Abstract
Study design
A multi-cohort, case–control rodent study.
Purpose
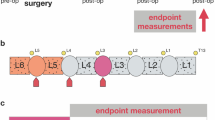
Investigate the long-term behavioural, histologic and radiologic consequences on the complete lumbar spine of L4/5 intervertebral disc (IVD) injury in mice and determine if increased physical activity mitigates the observed changes.
Methods
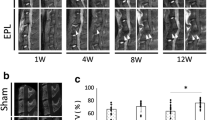
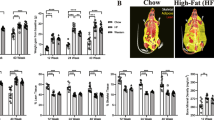
Cohorts of 2-month-old CD1 female mice underwent a single ventral puncture of the L4/5 IVD. 0.5-, 3- or 12-months after injury, general health (body weight and locomotor capacity), behavioural signs of axial discomfort (tail suspension, grip strength and FlexMaze assays) and radiating pain (von Frey and acetone tests) were assessed. Experimental groups with free access to an activity wheel in their home cages were including in the 12-month cohort. Lumbar disc status was determined using colorimetric staining and radiologic (X-ray and T2-MRI) analysis. Innervation was measured by immunoreactivity for PGP9.5 and calcitonin gene‐related peptide.
Results
No changes in general health or persistent signs of axial discomfort were observed up to one year post-injury. In contrast, signs of radiating pain developed in injured mice at 3 months post-injury, persisted up to 12 months and were reversed by long-term physical activity. At 12-months post-injury, degeneration was observed in non-injured lumbar discs. Secondary degenerating IVDs were similar to the injured discs by X-ray (narrowing) and T2-MRI (internal disc disruption) but did not show abnormal innervation. Increased physical activity had no impact on mechanically injured IVDs, but attenuated disc narrowing at other lumbar levels.
Conclusions
Mechanical injury of L4/5-IVDs induces delayed radiating pain and degeneration of adjacent discs; increased physical activity positively mitigated both.



Similar content being viewed by others
Availability of data and material
Available upon request.
References
Global Burden Of Disease Study C (2015) Global, regional, and national incidence, prevalence, and years lived with disability for 301 acute and chronic diseases and injuries in 188 countries, 1990–2013: a systematic analysis for the global burden of disease study 2013. Lancet 386:743–800. https://doi.org/10.1016/S0140-6736(15)60692-4
Ja H, Mw VT, Av M, Bw K (2005) Meta-analysis: exercise therapy for nonspecific low back Pain. Ann Intern Med 142:765–775. https://doi.org/10.7326/0003-4819-142-9-200505030-00013
Millecamps M, Ls S (2018) Delayed onset of persistent discogenic axial and radiating pain after a single-level lumbar intervertebral disc injury in mice. Pain 159:1843–1855. https://doi.org/10.1097/J.Pain.0000000000001284
Lee S, Millecamps M, Dz F, Ls S (2020) Long-term histological analysis of innervation and macrophage infiltration in a mouse model of intervertebral disc injury-induced low back pain. J Orthop Res 38:1238–1247. https://doi.org/10.1002/Jor.24560
Zimmermann M (1986) Ethical considerations in relation to pain in animal experimentation. Acta Physiol Scand Suppl 554:221–233
Millecamps M, Tajerian M, Naso L, Sage Eh, Ls S (2012) Lumbar intervertebral disc degeneration associated with axial and radiating low back pain in ageing sparc-null mice. Pain 153:1167–1179. https://doi.org/10.1016/J.Pain.2012.01.027
Millecamps M, Jt C, Ap M, Ls S (2015) Behavioral signs of axial low back pain and motor impairment correlate with the severity of intervertebral disc degeneration in a mouse model. Spine J 15:2524–2537. https://doi.org/10.1016/J.Spinee.2015.08.055
Miyagi M, Millecamps M, Danco At, Ohtori S, Takahashi K, Stone Ls (2014) Issls prize winner: increased innervation and sensory nervous system plasticity in a mouse model of low back pain due to intervertebral disc degeneration. Spine (Phila Pa 1976) 39:1345–1354. https://doi.org/10.1097/Brs.0000000000000334
Tajerian M, Millecamps M, Ls S (2012) Morphine and clonidine synergize to ameliorate low back pain in mice. Pain Res Treat 2012:150842. https://doi.org/10.1155/2012/150842
Shi C, Qiu S, Sm R, Das V, Zhu B, Aa W, Aj VW, Mwale F, Jc I, Sakai D, Votta-Velis G, Yuan W, Hj Im (2018) Animal models for studying the etiology and treatment of low back pain. J Orthop Res 36:1305–1312. https://doi.org/10.1002/Jor.23741
Olmarker K (2008) Puncture of a lumbar intervertebral disc induces changes in spontaneous pain behavior: an experimental study in rats. Spine (Phila Pa 1976) 33:850–855. https://doi.org/10.1097/Brs.0b013e31816b46ca
Allen Kd, Tm G, Rm R, Wc W, Vb K, Jl H, Lm B, La S (2009) Decreased physical function and increased pain sensitivity in mice deficient for type Ix collagen. Arthritis Rheum 60:2684–2693. https://doi.org/10.1002/Art.24783
Shi C, Das V, Li X, Kc R, Qiu S, Os I, Rl R, Js K, Mwale F, Aa W, Zhu B, Zhao L, Aj VW, Ji M, Lu J, Votta-Velis G, Yuan W, Hj Im (2018) Development of an in vivo mouse model of discogenic low back pain. J Cell Physiol 233:6589–6602. https://doi.org/10.1002/Jcp.26280
Millecamps M, Tajerian M, Sage Eh, Stone Ls (2011) Behavioral signs of chronic back pain in the sparc-null mouse. Spine (Phila Pa 1976) 36:95–102. https://doi.org/10.1097/Brs.0b013e3181cd9d75
Brinjikji W, Fe D, Jg J, Cm C, Df K, Murad Mh, Luetmer Ph (2015) Mri findings of disc degeneration are more prevalent in adults with low back pain than in asymptomatic controls: a systematic review and meta-analysis. Ajnr Am J Neuroradiol 36:2394–2399. https://doi.org/10.3174/Ajnr.A4498
Konstantinou K, Dunn Km, Ogollah R, Vogel S, Hay Em, Team Asr (2015) Characteristics of patients with low back and leg pain seeking treatment in primary care: baseline results from the Atlas cohort study. BMC Musculoskelet Disord 16:332. https://doi.org/10.1186/S12891-015-0787-8
Hawasli Ah, Chang J, Ck Y, Steger-May K, Lg L, Ig D (2016) Interpedicular height as a predictor of radicular pain in adult degenerative scoliosis. Spine J 16:1070–1078. https://doi.org/10.1016/J.Spinee.2016.04.017
Lai A, Ho L, Tw E-R, Watanabe H, Salandra J, Ba W, Laudier D, Ac H, Gm P, Jc I (2019) Dietary polyphenols as a safe and novel intervention for modulating pain associated with intervertebral disc degeneration in an in-vivo rat model. PLoS ONE 14:E0223435. https://doi.org/10.1371/Journal.Pone.0223435
Aa G (2016) Adjacent segment degeneration after posterior lumbar fusion: an analysis of possible risk factors. Clin Neurol Neurosurg 143:15–18. https://doi.org/10.1016/J.Clineuro.2016.02.004
Fm S-P, Ra D, Ec B (2014) Adjacent segment disease perspective and review of the literature. Ochsner J 14:78–83
As H, Robbins M (2004) Adjacent segment degeneration and adjacent segment disease: the consequences of spinal fusion? Spine J 4:190s–194s. https://doi.org/10.1016/J.Spinee.2004.07.007
Dc N, Marcia S, Ardura F, Is L, Marras M, Saba L (2016) Diffusion-weighted Mri assessment of adjacent disc degeneration after thoracolumbar vertebral fractures. Cardiovasc Intervent Radiol 39:1306–1314. https://doi.org/10.1007/S00270-016-1369-3
Hoogendoorn Rj, Helder Mn, Wuisman Pi, Bank Ra, Everts Ve, Smit Th (2008) Adjacent segment degeneration: observations in a goat spinal fusion study. Spine (Phila Pa 1976) 33:1337–1343. https://doi.org/10.1097/Brs.0b013e318173438f
Cc L, Fm T, Zhou Z, Wang P, Gou Y, Zhang H, Wy W, Shen Y, Yz Z, Zhang L (2015) Protective effect of calcitonin on lumbar fusion-induced adjacent-segment disc degeneration in ovariectomized rat. BMC Musculoskelet Disord 16:342. https://doi.org/10.1186/S12891-015-0788-7
Acknowledgements
The authors thank the staff of McGill University’s Comparative Medicine and Animal Resources Centre, Alan Edwards Centre for Research on Pain, Douglas Brain Imaging Centre, and Centre for Bone and Periodontal Research for their support and expertise.
Funding
This work was supported by Canadian Institutes for Health Research Grants MOP-126046 and MOP-142291 to LSS and MM. SL was supported by the Catherine Bushnell postdoctoral fellowship from the Louise and Alan Edwards Foundation.
Author information
Authors and Affiliations
Contributions
Data collection MM, PL, DF; Methodology, Material preparation, Project administration, Formal analysis: MM; Funding acquisition and Conceptualization by MM and LSS, Supervision and manuscript review & editing: LSS. All authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no competing interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Millecamps, M., Lee, S., Foster, D.Z. et al. Disc degeneration spreads: long-term behavioural, histologic and radiologic consequences of a single-level disc injury in active and sedentary mice. Eur Spine J 30, 2238–2246 (2021). https://doi.org/10.1007/s00586-021-06893-2
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00586-021-06893-2