Abstract
Key message
In Hancornia speciosa, the latex is synthesized in the cytosol of the ground meristem cells and in plastids of the fusiform derivatives of secondary phloem. Laticifer development is related to climatic seasonality.
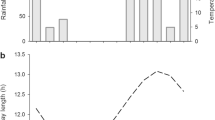
Hancornia speciosa is a latescent species that grows in the Brazilian Cerrado (neotropical savanna) and shows significant potential for rubber production and the extraction of bioactive compounds from its latex. We examined the development of laticifers in its stem apex and secondary phloem in relation to climate seasonality. Morphometric evaluations of elongating branches and micromorphometric evaluations of the cambial zone were carried out monthly for 1 year, with structural and ultrastructural analyses of the stem apex and secondary phloem. Laticifer development in both the stem apex and secondary phloem is related to increasing day length and maximum temperature and humidity. Laticifers are formed by anastomosis of the transverse and longitudinal walls of the ground meristem cells of the stem apex, and the fusiform derivatives of secondary phloem in the cambial zone. The process of latex secretion involves the synthesis of terpenic droplets in the cytoplasm of the ground meristem cells of the stem apex and in the plastids of the fusiform derivatives of secondary phloem. Latex is an emulsion formed by the cytosol terpenic droplets engulfed by vacuoles contaning mucilages and proteins secreted by dictyosomes and rough endoplasmic reticulum, and alkaloids secreted by the phloem cells. This work expands our knowledge concerning the development of laticifers in Apocynaceae and contributes to our better understanding of the influence of environmental factors on latex secretion.








Similar content being viewed by others
References
Almeida LM, Floriano JF, Ribeiro TP, Magno LN, Mota LSLS, Peixoto N, Mrué F, Melo-Reis P, Junior RSL, Graeff CFO (2014) Hancornia speciosa latex for biomedical applications: physical and chemical properties, biocompatibility assessment and angiogenic activity. J Mater Sci Mater Med 25:2153–2162. https://doi.org/10.1007/s10856-014-5255-8
Almeida AL, de Freitas PF, Ferreira CP, Ventrella MC (2020) Syncytial development of annatto (Bixa orellana L.) pigment gland: a curious type of anastomosed articulated laticifer. Flora 274:151727. https://doi.org/10.1016/j.flora.2020.151727
Alonso AAA, Mendonça MS, Reis RS, Biondo PLTA, Alonso RRP (2013) Laticifers distribution in secondary phloem of the Amazon wood species. Comunicata Scientiae 4:212–215
APG (2016) An update of the Angiosperm Phylogeny Group classification for the orders and families of flowering plants: APG IV. Bot J Lin Soc 181:1–20. https://doi.org/10.1111/boj.12385
Aref IM, Khan PR, Al-Mefarrej H, Al-Shahrani T, Ismail A, Iqbal M (2014) Cambial periodicity and wood production in Acacia ehrenbergiana Hayne growing on dry sites of Saudi Arabia. J Environ Biol 35:301–310
BFG (2015) Growing knowledge: an overview of Seed Plant diversity in Brazil. Rodriguésia 66:1085–1113. https://doi.org/10.1590/2175-7860201566411
Cai X, Li W, Yin L (2009) Ultrastructure and cytochemical localization of acid phosphatase of laticifers in Euphorbia kansui Liou. Protoplasma 238:3–10. https://doi.org/10.1007/s00709-009-0065-4
Cain AJ (1947) The use of Nile Blue in the examination of lipoids. J Cell Sci 3:383–392
Calle Z, Schlumpberger BO, Piedrahita L, Leftin A, Hammer SA, Tye A, Borchert R (2010) Seasonal variation in daily insolation induces synchronous bud break and flowering in the tropics. Trees 24:865–877. https://doi.org/10.1007/s00468-010-0456-3
Canaveze Y, Machado SR (2016) The occurrence of intrusive growth associated with articuled laticifers in Tabernaemontana catharinensis A.DC. A new record for Apocynaceae. Int J Plant Sci 177:1–23. https://doi.org/10.1086/685446
Canaveze Y, Mastroberti AA, Mariath JEA, Machado SR (2019) Cytological differentiation and cell wall involvement in the growth mechanisms of articulated laticifers in Tabernaemontana catharinensis A.DC. (Apocynaceae). Protoplasma 256:131–146. https://doi.org/10.1007/s00709-018-1284-3
Castelblanque L, Balaguer B, Marti C, Rodríguez JJ, Orozco M, Vera P (2016) Novel insights into the organization of laticifer cells: a cell comprising a unified whole system. Plant Physiol 172:1032–1044. https://doi.org/10.1104/pp.16.00954
David R, Carde JP (1964) Coloration différentielle dês inclusions lipidique et terpéniques des pseudophylles du pine maritime au moyen du réactif Nadi. CR Acad Sci Paris, Série D 258:1338–1340
Fahn A (1979) Secretory tissues in plants. Academic Press, San Francisco
Freitas CDT, Souza DP, Araújo ES, Cavalheiro MG, Oliveira LS, Ramos MV (2010) Anti-oxidative and proteolytic activities and protein profile of laticifer cells of Cryptostegia grandiflora, Plumeria rubra and Euphorbia tirucalli. Braz J Plant Physiol 22:11–22. https://doi.org/10.1590/S1677-04202010000100002
Gama TSS, Rubiano VS, Demarco D (2017) Laticifer development and its growth mode in Allamanda blanchetii ADC (Apocynaceae). J Torrey Bot Soc 144:303–312. https://doi.org/10.3159/Torrey-D-16-00050
Gimenez AM (2004) Anatomía de leño y corteza de Tabernaemontana catharinensis A. DC (Apocynaceae). Quebracho 11:22–32
Gonçalves MP, Mercadante-Simões MO, Ribeiro LM (2018) Ontogeny of anastomosed laticifers in the stem apex of Hancornia speciosa (Apocynaceae): a topographic approach. Protoplasma 255:1713–1724. https://doi.org/10.1007/s00709-018-1262-9
Gondaliya AD, Rajput KS (2016a) Stem anatomy and development of intraxylary phloem in Vallaris solanacea (Roth) Kuntze (Apocynaceae). J Indian bot Soc 95:202–215
Gondaliya AD, Rajput KS (2016b) Stem anatomy and development of inter- and intraxylary phloem in Leptadenia pyrotechnica (Forssk.) Decne. (Asclepiadaceae). Plant Biosystems 151:855–865. https://doi.org/10.1080/11263504.2016.1218968
Hagel JM, Yeung EC, Facchini PJ (2008) Got milk? The secret life of laticifers. Trends Plant Sci 13:631–639. https://doi.org/10.1016/j.tplants.2008.09.005
Jensen WA (1962) Botanical histochemistry. WH Freeman, San Francisco
Karnovisky MJ (1965) A formaldehyde-glutaraldehyde fixative of high osmolality for use in electron microscopy. J Cell Bio 27:137–138
Krentkowski FL, Duarte MR (2012) Morpho-anatomical analysis of Aspidosperma olivaceum and A. polyneuron. Apocynaceae Braz J Pharmacog 22:937–945
Krepkowski J, Bräuning A, Gebrekirstos A, Strobl S (2011) Cambial growth dynamics and climatic control of different tree life forms in tropical mountain forest in Ethiopia. Trees 25:59–70. https://doi.org/10.1007/s00468-010-0460-7
Kumar SP, Rajeswari B, Rao AP, Reddy LV, Khan PSSV (2012) Anatomical and ultrastructural peculiarities in the laticifers of Euphorbia antiquorum L.: a potential source for the biofuel production. Curr Bot 4:12–17
Lara NOT, Marcati CR (2016) Cambial dormancy lasts 9 months in a tropical evergreen species. Trees 30:1331–1339. https://doi.org/10.1007/s00468-016-1369-6
Leme FM, Borella PH, Marinho CR, Teixeira SP (2020) Expanding the laticifer knowledge in Cannabaceae: distribution, morphology, origin, and latex composition. Protoplasma 257:1183–1199. https://doi.org/10.1007/s00709-020-01500-5
Lopes KLB, Thadeo M, Azevedo AA, Soares AA, Meira RMSA (2009) Articulated laticifers in the vegetative organs of Mandevilla atroviolacea(Apocynaceae, Apocynoideae). Can J Bot 87:202–209. https://doi.org/10.1139/b08-126
Marcati CR, Machado SR, Podadera DS, Podadera DS, Lara NOT, Bosio F, Wiedenhoeft AC (2016) Cambial activity in dry and rainy season on branches from woody species growing in Brazilian Cerrado. Flora 223:1–10. https://doi.org/10.1016/j.flora.2016.04.008
Marinho CR, Teixeira SP (2019) Cellulases and pectinases act together on the development of articulated laticifers in Ficus montana and Maclura tinctoria (Moraceae). Protoplasma 256:1093–1107. https://doi.org/10.1007/s00709-019-01367-1
Mazia D, Brewer PA, Alfert M (1953) The cytochemical staining and measurement of protein with mercuric bromophenol blue. Biol Bull 104:57–67
Medeiros ES, Galiani PD, Moreno RMB, Mattoso L, Malmonge J (2010) A comparative study of the non-isothermal degradation of natural rubber from Mangabeira (Hancornia speciosa Gomes) and Seringueira (Hevea brasiliensis). J Therm Anal Calorim 100:1045–1050. https://doi.org/10.1007/s10973-009-0477-6
Mercadante-Simões MO, Paiva EAS (2013) Leaf colleters in Tontelea micrantha (Celastraceae, Salacioideae): Ecological, morphologicaland structural aspects. C R Biol 336:400–406. https://doi.org/10.1016/j.crvi.2013.06.007
Mercadante-Simões MO, Paiva EAS (2015) Anatomy and ultrastructure of the floral nectary of Tontelea micrantha (Celastraceae: Salacioideae): Floral Nectary of Tontelea micrantha. Plant Species Biol 31:117–124. https://doi.org/10.1111/1442-1984.12093
Milanez FR (1961) Contribuição ao conhecimento anatômico de Cryptostegia grandiflora-II. Sobre os laticíferos da estrutura primária (Asclepiaceae). Rodriguésia 35/36:99–128
Milanez FR (1966) Contribuição ao conhecimento anatômico de Cryptostegia grandiflora – III. Nota sobre a estrutura secundária. Rodriguésia 25:335–350
Naidoo C, Naidoo Y, Dewir YH (2020) The secretory apparatus of Tabernaemontana ventricosa Hochst ex A.DC. (Apocynaceae): laticifer identification characterization and distribution. Plants 9:686. https://doi.org/10.3390/plants9060686
O’Brien TP, Feder N, Mccully ME (1964) Polychromatic staining of plant cell walls by toluidine blue O. Protoplasma 59:368–373. https://doi.org/10.1007/BF01248568
Oliveira JM, Santarosa E, Pillar VD, Roig FA (2009) Seasonal cambium activity in the subtropical rain forest tree Araucaria angustifolia. Trees 23:107–115. https://doi.org/10.1007/s00468-008-0259-y
Pace MR, Cunha Neto IL, Santos-Silva LNN, Melo-de-Pinna FA, Acevedo-Rodríguez P, Almeida RF, Amorim M, Angyalossy V (2019) First report of laticifers in lianas of Malpighiaceae and their phylogenetic implications. Am J Bot 106:1156–1172. https://doi.org/10.1002/ajb2.1350
Pearse AGE (1980) Histhochemistry theoretical and applied: preparative and optical technology. Churchill Livingston, Edinburgh
Pereira AC, Pereira ABD, Moreira CCL, Botion LM, Lemos VS, Braga FC, Cortes SF (2015) Hancornia speciosa Gomes (Apocynaceae) as a potential anti-diabetic drug. J Ethnopharmacol 161:30–35. https://doi.org/10.1016/j.jep.2014.11.050
Pirolla-Souza AO, ArrudaPaceFarinaccio RCMRMA (2019) Leaf anatomical characters of Rhabdadenia (Rhabdadenieae, Apocynaceae), their taxonomic implications, and notes on the presence of articulated laticifers in the genus. Plant Syst Evol 305:797–810. https://doi.org/10.1007/s00606-019-01608-z
Pizzolato TD, Lillie RD (1973) Mayer’s tannic acid-ferric chloride stain for mucins. J Histochem Cytochem 21:56–64
Rajeswari B, Kumar SP, Rao AP, Khan PSSV (2013) A distribution and ultrastructure of laticifers in the phylloclade of Euphorbia caducifolia Haines, a potential hydrocarbon yielding CAM plant. Am J Plant Sci 5:70–79. https://doi.org/10.4236/ajps.2014.51011
Ramos MV, Demarco D, Souza ICC, Freitas CDT (2019) Laticifers, latex, and their role in plant defense. Trends Plant Sci 24:553–567. https://doi.org/10.1016/j.tplants.2019.03.006
Rao KS, Rajput KS, Kim YS (2013) Secondary growth and occurrence of laticifers in the root of papaya (Carica papaya L). Acta Bot Gallica 160:253–258. https://doi.org/10.1080/12538078.2013.830072
Robards AW (1978) An introduction to techniques for scanning electron microscopy of plant cells. In: Hall JL (ed) Electron microscopy and cytochemistry of plant cells. Elsevier, New York, pp 343–403
Roland AM (1978) General preparations and staining of thin sections. In: Hall JL (ed) Electron microscopy and cytochemistry of plant cells. Elsevier, New York, pp 1–62
Sando T, Hayashi T, Takeda T, Akiyama Y, Nakazawa Y, Fukusaki E, Kobayashi A (2009) Histochemical study of detailed laticifer structure and rubber biosynthesis-related protein localization in Hevea brasiliensis using spectral confocal laser scanning microscopy. Planta 230:215–225. https://doi.org/10.1007/s00425-009-0936-0
Souza TR, Pereira IR, Oliveira AHC, Gonçalves PJ, Almeida LM (2016) Caracterização química do látex de Hancornia speciosa e produtividade associada às características fenotípicas. Rev Agrot 7:61–66. https://doi.org/10.12971/2179-5959
Svendsen AB, Verpoorte R (1983) Chromatography of alkaloids. Elsevier, New York
Tan D, Hu X, Fu L, Kumpeangkeaw A, Ding Z, Sun X, Zhang J (2017) Comparative morphology and transcriptome analysis reveals distinct functions of the primary and secondary laticifer cells in the rubber tree. Sci Rep 7:3126. https://doi.org/10.1038/s41598-017-03083-3
Teixeira SP, Marinho CR, Leme FM (2020) Structural diversity and distribution of laticifers In: Advances in Botanical Research, Elsevier, p 27–54. https://doi.org/10.1016/bs.abr.2019.09.003
Vidal BC (1970) Dichroism in collagen bundles stained with Xylidine Ponceau 2R. Ann Histochim 15:289–296
Volland-Voigt F, Bräuning A, Ganzhi O, Peters T, Maza H (2011) Radial stem variations of Tabebuia chrysantha (Bignoniaceae) in different tropical forest ecosystems of southern Ecuador. Trees 25:39–48. https://doi.org/10.1007/s00468-010-0461-6
Acknowledgements
The authors thank the Conselho Nacional de Desenvolvimento Científico e Tecnológico for funding (423340/2018-2) and granting Scientific Initiation Scholarships (PRP02/2018 PIBIC-CNPq/UNIMONTES, PRP03/2018PIBIC-AF-CNPq/UNIMONTES, PRP02/2019 PIBIC-CNPq/UNIMONTES, PRP 03/2019PIBIC-AF-CNPq/UNIMONTES) and Research Productivity Grants to MOMS (423340/2018-2) and LMR (302216/2018-9); the Centro de Microscopia Eletrônica da Universidade Federal de Minas Gerais—CM/UFMG for the ultrastructural analyses; and Waldimar Ferreira Ruas and Cleidimar Pereira Farias Cardoso for their support during fieldwork and laboratory tests.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Communicated by B. Fernandez-Marin.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Souza, A.I.R.d., Cordeiro, K.R., Gonçalves, M.P. et al. The development of anastomosed laticifers in the stem apical meristem and vascular cambium of Hancornia speciosa (Apocynaceae) is related to climatic seasonality. Trees 35, 1317–1328 (2021). https://doi.org/10.1007/s00468-021-02118-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00468-021-02118-7