Abstract
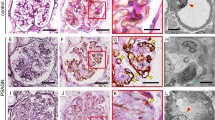
Cellular crescents are defined as two or more layers of proliferating cells in Bowman’s space and are a hallmark of inflammatory active glomerulonephritis and a histologic marker of severe glomerular injury. In general, the percentage of glomeruli that exhibit crescents correlates with the severity of kidney failure and other clinical manifestations of nephritic syndrome. In general, a predominance of active crescents is associated with rapidly progressive glomerulonephritis and a poor outcome. The duration and potential reversibility of the underlying disease correspond with the relative predominance of cellular or fibrous components in the crescents, the initial location of the immunologic insult inside the glomerulus, and the sort of involved cells and inflammatory mediators. However, the presence of active crescents may not have the same degree of significance in the different types of glomerulopathies. The pathophysiology of parietal cell proliferation may have dissimilar origins, underscoring the fact that the resultant crescents are a non-specific morphological pattern of glomerular injury with different implications in clinical prognosis in the scope of glomerular diseases.

Similar content being viewed by others
References
Anguiano L, Kain R, Anders HJ (2020) The glomerular crescent: triggers, evolution, resolution, and implications for therapy. Curr Opin Nephrol Hypertens 29:302–309
Eymael J, Sharma S, Loeven MA, Wetzels JF, Mooren F, Florquin S, Deegens JK, Willemsen BK, Sharma V, van Kuppevelt TH, Bakker MA, Ostendorf T, Moeller MJ, Dijkman HB, Smeets B, van der Vlag J (2018) CD44 is required for the pathogenesis of experimental crescentic glomerulonephritis and collapsing focal and segmental glomerulosclerosis. Kidney Int 93:626–642
Fogo AB, Lusco MA, Najafian B, Alpers CE (2016) AJKD Atlas of renal pathology: pauci-immune necrotizing crescentic glomerulonephritis. Am J Kidney Dis 68:e31–e32
Puelles VG, Fleck D, Ortz L, Papadouri S et al (2019) Novel 3D analysis using optical tissue clearing documents the evolution of murine rapidly progressive glomerulonephritis. Kidney Int 96:505–516
Lan HY, Nikolic-Paterson DJ, Atkins RC (1992) Involvement of activated periglomerular leukocytes in the rupture of Bowman’s capsule and glomerular crescent progression in experimental glomerulonephritis. Lab Investig 67:743–751
Jennette JC (2003) Rapidly progressive crescentic glomerulonephritis. Kidney Int 63:1164–1177
Kitching AR, Holdsworth SR, Tipping PG (1999) IFN-gamma mediates crescent formation and cell-mediated immune injury in murine glomerulonephritis. J Am Soc Nephrol 10:752–759
Hopfer H, Holzer J, Hünemörder S, Paust HJ, Sachs M, Meyer-Schwesinger C, Turner JE, Panzer U, Mittrücker HW (2012) Characterization of the renal CD4+ T-cell response in experimental autoimmune glomerulonephritis. Kidney Int 82:60–71
Kluger MA, Luig M, Wegscheid C, Goerke B et al (2014) Stat3 programs Th17-specific regulatory T cells to control GN. J Am Soc Nephrol 25:1291–1302
Giorgini A, Brown HJ, Sacks SH, Robson MG (2010) Toll-like receptor 4 stimulation triggers crescentic glomerulonephritis by multiple mechanisms including a direct effect on renal cells. Am J Pathol 177:644–653
Naish P, Penn GB, Evans DJ, Peters DK (1972) The effect of defibrination on nephrotoxic serum nephritis in rabbits. Clin Sci 42:643–646
Tipping PG, Erlich JH, Apostolopoulos J, Mackman N, Loskutoff D, Holdsworth SR (1995) Glomerular tissue factor expression in crescentic glomerulonephritis. Correlations between antigen, activity, and mRNA. Am J Pathol 147:1736–1748
Cunningham MA, Kitching AR, Tipping PG, Holdsworth SR (2004) Fibrin independent proinflammatory effects of tissue factor in experimental crescentic glomerulonephritis. Kidney Int 66:647–654
Kitching AR, Holdsworth SR, Ploplis VA, Plow EF, Collen D, Carmeliet P, Tipping PG (1997) Plasminogen and plasminogen activators protect against renal injury in crescentic glomerulonephritis. J Exp Med 185:963–968
Atkins RC, Nikolic-Paterson DJ, Song Q, Lan HY (1996) Modulators of crescentic glomerulonephritis. J Am Soc Nephrol 7:2271–2278
Moussa L, Apostolopoulos J, Davenport P, Tchongue J, Tipping PG (2007) Protease-activated receptor-2 augments experimental crescentic glomerulonephritis. Am J Pathol 171:800–808
Tipping PG, Holdsworth SR (1986) The participation of macrophages, glomerular procoagulant activity, and factor VIII in glomerular fibrin deposition. Studies on anti-GBM antibody-induced glomerulonephritis in rabbits. Am J Pathol 124:10–17
Lloyd CM, Dorf ME, Proudfoot A, Salant DJ, Gutierrez-Ramos JC (1997) Role of MCP-1 and RANTES in inflammation and progression to fibrosis during murine crescentic nephritis. J Leukoc Biol 62:676–680
Segerer S, Cui Y, Hudkins KL, Goodpaster T, Eitner F, Mack M, Schlöndorff D, Alpers CE (2000) Expression of the chemokine monocyte chemoattractant protein-1 and its receptor chemokine receptor 2 in human crescentic glomerulonephritis. J Am Soc Nephrol 11:2231–2242
Nishikawa K, Guo YJ, Miyasaka M, Tamatani T, Collins AB, Sy MS, McCluskey RT, Andres G (1993) Antibodies to intercellular adhesion molecule 1/lymphocyte function-associated antigen 1 prevent crescent formation in rat autoimmune glomerulonephritis. J Exp Med 177:667–677
Timoshanko JR, Kitching AR, Semple TJ, Holdsworth SR, Tipping PG (2005) Granulocyte macrophage colony-stimulating factor expression by both renal parenchymal and immune cells mediates murine crescentic glomerulonephritis. J Am Soc Nephrol 16:2646–2656
Song CY, Kim BC, Hong HK, Lee HS (2007) TGF-beta type II receptor deficiency prevents renal injury via decrease in ERK activity in crescentic glomerulonephritis. Kidney Int 71:882–888
Han Y, Ma FY, Tesch GH, Manthey CL, Nikolic-Paterson DJ (2013) Role of macrophages in the fibrotic phase of rat crescentic glomerulonephritis. Am J Physiol Ren Physiol 304:F1043–F1053
Sanders JS, van Goor H, Hanemaaijer R, Kallenberg CG, Stegeman CA (2004) Renal expression of matrix metalloproteinases in human ANCA-associated glomerulonephritis. Nephrol Dial Transplant 19:1412–1419
Hochheiser K, Engel DR, Hammerich L, Heymann F, Knolle PA, Panzer U, Kurts C (2011) Kidney dendritic cells become pathogenic during crescentic glomerulonephritis with proteinuria. J Am Soc Nephrol 22:306–316
Evers BD, Engel DR, Böhner AM, Tittel AP, Krause TA, Heuser C, Garbi N, Kastenmüller W, Mack M, Tiegs G, Panzer U, Boor P, Ludwig-Portugall I, Kurts C (2016) CD103+ kidney dendritic cells protect against crescentic GN by maintaining IL-10-producing regulatory T cells. J Am Soc Nephrol 27:3368–3382
Li HL, Hancock WW, Dowling JP, Atkins RC (1991) Activated (IL-2R+) intraglomerular mononuclear cells in crescentic glomerulonephritis. Kidney Int 39:793–798
Kitching AR, Turner AL, Wilson GR, Semple T, Odobasic D, Timoshanko JR, O'Sullivan KM, Tipping PG, Takeda K, Akira S, Holdsworth SR (2005) IL-12p40 and IL-18 in crescentic glomerulonephritis: IL-12p40 is the key Th1-defining cytokine chain, whereas IL-18 promotes local inflammation and leukocyte recruitment. J Am Soc Nephrol 16:2023–2033
Smeets B, Uhlig S, Fuss A, Mooren F, Wetzels JF, Floege J, Moeller MJ (2009) Tracing the origin of glomerular extracapillary lesions from parietal epithelial cells. J Am Soc Nephrol 20:2604–2615
Shirato I, Asanuma K, Takeda Y, Hayashi K, Tomino Y (2000) Protein gene product 9.5 is selectively localized in parietal epithelial cells of Bowman’s capsule in the rat kidney. J Am Soc Nephrol 11:2381–2386
Fujigaki Y, Sun DF, Fujimoto T, Suzuki T, Goto T, Yonemura K, Morioka T, Yaoita E, Hishida A (2002) Mechanisms and kinetics of Bowman’s epithelial-myofibroblast transdifferentiation in the formation of glomerular crescents. Nephron 92:201–213
Appel D, Kershaw DB, Smeets B, Yuan G, Fuss A, Frye B, Elger M, Kriz W, Floege J, Moeller MJ (2009) Recruitment of podocytes from glomerular parietal epithelial cells. J Am Soc Nephrol 20:333–343
Le Hir M, Keller C, Eschmann V, Hähnel B, Hosser H, Kriz W (2001) Podocyte bridges between the tuft and Bowman’s capsule: an early event in experimental crescentic glomerulonephritis. J Am Soc Nephrol 12:2060–2071
Bariéty J, Bruneval P, Meyrier A, Mandet C, Hill G, Jacquot C (2005) Podocyte involvement in human immune crescentic glomerulonephritis. Kidney Int 68:1109–1119
Thorner PS, Ho M, Eremina V, Sado Y, Quaggin S (2008) Podocytes contribute to the formation of glomerular crescents. J Am Soc Nephrol 19:495–502
Ronconi E, Sagrinati C, Angelotti ML, Lazzeri E, Mazzinghi B, Ballerini L, Parente E, Becherucci F, Gacci M, Carini M, Maggi E, Serio M, Vannelli GB, Lasagni L, Romagnani S, Romagnani P (2009) Regeneration of glomerular podocytes by human renal progenitors. J Am Soc Nephrol 20:322–332
Succar L, Boadle RA, Harris DC, Rangan GK (2016) Formation of tight junctions between neighboring podocytes is an early ultrastructural feature in experimental crescentic glomerulonephritis. Int J Nephrol Renovasc Dis 9:297–312
Kriz W, Hähnel B, Hosser H, Ostendorf T, Gaertner S, Kränzlin B, Gretz N, Shimizu F, Floege J (2003) Pathways to recovery and loss of nephrons in anti-Thy-1 nephritis. J Am Soc Nephrol 14:1904–1926
Tam FWK, Smith J, Morel D, Karkar AM, Thompson EM, Cook HT, Pusey CD (1999) Development of scarring and renal failure in a rat model of crescentic glomerulonephritis. Nephrol Dial Transplant 14:1658–1666
Kitching AR, Aikhan MA (2018) CD8+ cells and glomerular crescent formation: outside-in as well as inside-out. J Clin Invest 128:3231–3233
Haas M, Verhave JC, Liu ZH, Alpers CE, Barratt J, Becker JU, Cattran D, H. Cook T, Coppo R, Feehally J, Pani A, Perkowska-Ptasinska A, Roberts ISD, Soares MF, Trimarchi H, Wang S, Yuzawa Y, Zhang H, Troyanov S, Katafuchi R (2017) The predictive value of crescents in IgA nephropathy: a large retrospective multicenter study. J Am Soc Nephrol 28:691–701
Trimarchi H, Barratt J, Cattran DC, Cook HT, Coppo R, Haas M, Liu ZH, Roberts ISD, Yuzawa Y, Zhang H, Feehally J (2017) Oxford classification for IgA nephropathy 2016. An update from the IgA Nephropathy Classification Working Group. Kidney Int 91:1014–1021
Couser WG (1988) Rapidly progressive glomerulonephritis: classification, pathogenetic mechanisms, and therapy. Am J Kidney Dis 11:449–464
Rizzo P, Novelli R, Rota C, Gagliardini E, Ruggiero B, Rottoli D, Benigni A, Remuzzi G (2017) The role of angiotensin II in parietal epithelial cell proliferation and crescent formation in glomerular diseases. Am J Pathol 187:1441–1450
Rizzo P, Perico N, Gagliardini E, Novelli R, Alison MR, Remuzzi G, Benigni A (2013) Nature and mediators of parietal epithelial cell activation in glomerulonephritides of human and rat. Am J Pathol 183:1769–1778
Kim S, Iwao H (2000) Molecular and cellular mechanisms of angiotensin II-mediated cardiovascular and renal diseases. Pharmacol Rev 52:11–34
Kaneko Y, Sakatsume M, Xie Y, Kuroda T, Igashima M, Narita I, Gejyo F (2003) Macrophage metalloelastase as a major factor for glomerular injury in anti-glomerular basement membrane nephritis. J Immunol 170:3377–3385
Ryu M, Migliorini A, Miosge N, Gross O, Shankland S, Brinkkoetter PT, Hagmann H, Romagnani P, Liapis H, Anders HJ (2012) Plasma leakage through glomerular basement membrane ruptures triggers the proliferation of parietal epithelial cells and crescent formation in non-inflammatory glomerular injury. J Pathol 228:482–494
Moeller MJ, Soofi A, Hartmann I, Le Hir M, Wiggins R, Kriz W, Holzman LB (2004) Podocytes populate cellular crescents in a murine model of inflammatory glomerulonephritis. J Am Soc Nephrol 15:61–67
Yoshioka K, TakemuraT AN, Miyamoto H, Iseki T, Maki S (1987) Cellular and non-cellular compositions of crescents in human glomerulonephritis. Kidney Int 32:284–291
Itami H, Hara S, Samejima K, Tsushima H, Morimoto K, Okamoto K, Kosugi T, Kawano T, Fujiki K, Kitada H, Hatakeyama K, Tsuruya K, Ohbayashi C (2020) Complement activation is associated with crescent formation in IgA nephropathy. Virchows Arch 477:565–572
Roos A, Rastaldi MP, Calvaresi N, Oortwijn BD, Schlagwein N, van Gijlswijk-Janssen DJ, Stahl GL, Matsushita M, Fujita T, van Kooten C, Daha MR (2006) Glomerular activation of the lectin pathway of complement in IgA nephropathy is associated with more severe renal disease. J Am Soc Nephrol 17:1724–1734
Hashimoto A, Suzuki Y, Suzuki H, Ohsawa I, Brown R, Hall S, Tanaka Y, Novak J, Ohi H, Tomino Y (2012) Determination of severity of murine IgA nephropathy by glomerular complement activation by aberrantly glycosylated IgA and immune complexes. Am J Pathol 181:1338–1347
Daha MR, van Kooten C (2000) Is there a role for locally produced complement in renal disease? Nephrol Dial Transplant 15:1506–1509
Lazareth H, Henique C, Lenoir O, Puelles VG et al (2019) The tetraspanin CD9 controls migration and proliferation of parietal epithelial cells and glomerular disease progression. Nat Commun 10:3303
Jennette JC, Nachman PH (2017) ANCA glomerulonephritis and vasculitis. Clin J Am Soc Nephrol 12:1680–1691
Ranasinghe R, Eri R (2018) Modulation of the CCR6-CCL20 axis: a potential therapeutic target in inflammation and cancer. Medicina 54:88
Jia ZJ, Wu FX, Huang QH, Liu JM (2012) Toll-like receptor 4: the potential therapeutic target for neuropathic pain. Zhongguo Yi Xue Ke Xue Yuan Xue Bao 34:168–173
Monnet E, Lapeyre G, Poelgeest EV, Jacqmin P, Graaf K, Reijers J, Moerland M, Burggraaf J, Min C (2017) Evidence of NI-0101 pharmacological activity, an anti-TLR4 antibody, in a randomized phase I dose escalation study in healthy volunteers receiving LPS. Clin Pharmacol Ther 101:200–208
Li L, Ni L, Heary RF, Elkabes S (2020) Astroglial TLR9 antagonism promotes chemotaxis and alternative activation of macrophages via modulation of astrocyte derived signals: implications for spinal cord injury. J Neuroinflammation 17:73
Yao W, Ba Q, Li X, Li H, Zhang S, Yuan Y, Wang F, Duan X, Li J, Zhang W, Wang H (2017) A natural CCR2 antagonist relieves tumor-associated macrophage-mediated immunosuppression to produce a therapeutic effect for liver cancer. EBioMedicine 22:58–67
de Zeeuw D, Bekker P, Henkel E, Hasslacher C, Gouni-Berthold I et al (2015) The effect of CCR2 inhibitor CCX140-B on residual albuminuria in patients with type 2 diabetes and nephropathy: a randomised trial. Lancet Diabetes Endocrinol 3:687–696
Huang X, Ni B, Xi Y (2019) Protease-activated receptor 2 (PAR-2) antagonist AZ3451 as a novel therapeutic agent for osteoarthritis. Aging 11:12533–12545
Author information
Authors and Affiliations
Contributions
The author entirely contributed to the development of the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The author declares no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Trimarchi, H. Crescents in primary glomerulonephritis: a pattern of injury with dissimilar actors. A pathophysiologic perspective. Pediatr Nephrol 37, 1205–1214 (2022). https://doi.org/10.1007/s00467-021-05199-1
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00467-021-05199-1