Abstract
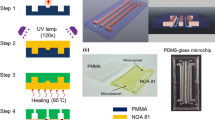
Toner-based fabrication technology has appeared as one of the simplest and fastest techniques to produce low-cost microfluidic devices. The instrumental simplicity and fabrication easiness make this fabrication technique quite suitable to be implemented in developing countries where the availability of resources is limited. In this study, the prototyping fidelity of poly(dimethylsiloxane) (PDMS) electrophoresis microchips has been evaluated using laser printed masters. The desirable layout of the microfluidic devices was first drawn in graphic software and then printed on the surface of transparency films by an office laser printer with 1,200-dpi resolution. The masters printed on transparency films were directly used to replicate the microfluidic structures in PDMS. The analysis of the dimensions of the printed toner lines has revealed that their thickness depends on the line width. For lines with width between 50 and 300 μm, the height ranged from 4.9 ± 0.2 to 10.4 ± 0.1 μm. The prototyping fidelity achieved for channels defined with width between 50 and 300 μm was higher than 96 %. Furthermore, it has been demonstrated that a single master can be used to replicate at least ten PDMS chips with great reproducibility. The analytical performance of the replicated PDMS microchips for electrophoresis separation has revealed good chip-to-chip and injection-to-injection reproducibility. Based on the results achieved, it is possible to conclude that toner-based masters can be a useful tool for researchers with restricted access to conventional microfabrication technologies.





Similar content being viewed by others
References
Abdelgawad M, Watson MW, Young EW, Mudrik JM, Ungrin MD, Wheeler AR (2008) Soft lithography: masters on demand. Lab Chip 8(8):1379–1385
Bao N, Zhang Q, Xu JJ, Chen HY (2005) Fabrication of poly(dimethylsiloxane) microfluidic system based on masters directly printed with an office laser printer. J Chromatogr A 1–2:270–275
Chen CS, Breslauer DN, Luna JI, Grimes A, Chin WC, Leeb LP, Khine M (2008) Shrinky-dink microfluidics: 3D polystyrene chips. Lab Chip 8(4):622–624
Coltro WKT, Piccin E, da Silva JAF, do Lago CL, Carrilho E (2007) A toner-mediated lithographic technology for rapid prototyping of glass microchannels. Lab Chip 7(7):931–934
Coltro WKT, de Jesus DP, da Silva JAF, do Lago CL, Carrilho E (2010) Toner and paper-based fabrication techniques for microfluidic applications. Electrophoresis 31(15):2487–2498
Coltro WKT, da Silva JAF, Carrilho E (2011) Rapid prototyping of polymeric electrophoresis microchips with integrated copper electrodes for contactless conductivity detection. Anal Methods UK 3(1):168–172
da Silva JAF, Guzman N, do Lago CL (2002) Contactless conductivity detection for capillary electrophoresis––hardware improvements and optimization of the input-signal amplitude and frequency. J Chromatogr A 942(1–2):249–258
da Silva ER, Segato TP, Coltro WKT, Lima RS, Carrilho E, Mazo LH (2013) Determination of glyphosate and AMPA on polyester-toner electrophoresis microchip with contactless conductivity detection. Electrophoresis 34(14):2107–2111
de Jesus DP, Blanes L, do Lago CL (2006) Microchip free-flow electrophoresis on glass substrate using laser-printing toner as structural material. Electrophoresis 27(24):4935–4942
de Souza FR, Alves GL, Coltro WKT (2012) Capillary-driven toner-based microfluidic devices for clinical diagnostics with colorimetric detection. Anal Chem 84(21):9002–9007
do Lago CL, da Silva HDT, Neves CA, Brito-Neto JGA, da Silva JAF (2003) A dry process for production of microfluidic devices based on the lamination of laser-printed polyester films. Anal Chem 75(15):3853–3858
do Lago CL, Neves CA, de Jesus DP, da Silva HDT, Brito-Neto JGA, da Silva JAF (2004) Microfluidic devices obtained by thermal toner transferring on glass substrate. Electrophoresis 25(21–22):3825–3831
Duarte GRM, Price CW, Augustine BH, Carrilho E, Landers JP (2011) Dynamic solid phase DNA extraction and PCR amplification in polyester-toner based microchip. Anal Chem 83(13):5182–5189
Duffy DC, McDonald JC, Schueller OJA, Whitesides GM (1998) Rapid prototyping of microfluidic systems in poly(dimethylsiloxane). Anal Chem 70(23):4974–4984
Easley CJ, Benninger RKP, Shaver JH, Head WS, Piston DW (2009) Rapid and inexpensive fabrication of polymeric microfluidic devices via toner transfer masking. Lab Chip 9(8):1119–1127
Gabriel EFM, do Lago CL, Gobbi ÂL, Carrilho E, Coltro WKT (2013) Characterization of microchip electrophoresis devices fabricated by direct-printing process with colored toner. Electrophoresis 34(15):2169–2176
Grimes A, Breslauer DN, Long M, Pegan J, Lee LP, Khine M (2008) Shrinky-dink microfluidics: rapid generation of deep and rounded patterns. Lab Chip 8(1):170–172
Hong C, Bao D, Thomas MS, Clift JM, Vullev VI (2008) Print-and-peel fabrication of microelectrodes. Langmuir 24(16):8439–8442
Jena RK, Yue CY, Lam YC, Tang PS, Gupta A (2012) Comparison of different molds (epoxy, polymer and silicon) for microfabrication by hot embossing technique. Sens Actuator B Chem 163(1):233–241
Kaigala GV, Ho S, Penterman R, Backhouse CJ (2007) Rapid prototyping of microfluidic devices with a wax printer. Lab Chip 7(3):384–387
Kuban P, Hauser PC (2013) Contactless conductivity detection for analytical techniques: developments from 2010 to 2012. Electrophoresis 34(1):55–69
Liu AL, He FY, Wang K, Zhou T, Lu Y, Xia XH (2005) Rapid method for design and fabrication of passive micromixers in microfluidic devices using a direct-printing process. Lab Chip 5(9):974–978
Lu Y, Hu YL, Xia XH (2009) Effect of surface microstructures on the separation efficiency of neurotransmitters on a direct-printed capillary electrophoresis microchip. Talanta 79(5):1270–1275
McDonald JC, Duffy DC, Anderson JR, Chiu DT, Wu HK, Schueller OJA, Whitesides GM (2000) Fabrication of microfluidic systems in poly(dimethylsiloxane). Electrophoresis 21(1):27–40
McDonald JC, Chabinyc ML, Metallo SJ, Anderson JR, Stroock AD, Whitesides GM (2002) Prototyping of microfluidic devices in poly(dimethylsiloxane) using solid-object printing. Anal Chem 74(7):1537–1545
Ouyang Y, Wang S, Li J, Riehl PS, Begley M, Landers JP (2013) Rapid patterning of ‘tunable’ hydrophobic valves on disposable microchips by laser printer lithography. Lab Chip 13(9):1762–1771
Tan AM, Rodgers K, Murrihy JP, O’Mathuna C, Glennon JD (2001) Rapid fabrication of microfluidic devices in poly(dimethylsiloxane) by photocopying. Lab Chip 1(1):7–9
Thomas M, Millare B, Clift JM, Vullev VI (2009) Print-and-peel fabrication for microfluidics. Abstr Papers Am Chem Soc 237:263
Thomas MS, Millare B, Clift JM, Bao D, Hong C, Vullev VI (2010) Print-and-peel fabrication for microfluidics: what’s in it for biomedical applications? Ann Biomed Eng 38(1):21–32
Vullev VI, Wan JD, Heinrich V, Landsman P, Bower PE, Xia B, Millare B, Jones G (2006) Nonlithographic fabrication of microfluidic devices. J Am Chem Soc 128(50):16062–16072
Yu H, He FY, Lu Y, Hu YL, Zhong HY, Xia XH (2008a) Improved separation efficiency of neurotransmitters on a native printed capillary electrophoresis microchip simply by manipulating electroosmotic flow. Talanta 75(1):43–48
Yu H, Lu Y, Zhou YG, Wang FB, He FY, Xia XH (2008b) A simple, disposable microfluidic device for rapid protein concentration and purification via direct-printing. Lab Chip 8(9):1496–1501
Acknowledgments
This project has been supported by Conselho Nacional de Desenvolvimento Científico e Tecnólogico (CNPq)—Grant 478911/2012-2––and Fundação de Amparo à Pesquisa do Estado de Goiás (FAPEG). The authors gratefully acknowledge the research fellowships granted from CNPq to D. P. J. (Grant No. 305318/2012-8) and W. K. T. C. (Grant No. 311744/2013-3) and scholarships granted from CNPq to E. O. L. and L. C. D. and from CAPES to L. E. P. B. We would like to thank the Laboratory of Microfabrication from Brazilian Nanotechnology Laboratory for using their facilities.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Lobo Júnior, E.O., Duarte, L.C., Paula Braga, L.E. et al. High fidelity prototyping of PDMS electrophoresis microchips using laser-printed masters. Microsyst Technol 21, 1345–1352 (2015). https://doi.org/10.1007/s00542-014-2190-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00542-014-2190-z