Abstract
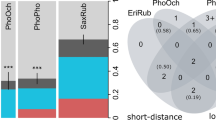
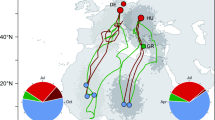
Mobile hosts like birds occupy a wide array of habitats in which they encounter various vector and parasite faunas. If the infection probability for vector-borne parasites varies among seasons and biomes, a migratory life can critically influence the infections of a host. The growing body of literature on avian blood parasites suggests that host migrations do not only influence prevalence of infection but can also evoke higher infection intensities and increased parasite diversity in migrant compared to resident host species. We investigated the prevalence, intensity and diversity of Plasmodium and Haemoproteus infections in three closely-related and sympatrically breeding sparrow species with different migration strategies ranging from residential house sparrow and partially migratory tree sparrow to the obligate migratory Spanish sparrow. With a prevalence of 49%, the migratory Spanish sparrows were significantly less frequently infected than the resident house sparrows (82%). The partially migratory tree sparrow showed an intermediate prevalence of 60%. The parasitaemias were similar in all three host species and indicated mostly chronic but also few acute infections. While we found Plasmodium parasites in all three sparrow species, only Spanish sparrows were infected with Haemoproteus parasites in our study. With nine clearly identified parasite lineages in our study and the highest number of lineages per infected individuals (i.e. relative diversity), Spanish sparrows harboured the most diverse parasite fauna. Our results suggest that migration strategies can affect Plasmodium and Haemoproteus infections of sparrows resulting in a lower parasite prevalence and higher parasite diversity in migratory hosts—at least during our host’s breeding period. A general scope for all annual cycle periods and across various bird taxa remains to be elucidated.



Similar content being viewed by others
References
Allander K, Sundberg J (1997) Temporal variation and reliability of blood parasite levels in captive yellowhammer males Emberiza citrinella. J Avian Biol 28:325–330
Altizer SM, Bartel RA, Han BA (2011) Animal migration and infectious disease risk. Science 331:296–302. https://doi.org/10.1126/science.1194694
Altizer SM, Dobson AP, Hosseini P, Hudson P, Pascual M, Rohani P (2006) Seasonality and the dynamics of infectious diseases. Ecol Lett 9:467–484
Bates D, Mächler M, Bolker B, Walker S (2015) Fitting linear mixed-effects models using lme4. J Stat Softw 67:1–48. https://doi.org/10.18637/jss.v067.i01
Bensch S, Hellgren O, Pérez-Tris J (2009) MalAvi: a public database of malaria parasites and related haemosporidians in avian hosts based on mitochondrial cytochrome b lineages. Mol Ecol Resour 9:1353–1358. https://doi.org/10.1111/j.1755-0998.2009.02692.x
Bichet C, Sorci G, Robert A, Julliard R, Lendvai AZ, Chastel O, Garnier S, Loiseau C (2014) Epidemiology of Plasmodium relictum infection in the house sparrow. J Parasitol 100:59–65. https://doi.org/10.1645/12-24.1
Bodenhofer U, Bonatesta E, Horejš-Kainrath C, Hochreiter S (2015) msa: an R package for multiple sequence alignment. Bioinformatics 31:3997–3999. https://doi.org/10.1093/bioinformatics/btv494
Boone AT, Rodewald PG, DeGroote LW (2010) Neotropical winter habitat of the Magnolia warbler: effects on molt, energetic condition, migration timing, and hematozoan infection during spring migration. Condor 112:115–122. https://doi.org/10.1525/cond.2010.090098
Bradley CA, Altizer SM (2005) Parasites hinder monarch butterfly flight: implications for disease spread in migratory hosts. Ecol Lett 8:290–300. https://doi.org/10.1111/j.1461-0248.2005.00722.x
Clarabuch O, Gonzalez-Solis J (1997) Parasitism as a migration cost. Biol e Conserv della Fauna 102:113–117
Cramp S (1994) Handbook of the birds of Europe, the Middle East and North Africa: crows and finches. Oxford University Press, Oxford
Dimitrov D, Zehtindjiev P, Bensch S (2010) Genetic diversity of avian blood parasites in SE Europe: cytochrome b lineages of the genera Plasmodium and Haemoproteus (Haemosporida) from Bulgaria. Acta Parasitol 55:201–209. https://doi.org/10.2478/s11686-010-0029-z
Eikenaar C, Hegemann A (2016) Migratory common blackbirds have lower innate immune function during autumn migration than resident conspecifics. Biol Lett 12:20160078. https://doi.org/10.1098/rsbl.2016.0078
Figuerola J, Green AJ (2000) Haematozoan parasites and migratory behaviour in waterfowl. Evol Ecol 14:143–153. https://doi.org/10.1023/A:1011009419264
Gylfe Å, Bergström SS, Lundström J, Olsen B, Lundstróm J, Olsen B (2000) Reactivation of Borrelia infection in birds. Nature 403:724–725. https://doi.org/10.1038/35001663
Hahn S, Bauer S, Dimitrov D, Emmenegger T, Ivanova K, Zehtindjiev P, Buttemer WA (2018) Low intensity blood parasite infections do not reduce the aerobic performance of migratory birds. Proc R Soc B 285:20172307. https://doi.org/10.1098/rspb.2017.2307
Hall RJ, Altizer SM, Bartel RA (2014) Greater migratory propensity in hosts lowers pathogen transmission and impacts. J Anim Ecol 83:1068–1077. https://doi.org/10.1111/1365-2656.12204
Hall T (1999) BioEdit: a user-friendly biological sequence alignment editor and analysis program for Windows 95/98/NT. Nucleic Acids Symp Ser 41:95–98
Hellgren O, Waldenström J, Bensch S (2004) A new PCR assay for simultaneous studies of Leucocytozoon, Plasmodium, and Haemoproteus from avian blood. J Parasitol 90:797–802. https://doi.org/10.1645/GE-184R1
Jarošík V, Honěk A, Magarey RD, Skuhrovec JJ, Honek A (2011) Developmental database for phenology models: related insect and mite species have similar thermal requirements. J Econ Entomol 104:1870–1876. https://doi.org/10.1603/EC11247
Johns S, Shaw AK (2016) Theoretical insight into three disease-related benefits of migration. Popul Ecol 58:213–221. https://doi.org/10.1007/s10144-015-0518-x
Koprivnikar J, Leung TLF (2015) Flying with diverse passengers: greater richness of parasitic nematodes in migratory birds. Oikos 124:399–405. https://doi.org/10.1111/oik.01799
Loehle C (1995) Social barriers to pathogen transmission in wild animal populations. Ecology 76:326–335. https://doi.org/10.2307/1941192
Loiseau C, Harrigan RJ, Robert A, Bowie RCK, Thomassen HA, Smith TB, Sehgal RNM, Francisco S, Angeles L, Angeles L, Africa S (2012) Host and habitat specialization of avian malaria in Africa. Mol Ecol 21:431–441. https://doi.org/10.1111/j.1365-294X.2011.05341.x
Marzal A, Ricklefs RE, Valkiūnas G, Albayrak T, Arriero E, Bonneaud C, Czirják GÁA, Ewen J, Hellgren O, Hořáková D, Iezhova TA, Jensen H, Križanauskienė A, Lima MR, de Lope F, Magnussen E, Martin LB, Møller AP, Palinauskas V, Pap PL, Pérez-Tris J, Sehgal RNM, Soler M, Szöllosi E, Westerdahl H, Zehtindjiev P, Bensch S, Zetindjiev P (2011) Diversity, loss, and gain of malaria parasites in a globally invasive bird. PLoS One 6:1–8. https://doi.org/10.1371/journal.pone.0021905
Mata VA, da Silva LPLP, Lopes RJ, Drovetski SV (2015) The Strait of Gibraltar poses an effective barrier to host-specialised but not to host-generalised lineages of avian Haemosporidia. Int J Parasitol 45:711–719. https://doi.org/10.1016/j.ijpara.2015.04.006
O’Connor EA, Cornwallis CK, Hasselquist D, Nilsson JÅ, Westerdahl H (2018) The evolution of immunity in relation to colonization and migration. Nat Ecol Evol 2:841–849. https://doi.org/10.1038/s41559-018-0509-3
Oksanen J, Blanchet FG, Friendly M, Kindt R, Legendre P, McGlinn D, Minchin PR, O’hara RB, Simpson GL, Solymos P, Stevens MHH, Szoecs E, Wagner H (2018) vegan: community ecology package. R Package. Version 2:5–2
Pérez-Rodríguez A, Fernández-González S, de la Hera I, Pérez-Tris J (2013) Finding the appropriate variables to model the distribution of vector-borne parasites with different environmental preferences: climate is not enough. Glob Chang Biol 19:n/a-n/a. https://doi.org/10.1111/gcb.12226
Pérez-Tris J, Bensch S (2005) Diagnosing genetically diverse avian malarial infections using mixed-sequence analysis and TA-cloning. Parasitology 131:15–23. https://doi.org/10.1017/S003118200500733X
R Core Team (2016) R: A language and environment for statistical computing
Schaefer H-C, Jetz W, Böhning-Gaese K (2007) Impact of climate change on migratory birds: community reassembly versus adaptation. Glob Ecol Biogeogr 17:071106211200001–??? https://doi.org/10.1111/j.1466-8238.2007.00341.x
Sehgal RNM (2015) Manifold habitat effects on the prevalence and diversity of avian blood parasites. Int J Parasitol Parasites Wildl 4:421–430. https://doi.org/10.1016/j.ijppaw.2015.09.001
Shurulinkov P (2005) Occurrence of haematozoan parasites of genus Hepatozoon (Apicomplexa: Hepatozoidae) in wild birds in Bulgaria. Acta Zool Bulg 57:245–252
Summers-Smith JD (1988 The Sparrows : a study of the genus Passer. Calton, Staffordshire, England: T & AD Poyser
Teitelbaum CS, Huang S, Hall RJ, Altizer SM (2018) Migratory behavior predicts greater parasite diversity in ungulates. Proc R Soc B Biol Sci
Valkiūnas G (2005) Avian malaria parasites and other Haemosporidia. CRC Press, Boca Raton
Valkiūnas G, Bensch S, Iezhova TA, Krizanauskiené A, Hellgren O, Bolshakov CV (2006) Nested cytochrome b polymerase chain reaction diagnostics underestimate mixed infections of avian blood haemosporidian parasites: microscopy is still essential. J Parasitol 92:418–422. https://doi.org/10.1645/GE-3547RN.1
Ventim R, Mendes L, Ramos JA, Cardoso H, Pérez-Tris J (2012a) Local haemoparasites in introduced wetland passerines. J Ornithol 153:1253–1259. https://doi.org/10.1007/s10336-012-0860-0
Ventim R, Tenreiro P, Grade N, Encarnação P, Araújo M, Mendes L, Pérez-Tris J, Ramos JA (2012b) Characterization of haemosporidian infections in warblers and sparrows at south-western European reed beds. J Ornithol 153:505–512. https://doi.org/10.1007/s10336-011-0767-11
Žiegytė R, Valkiūnas G (2014) Recent advances in vector studies of avian haemosporidian parasites. Ekologija 60:73–83. https://doi.org/10.6001/ekologija.v60i4.3042
Acknowledgments
We thank Bill Buttemer, Karina Ivanova, Martin Marinov and Strahil Peev for their support with field work as well as Zubera and Lyatif Ismail for their great hospitality. Finally, we would like to thank two anonymous reviewers for their constructive comments and inputs.
Funding
This study was funded by the Swiss National Science Foundation (SNSF) as a part of the project 31003A_160265. While TE, SB and SH were exclusively funded by SNSF, DD and PZ were co-funded from the Bulgarian National Science foundation under contract DN01/6 and JOM was funded by the Swiss Ornithological Institute. This study is report no. 63 of the Biological Experimental Station ‘Kalimok’.
Author information
Authors and Affiliations
Contributions
TE, SB and SH designed the study; PZ and DD arranged permissions and licences; TE, DD and SH organised and carried out the field work; JOM carried out the molecular lab work; TE and SH performed the screening by microscopy; TE implemented the data analysis and drafted the manuscript. All authors have revised the draft and agreed on the final version of the article.
Corresponding author
Ethics declarations
Ethical statement
All procedures were in accordance with the local animal ethics guidelines and were permitted by the Bulgarian Ministry of Environment and Waters (licence no. 574/27.03.2014).
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Handling Editor: Bill Chobotar
Rights and permissions
About this article
Cite this article
Emmenegger, T., Bauer, S., Dimitrov, D. et al. Host migration strategy and blood parasite infections of three sparrow species sympatrically breeding in Southeast Europe. Parasitol Res 117, 3733–3741 (2018). https://doi.org/10.1007/s00436-018-6072-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00436-018-6072-7