Abstract
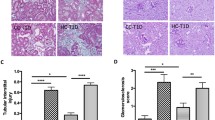
Although several studies have focused on the effects of nutritional status during intrauterine development, few have addressed the impact of maternal diabetes mellitus on renal function and morphology in the young offspring. In the present study, renal morpho-functional aspects were studied in the offspring of diabetic rats. Diabetes was induced in female rats with a single dose of streptozotocyn (STZ), 10 days before mating. After weaning, the offspring (DO) had free access to food and water. Arterial blood pressure was measured, by tail plethysmography, from 2 months on. Renal function was evaluated in 2- and 3-month-old rats in the DO group and in controls (C). Analysis of renal morphology was carried out in newborn and in 1-, 2- and 3-month-old rats in both groups. Although the nephron number was not changed in the DO group, glomerular hypertrophy was observed from 2 months on. At the same age, the glomerular filtration rate was significantly reduced in DO, and blood pressure was significantly increased, when compared to C. Glucose tolerance test (GTT) from DO showed a different profile when compared to C. The number of PCNA positive cells in renal tissue was similar in both groups. Our data suggests that exposure to intrauterine diabetes may be an important cause of both impaired renal function and hypertension in offspring, without changes in the nephron number.


Similar content being viewed by others
References
Barker DJ, Osmond C, Golding J, Kuh D, Wadsworth ME (1989) Growth in utero, blood pressure in childhood and adult life and mortality from cardiovascular disease. BMJ 298:564–567
Barker DJ (1995) Fetal origins of coronary heart disease. BMJ 311:171–174
Campbell DM, Hall MH, Barker DJ, Cross J, Shiell AW, Godfrey KM (1996) Diet in pregnancy and the offspring’s blood pressure 40 years later. Br J Obstet Gynaecol 103:273–280
Barker DJ (1998) In utero programming of chronic disease. Clin Sci 95:115–128
Pedersen L, Tygstrup I, Pedersen J (1964) Congenital malformations in newborn infants of diabetic women. Lancet 13:1124–1126
Martinez-Frias ML (1994) Epidemiological analysis of outcomes of pregnancy in diabetic mothers:identification of the most frequent congenital anomalies. Am J Med Gen 51:108–113
Travers J, Pratten M, Beck F (1989) Effects of low insulin levels on rat embryonic growth and development. Diabetes 38:773–778
Amri K, Freund N, Vilar J, Merlet-Benichou C, Lelièvre-Pegorier M (1999) Adverse effects of hyperglycemia on kidney development in rats. Diabetes 48:2240–2245
Hoy WE, Rees M, Kile E, Mathews JD, Wang Z (1999) A new dimension to the Barker hypothesis: low birth weight and susceptibility to renal disease. Kidney Int 56:1072–1077
Rocha SO, Gomes GN, Forti ALL, Franco MCP, Fortes ZB, Cavanal MF, Gil FZ (2005) Long-term effects of maternal diabetes on vascular reactivity and renal function in rat male offspring. Pediatr Res 58:1274–1279
Brenner BM, Cheetow GM (1994) Congenital oligonephropathy and the etiology of adult hypertension and progressive renal injury. Am J Kidney Dis 23:171–175
Brenner BM, Garcia DL, Anderson S (1988) Glomeruli and blood pressure. Less of one, more of the other? Am J Hypertension 1:335–347
Lucas SR, Zaladek Gil F, Costa Silva VL, Miraglia SM (1991) Functional and morphometric evaluation of intrauterine under-nutrition on kidney development of the progeny. Braz J Med Biol Res 24:967–970
Lucas SR, Miraglia SM, Zaladek Gil F, Coimbra TM (2001) Intrauterine food restriction as a determinant of nephrosclerosis. Am J Kidney Dis 37:467–476
Malavazi AG, Barão MA, Gomes GN, Odo LN, Franco MC, Nigro D, Lucas SR, Laurindo FR, Brandizzi LI, Zaladek Gil F (2002) L-arginine effects on blood pressure and renal function of intrauterine restricted rats. Pediatr Nephrol 17:856–862
Gil FZ, Lucas SR, Gomes GN, Cavanal MF, Coimbra TR (2005) Effects of intrauterine food restriction and long-term dietary supplementation with L-arginine on age-related changes in renal function and structure of rats. Pediatr Res 57:724–731
Nascimento-Gomes G, Zaladek Gil F, Mello-Aires M (1997) Alterations of the renal handling of H+ in diabetic rats. Kidney Blood Press Res 20:251–257
Franco MC, Akamine EH, Di Marco GS, Casarini DE, Fortes ZB, Tostes RC, Carvalho MH, Nigro D (2003) NADPH oxidase and enhanced superoxide generation in intrauterine undernourished rats: involvement of the renin-angiotensin system. Cardiovasc Res 59:767–775
Nold JL, Georgieff MK (2004) Infants of diabetic mothers. Pediatr Clin North Am 51:619–637
Carey AV, Carey RM, Gomez RA (1992) Expression of alpha smooth muscle actin in the developing kidney vasculature. Hypertension 19(suppl II):168–175
Hall PA, Levison DA, Woods AL, Yu CC, Kellock DB, Watkins JA, Barnes DM, Gillett CE, Camplejohn R, Dover R (1990) Proliferating cell nuclear antigen (PCNA) immunolocalization in paraffin sections: the index of cell proliferation with evidence of deregulated expression in some neoplasms. J Pathol 162:285–294
Srinivasan M, Aalinkeel R, Song F, Patel MS (2003) Programming of islet functions in the progeny of hyperinsulinemic/obese rats. Diabetes 52:984–990
Marcantoni C, Jafar TH, Oldrizzi L, Levey AS, Maschio G (2000) The role of systemic hypertension in the progression of nondiabetic renal disease. Kidney Int Suppl 75:S44–S48
Tolbert EM, Weisstuch J, Feiner HD, Dworkin LD (2000) Onset of glomerular hypertension with aging precedes injury in the spontaneously hypertensive rat. Am J Physiol Renal Physiol 278:F839–F846
Moeller I, Allen AM, Chai SY, Zhuo J, Mendelsohn FA (1998) Bioactive angiotensin peptides. J Hum Hypertens 12:289–293
Lemos VS, Silva DM, Walther T, Alenina N, Bader M, Santos RA (2005) The endothelium-dependent vasodilator effect of the nonpeptide Ang(1–7) mimic AVE 0991 is abolished in the aorta of mas-knockout mice. J Cardiovasc Pharmacol 46:274–279
Li P, Chappell MC, Ferrario CM, Brosnihan KB (1997) Angiotensin-(1–7) augments bradykinin-induced vasodilation by competing with ACE and releasing nitric oxide. Hypertension 29:394–400
Woods LL, Weeks DA, Rasch R (2001) Hypertension after neonatal uninephrectomy in rats precedes glomerular damage. Hypertension 38:337–342
Acknowledgements
This study was supported by FAPESP (Fundação de Amparo à Pesquisa do Estado de São Paulo) and CNPq (Conselho Nacional de Desenvolvimento Científico e Tecnológico, Brazil).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Magaton, A., Gil, F.Z., Casarini, D.E. et al. Maternal diabetes mellitus - early consequences for the offspring. Pediatr Nephrol 22, 37–43 (2007). https://doi.org/10.1007/s00467-006-0282-4
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00467-006-0282-4