Abstract
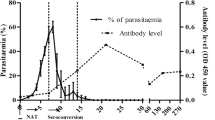
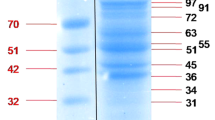
Babesiosis among humans is on the rise in North America. Current diagnostic assays for the screening of babesiosis require blood collection by venipuncture, which is an invasive method. Urine on the other hand is a desirable biospecimen for biomarker analysis of Babesia microti infections because it can be collected periodically and non-invasively. Our group uses a new class of biomarker harvesting nanocage technology, which, when combined with mass spectrometry (MS), can determine the presence of parasite proteins shed in different bodily fluids of mammalian hosts, including urine. Using the hamster model of babesiosis, our nanoparticle-MS approach identified several B. microti proteins in erythrocytes, plasma, and urine samples. Surface and secreted antigens previously shown to elicit host immune responses against the parasite were particularly abundant in erythrocytes and plasma compared to other proteins. Two of these antigens, BmSA1 and BMR1_03g00947, showed different localization patterns by immunofluorescence of infected erythrocytes. Hamster urine samples from parasitemic animals harbored lower numbers of B. microti proteins compared to erythrocytes and plasma, with glycolytic enzymes, cytoskeletal components, and chaperones being the most frequently detected proteins. By applying novel nanoparticle-MS methods, a high level of analytical sensitivity can be achieved to detect multiple B. microti proteins in blood and urine. This is generally difficult to obtain with other techniques due to the masking of parasite biomarkers by the complex biomolecular matrix of bodily fluids from the host.





Similar content being viewed by others
References
Blum S, Gattringer R, Haschke E, Walochnik J, Tschurtschenthaler G, Lang F, Oberbauer R (2011) The case: hemolysis and acute renal failure. Babesiosis Kidney Int 80(6):681–683. https://doi.org/10.1038/ki.2011.184
Chan K, Marras SA, Parveen N (2013) Sensitive multiplex PCR assay to differentiate Lyme spirochetes and emerging pathogens Anaplasma phagocytophilum and Babesia microti. BMC Microbiol 13:295. https://doi.org/10.1186/1471-2180-13-295
Cullen JM, Levine JF (1987) Pathology of experimental Babesia microti infection in the Syrian hamster. Lab Anim Sci 37(5):640–643
Douglas TA, Tamburro D, Fredolini C, Espina BH, Lepene BS, Ilag L, Espina V, Petricoin EF, Liotta LA, Luchini A (2011) The use of hydrogel microparticles to sequester and concentrate bacterial antigens in a urine test for Lyme disease. Biomaterials 32(4):1157–1166. https://doi.org/10.1016/j.biomaterials.2010.10.004
Filbin MR, Mylonakis EE, Callegari L, Legome E (2001) Babesiosis. J Emerg Med 20(1):21–24
Fredolini C, Meani F, Reeder KA, Rucker S, Patanarut A, Botterell PJ, Bishop B, Longo C, Espina V, Petricoin EF, Liotta LA, Luchini A (2008) Concentration and preservation of very low abundance biomarkers in urine, such as human growth hormone (hGH), by Cibacron blue F3G-A loaded hydrogel harticles. Nano Res 1(6):502–518. https://doi.org/10.1007/s12274-008-8054-z
Gray J, von Stedingk LV, Gurtelschmid M, Granstrom M (2002) Transmission studies of Babesia microti in Ixodes ricinus ticks and gerbils. J Clin Microbiol 40(4):1259–1263
Gray EB, Niknafs AM, Herwaldt BL (2019) Surveillance for Babesiosis - United States, 2017. Annual Summary. https://www.cdc.gov/parasites/babesiosis/resources/babesiosis_surveillance_summary_2017b.pdf. Accessed 5 Jan 2020
Kim B, Araujo R, Howard M, Magni R, Liotta LA, Luchini A (2018) Affinity enrichment for mass spectrometry: improving the yield of low abundance biomarkers. Expert Rev Proteomics 15(4):353–366. https://doi.org/10.1080/14789450.2018.1450631
Krause PJ (2019) Human babesiosis. Int J Parasitol 49(2):165–174. https://doi.org/10.1016/j.ijpara.2018.11.007
Lobo CA, Cursino-Santos JR, Alhassan A, Rodrigues M (2013) Babesia: an emerging infectious threat in transfusion medicine. PLoS Pathog 9(7):e1003387. https://doi.org/10.1371/journal.ppat.1003387
Longo C, Patanarut A, George T, Bishop B, Zhou W, Fredolini C, Ross MM, Espina V, Pellacani G, Petricoin EF, Liotta LA (2009) Core-shell hydrogel particles harvest, concentrate and preserve labile low abundance biomarkers. PLoS One 4(3):e4763. https://doi.org/10.1371/journal.pone.0004763
Magni R, Espina BH, Shah K, Lepene B, Mayuga C, Douglas TA, Espina V, Rucker S, Dunlap R, Petricoin EF, Kilavos MF, Poretz DM, Irwin GR, Shor SM, Liotta LA, Luchini A (2015) Application of Nanotrap technology for high sensitivity measurement of urinary outer surface protein a carboxyl-terminus domain in early stage Lyme borreliosis. J Transl Med 13:346. https://doi.org/10.1186/s12967-015-0701-z
Magni R, Luchini A, Liotta L, Molestina RE (2019) Analysis of the Babesia microti proteome in infected red blood cells by a combination of nanotechnology and mass spectrometry. Int J Parasitol 49(2):139–144. https://doi.org/10.1016/j.ijpara.2018.08.004
Mylonakis E (2001) When to suspect and how to monitor babesiosis. Am Fam Physician 63(10):1969–1974
Oz HS, Hughes WT (1996) Acute fulminating babesiosis in hamsters infected with Babesia microti. Int J Parasitol 26(6):667–670
Piesman J, Karakashian SJ, Lewengrub S, Rudzinska MA, Spielmank A (1986) Development of Babesia microti sporozoites in adult Ixodes dammini. Int J Parasitol 16(4):381–385
Silva JC, Cornillot E, McCracken C, Usmani-Brown S, Dwivedi A, Ifeonu OO, Crabtree J, Gotia HT, Virji AZ, Reynes C, Colinge J, Kumar V, Lawres L, Pazzi JE, Pablo JV, Hung C, Brancato J, Kumari P, Orvis J, Tretina K, Chibucos M, Ott S, Sadzewicz L, Sengamalay N, Shetty AC, Su Q, Tallon L, Fraser CM, Frutos R, Molina DM, Krause PJ, Ben Mamoun C (2016) Genome-wide diversity and gene expression profiling of Babesia microti isolates identify polymorphic genes that mediate host-pathogen interactions. Sci Rep 6:35284. https://doi.org/10.1038/srep35284
Tonkin CJ, van Dooren GG, Spurck TP, Struck NS, Good RT, Handman E, Cowman AF, McFadden GI (2004) Localization of organellar proteins in Plasmodium falciparum using a novel set of transfection vectors and a new immunofluorescence fixation method. Mol Biochem Parasitol 137(1):13–21. https://doi.org/10.1016/j.molbiopara.2004.05.009
Tonnetti L, Young C, Kessler DA, Williamson PC, Reik R, Proctor MC, Bres V, Deisting B, Bakkour S, Schneider W, Diner S, Busch MP, Stramer SL, Linnen JM (2019) Transcription-mediated amplification blood donation screening for Babesia. Transfusion. https://doi.org/10.1111/trf.15630
Vannier EG, Diuk-Wasser MA, Ben Mamoun C, Krause PJ (2015) Babesiosis. Infect Dis Clin N Am 29(2):357–370. https://doi.org/10.1016/j.idc.2015.02.008
Wang G, Villafuerte P, Zhuge J, Visintainer P, Wormser GP (2015) Comparison of a quantitative PCR assay with peripheral blood smear examination for detection and quantitation of Babesia microti infection in humans. Diagn Microbiol Infect Dis 82(2):109–113. https://doi.org/10.1016/j.diagmicrobio.2015.03.010
Wilson M, Glaser KC, Adams-Fish D, Boley M, Mayda M, Molestina RE (2015) Development of droplet digital PCR for the detection of Babesia microti and Babesia duncani. Exp Parasitol 149:24–31. https://doi.org/10.1016/j.exppara.2014.12.003
Acknowledgments
We thank Debra Adams-Fish from the ATCC® Specialty Laboratory Facilities for her assistance in the animal experiments. ATCC® is a registered trademark of the American Type Culture Collection.
Funding
R.E. Molestina was supported by the ATCC® Internal Research and Development Program. A. Luchini, L. Liotta, and R. Magni were supported by grants W81XWH-17-1-0175 from the US Army Medical Research Acquisition Activity, and R21HD097472, R01AR068436, and R21AI138135 from the US National Institutes of Health.
Author information
Authors and Affiliations
Contributions
R.E. Molestina contributed to the study conception and design, animal experiments, biological material preparation, results figures, and first draft of the manuscript. Nanoparticle synthesis, protein capture, MS analysis, and data collection were performed by R. Magni. All authors commented on previous versions of the manuscript, provided edits, and read and approved the final manuscript.
Corresponding author
Additional information
Section Editor: Julia Walochnik
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Magni, R., Luchini, A., Liotta, L. et al. Proteomic analysis reveals pathogen-derived biomarkers of acute babesiosis in erythrocytes, plasma, and urine of infected hamsters. Parasitol Res 119, 2227–2235 (2020). https://doi.org/10.1007/s00436-020-06712-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00436-020-06712-5