa quadricornis n. sp. (Multivalvulida) from the muscle of goldspotted trevally (Carangoides fulvoguttatus) and Kudoa permulticapsula n. sp. (Myltivalvulida) from the muscle of Spanish mackerel (Scomberomorus commerson) from the Great Barrier Reef, Australia. J Parasitol 89:168–173
Whipps CM, Grossel G, Adlard RD, Yokoyama H, Bryant MS, Munday BL, Kent ML (2004) Phylogeny of the Multivalvulidae (Myxozoa: Myxosporea) based on comparative ribosomal DNA sequence analysis. J Parasitol 90:618–622
Yokoyama H (2003) A review: gaps in our knowledge on myxozoan parasites of fishes. Fish Pathol 38:125–136
Yokoyama H, Whipps CM, Kent ML, Mizuno K, Kawakami H (2004) Kudoa thyrsites from Japanese flounder and Kudoa lateolabracis n. sp. from Chinese sea bass: causative myxozoans of post-mortem myoliquefaction. Fish Pathol 39:79–85
Zhou LS, Li-Chan ECY (2009) Effect of Kudoa spores, endogenous protease activity and frozen storage on cooked texture of minced Pacific hake (Merluccius productus). Food Chem 11:1076–1082
Acknowledgments
This study was supported in part by a Grant-in-Aid (H23-shokuhin-ippan-007) from the Ministry of Health, Labour and Welfare of Japan.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Li, YC., Sato, H., Tanaka, S. et al. Characterization of the ribosomal RNA gene of Kudoa neothunni (Myxosporea: Multivalvulida) in tunas (Thunnus spp.) and Kudoa scomberi n. sp. in a chub mackerel (Scomber japonicus). Parasitol Res 112, 1991–2003 (2013). https://doi.org/10.1007/s00436-013-3357-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00436-013-3357-8