Abstract
Purpose
There is inconsistent management of cancer-related fatigue (CRF) by health professionals worldwide. This research aims to identify the most appropriate guidelines for the management of cancer-related fatigue.
Methods
A systematic search of international literature identified evidence-based clinical practice guidelines for CRF. Four reviewers independently appraised the highest quality guidelines using the AGREE-II instrument and National Heath and Medical Research Council (NHMRC) guideline standards.
Results
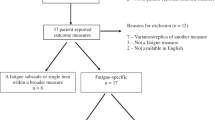
Five guidelines met the inclusion criteria. Of these, the 2015 Canadian Association of Psychosocial Oncology (CAPO) CRF guidelines and the 2014 American Society of Clinical Oncology (ASCO) fatigue guidelines for cancer survivors were selected for in-depth appraisal. The CAPO guideline scored higher than the ASCO for five domains of the AGREE-II. For one domain, the differences were statistically significant (p ≤ 0.05). The CAPO guideline met 37 of 47 NHMRC mandatory guideline standards and the ASCO guideline met 20. The difference in the proportion of standards met was statistically significant for one domain (p ≤ 0.05). Both guidelines had low scores for applicability and implementation.
Conclusions
Currently, the CAPO guideline for cancer-related fatigue has the strongest evidence for use. To enhance implementation, further strategies for guideline dissemination and application are needed.

Similar content being viewed by others
Notes
National Guideline Clearinghouse www.guideline.gov; Clinical Practice Guideline Portal www.clinicalguidelines.gov.au; NICE Guidance www.nice.org.uk/guidance; Guidelines International Network www.g-i-n.net
References
Prue G, Rankin J, Allen J, Gracey J, Cramp F (2006) Cancer-related fatigue: a critical appraisal. Eur J Cancer 42(7):846–863. doi:10.1016/j.ejca.2005.11.026
Wu H-S, Harden JK (2015) Symptom burden and quality of life in survivorship: a review of the literature. Cancer Nurs 38(1):E29–E54. doi:10.1097/NCC.0000000000000135
NCCN (2015) Cancer-related fatigue version 2.2015. http://www.nccn.org/professionals/physician_gls/pdf/fatigue.pdf. Accessed 16/7/2015
Berger AM, Mitchell SA, Jacobsen PB, Pirl WF (2015) Screening, evaluation, and management of cancer-related fatigue: ready for implementation to practice? CA Cancer J Clin. doi:10.3322/caac.21268
Silver JK, Gilchrist LS (2011) Cancer rehabilitation with a focus on evidence-based outpatient physical and occupational therapy interventions. Am J Phys Med Rehabil 90(5 Suppl 1):S5–15. doi:10.1097/PHM.0b013e31820be4ae
Pearson EJM, Morris ME, McKinstry CE (2015) Cancer related fatigue: a survey of health practitioner knowledge and practice. Support Care Cancer 23(12):3521–3529. doi:10.1007/s00520-015-2723-8
Merriam-Webster (2014) Merriam-Webster online dictionary. doi:http://www.merriam-webster.com/
Institutes of Medicine (2011) Clinical practice guidelines we can trust. The National Academies Press, Washington, DC doi:http://nap.edu/catalog/13058.htm
Woolf S, Schünemann HJ, Eccles MP, Grimshaw JM, Shekelle P (2012) Developing clinical practice guidelines: types of evidence and outcomes; values and economics, synthesis, grading, and presentation and deriving recommendations. Implement Sci 7:61. doi:10.1186/1748-5908-7-61
Clubb AB, Dahm P (2011) How to critically appraise a clinical practice guideline. Indian J Urol 27(4):498–502. doi:10.4103/0970-1591.91441
National Health and Medical Research Council (2011) Procedures and requirements for meeting the 2011 NHMRC standard for clinical practice guidelines. National Health and Medical Research Council, Melbourne doi:https://www.nhmrc.gov.au/guidelines/publications/cp133-and-cp133a
Howell D, Keller-Olaman S, Oliver TK, Hack TF, Broadfield L, Biggs K, Chung J, Gravelle D, Green E, Hamel M, Harth T, Johnston P, McLeod D, Swinton N, Syme A, Olson K (2013) A pan-Canadian practice guideline and algorithm: screening, assessment, and supportive care of adults with cancer-related fatigue. Curr Oncol 20(3):e233–e246. doi:10.3747/co.20.1302
Bower JE, Bak K, Berger AM, Breitbart W, Escalante CP, Ganz PA, Schnipper HH, Lacchetti C, Ligibel JA, Lyman GH, Ogaily MS, Pirl WF, Jacobsen PB (2014) Screening, assessment, and management of fatigue in adult survivors of cancer: an American Society of Clinical Oncology clinical practice guideline adaptation. J Clin Oncol 32(17):1840–1850. doi:10.1200/JCO.2013.53.4495
NCCN (2014) Survivorship version 1.2015. National Comprehensive Cancer Network. http://www.nccn.org/professionals/physician_gls/pdf/survivorship.pdf. Accessed 17/7/2015
Mitchell SA, Hoffman AJ, Clark JC, DeGennaro RM, Poirier P, Robinson CB, Weisbrod BL (2014) Putting evidence into practice: an update of evidence-based interventions for cancer-related fatigue during and following treatment. Clin J Oncol Nurs 18(Suppl):38–58. doi:10.1188/14.cjon.s3.38-58
Howell D, Keshavarz H, Broadfield L, Hack T, Hamel M, Harth T, Jones J, McLeod D, Olson K, Phan S, Sawka A, Swinton N, Ali M, on behalf of the Cancer Journey Advisory Group of the Canadian Partnership Against Cancer (2015) A pan Canadian practice guideline for screening, assessment, and management of cancer-related fatigue in adults Version 2-2015. Canadian Association of Psychosocial Oncology, Toronto doi:http://www.capo.ca/pdf/CRF_Guideline.pdf
Brouwers MC, Kho ME, Browman GP, Burgers JS, Cluzeau F, Feder G, Fervers B, Graham ID, Grimshaw J, Hanna SE, Littlejohns P, Makarski J, Zitzelsberger L (2010) AGREE II: advancing guideline development, reporting and evaluation in health care. Can Med Assoc J 182(18):E839–E842. doi:10.1503/cmaj.090449
The ADAPTE Collaboration (2009) The ADAPTE process: resource toolkit for guideline adaptation version 2.0. http://www.g-i-n.net. Accessed 13 Nov 2014
Cronin P, Rawson JV, Heilbrun ME, Lee JM, Kelly AM, Sanelli PC, Bresnahan BW, Paladin AM (2014) How to critically appraise the clinical literature. Acad Radiol 21(9):1117–1128. doi:10.1016/j.acra.2014.05.004
Siering U, Eikermann M, Hausner E, Hoffmann-Esser W, Neugebauer EA (2013) Appraisal tools for clinical practice guidelines: a systematic review. PLoS One 8(12):e82915. doi:10.1371/journal.pone.0082915
The AGREE Research Trust (2013) Appraisal of guidelines for REsearch and evaluation II. AGREE II Instrument. www.agreetrust.org. Accessed 6 August 2015
Pallant JF (2013) SPSS survival manual - a step by step guide to data analysis using IBM SPSS, 5 edn. Allen & Unwin, Crows Nest
Hallgren KA (2012) Computing inter-rater reliability for observational data: an overview and tutorial. Tutor Quant Methods Psychol 8(1):23–34 doi:nihms37295.pdf
Light RJ (1971) Measures of response agreement for qualitative data: some generalizations and alternatives. Psychol Bulletin 76(5):365–377. doi:10.1037/h0031643
Liddell FDK (1983) Simplified exact analysis of case-referent studies: matched pairs; dichotomous exposure. J Epidemiol Community Health 37(1):82–84. doi:10.1136/jech.37.1.82
Mitchell SA, Friese CR (2012) ONS PEP (Putting Evidence into Practice) weight of evidence classification schema: decision rules for summative evaluation of a body of evidence. https://www.ons.org/practice-resources/pep/evaluation-process. Accessed July 3 2015
Guyatt GH, Oxman AD, Vist GE, Kunz R, Falck-Ytter Y, Alonso-Coello P, Schünemann HJ (2008) GRADE: an emerging consensus on rating quality of evidence and strength of recommendations. BMJ 336(7650):924–926. doi:10.1136/bmj.39489.470347.AD
Jacobs C, Graham ID, Makarski J, Chasse M, Fergusson D, Hutton B, Clemons M (2014) Clinical practice guidelines and consensus statements in oncology - an assessment of their methodological quality. PLoS One 9(10). doi:10.1371/journal.pone.0110469
Harris SR, Schmitz KH, Campbell KL, McNeely ML (2012) Clinical practice guidelines for breast cancer rehabilitation. Cancer 118(S8):2312–2324. doi:10.1002/cncr.27461
Landis JR, Koch GG (1977) An application of hierarchical kappa-type statistics in the assessment of majority agreement among multiple observers. Biometrics 33(2):363–374. doi:10.2307/2529786
Cicchetti DV (1994) Guidelines, criteria, and rules of thumb for evaluating normed and standardized assessment instruments in psychology. Psychol Assess 6(4):284–290. doi:10.1037/1040-3590.6.4.284
Cohen J (1988) Statistical power analysis for the behavioral sciences, 2 edn. Academic Press Inc, Hillsdale, NJ
Nunnally JC (1994) Psychometric theory. McGraw-Hill series in psychology, 3rd edn. McGraw-Hill, New York
Eisinga R, Grotenhuis M, Pelzer B (2013) The reliability of a two-item scale: Pearson, Cronbach, or Spearman-Brown? Int J Public Health 58(4):637–642. doi:10.1007/s00038-012-0416-3
Reames BN, Krell RW, Ponto SN, Wong SL (2013) Critical evaluation of oncology clinical practice guidelines. J Clin Oncol 31(20):2563–2568. doi:10.1200/JCO.2012.46.8371
Kastner M, Estey E, Hayden L, Chatterjee A, Grudniewicz A, Graham ID, Bhattacharyya O (2014) The development of a guideline implementability tool (GUIDE-IT): a qualitative study of family physician perspectives. BMC Fam Pract 15:19. doi:10.1186/1471-2296-15-19
Shiffman RN, Dixon J, Brandt C, Essaihi A, Hsiao A, Michel G, O’Connell R (2005) The GuideLine implementability appraisal (GLIA): development of an instrument to identify obstacles to guideline implementation. BMC Med Inform Decis Mak 5:23. doi:10.1186/1472-6947-5-23
National Institute of Clinical Studies (2006) Assessing the implementability of guidelines. NICS, Melbourne doi:http://www.nhmrc.gov.au/
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Electronic supplementary material
ESM 1
(DOCX 168 kb)
Rights and permissions
About this article
Cite this article
Pearson, E.J.M., Morris, M.E. & McKinstry, C.E. Cancer-related fatigue: appraising evidence-based guidelines for screening, assessment and management. Support Care Cancer 24, 3935–3942 (2016). https://doi.org/10.1007/s00520-016-3228-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00520-016-3228-9