Abstract
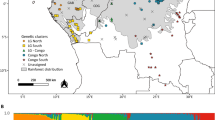
Colonising populations do not always exhibit founder effects. Common explanations are high levels of immigration and/or reproduction, but few empirical tests have been done. We measured genetic diversity of Nassauvia argentea in terms of variation and divergence of plant populations that have colonised Volcán Lonquimay, Chile, following its latest eruption in 1988. Fifteen individuals from each of ten populations were analysed using amplified fragment length polymorphism (AFLP) markers. Genetic variation and divergence were lower in colonising populations than established ones, but not significantly so (ANOVA and Kruskal-Wallis tests, p < 0.05). No consistent or significant trends were obtained from regressions with demographic variables. Bayesian analysis of population structure reveals close relatedness among populations of all ages on the volcano. We concluded that no conspicuous founder effect has occurred in the genetic diversity of populations colonising a newly derived volcanic environment. An important implication of this is the role of proximity to source regions and species vigour in moulding genetic diversity of colonisers from different species.



Similar content being viewed by others
References
Arrigo N, Tuszynski JW, Ehrich D, Gerdes T, Alvarez N (2009) Evaluating the impact of scoring parameters on the structure of intra-specific genetic variation using RawGeno, an R package for automating AFLP scoring. BMC Bioinformatics 10:33
Arroyo MTK, Squeo F (1990) Relationship between plant breeding systems and pollination. In: Kawano S (ed) Biological approaches and evolutionary trends in plants. Academic Press, London, pp 205–227
Arroyo MTK, Primack R, Armesto J (1982) Community studies in pollination ecology in the high temperate Andes of central Chile. I. Pollination mechanisms and altitudinal variation. Am J Bot 69:82–97
Barton NH, Charlesworth B (1984) Genetic revolutions, founder effects and speciation. Annual Rev Ecol Syst 15:133–164
Barton NH, Mallet J (1996) Natural selection and random genetic drift as causes of evolution on islands [and discussion]. Philos Trans Ser B 351:785–795
Benham J (2001) Genographer. Version 1.6.0. Montana State University, Montana
Bonin A, Ehrich D, Manel S (2007) Statistical analysis of amplified fragment length polymorphism data: a toolbox for molecular ecologists and evolutionists. Mol Ecol 16:3737–3758
Bryant D, Moulton V (2004) Neighbor-Net: an agglomerative method for the construction of phylogenetic networks. Mol Biol Evol 21:255–265
Cabrera AL (1982) Revisión del género Nassauvia (Compositae). Darwiniana 24:283–379
Carson HL (1968) The population flush and its genetic consequences. In: Lewontin RC (ed) Population biology and ecology. Syracuse University Press, New York, pp 123–137
Carson HL, Wisotzkey RG (1989) Increase in genetic variance following a population bottleneck. Amer Naturalist 134:668–673
Castor C (2002) Patrones, procesos y mecanismos de dispersión secundaria en plantas andinas de Chile central. PhD thesis, Universidad de Chile, Chile
Chakraborty R, Nei M (1977) Bottleneck effects on average heterozygosity and genetic distance with the stepwise mutation model. Evolution 31:347–356
Charlesworth D (2003) Effects of inbreeding on the genetic diversity of populations. Philos Trans Ser B 358:1051–1070
Comes HP, Tribsch A, Bittkau C (2008) Plant speciation in continental island floras as exemplified by Nigella in the Aegean Archipelago. Philos Trans Ser B 363:3083–3096
Corander J, Marttinen P (2006) Bayesian identification of admixture events using multilocus molecular markers. Mol Ecol 15:2833–2843
Corander J, Waldmann P, Sillanpää MJ (2003) Bayesian analysis of genetic differentiation between populations. Genetics 163:367–374
Coyne JA, Orr HA (2004) Speciation. Sinauer Associates, Massachussetts
Curtin CD, Bellon JR, Henschke PA (2007) Genetic diversity of Dekkera bruxellensis yeasts isolated from Australian wineries. FEMS Yeast Res 7:471–481
del Moral R, Clampitt CA (1985) Growth of native plant species on recent volcanic substrates from Mount St. Helens. Amer Midl Naturalist 114:374–383
del Moral R, Lacher IL (2005) Vegetation patterns 25 years after the eruption of Mount St. Helens, Washington, USA. Amer J Bot 92:1948–1956
Deshpande O, Batzoglou S, Feldman M, Cavalli-Sforza L (2009) A serial founder effect model for human settlement out of Africa. Proc Roy Soc Biol Sci Ser B 276:291–300
Després L, Loriot S, Gaudeul M (2002) Geographic pattern of genetic variation in the European globeflower Trollius europaeus L. (Ranunculaceae) inferred from amplified fragment length polymorphism markers. Mol Ecol 11:2337–2347
Doyle JJ, Doyle JL (1987) A rapid DNA isolation procedure for small quantities of fresh leaf tissue. Phytochem Bull 19:11–15
Ehrich D (2006) AFLPdat: a collection of R functions for convenient handling of AFLP data. Mol Ecol Notes 6:603–604
Excoffier L, Laval G, Schneider S (2006) Arlequin Version 3.1 User Manual. Computational and Molecular Population Genetics Lab (CMPG), Institute of Zoology, University of Berne
Fridriksson S, Magnusson B (1992) Development of the ecosystem on Surtsey with references to Anak Krakatau. GeoJournal 28:287–291
González-Ferrán O (1995) Volcanes de Chile. Instituto Geográfico Militar, Santiago
Gross JP (2006) Nortest: tests for normality. R package version 1.0
Hamrick JL, Nason JD (1996) Consequences of dispersal in plants. In: Rhodes OE Jr, Chester RK, Smith MH (eds) Population dynamics in ecological space and time. University of Chicago Press, Chicago, pp 203–236
Holland BS, Clarke AC, Meudt HM (2008) Optimizing automated AFLP scoring parameters to improve phylogenetic resolution. Syst Biol 57:347–366
Holsinger KE, Wallace LE (2004) Bayesian approaches for the analysis of population genetic structure: an example from Platanthera leucophaea (Orchidaceae). Mol Ecol 13:887–894
Holsinger KE, Lewis PO, Dey DK (2002) A Bayesian approach to inferring population structure from dominant markers. Mol Ecol 11:1157–1164
Honnay O, Jacquemyn H, Van Looy K, Vandepitte K, Breyne P (2009) Temporal and spatial genetic variation in a metapopulation of the annual Erysimum cheiranthoides on stony river banks. J Ecol 97:131–141
Huson DH, Bryant D (2006) Application of phylogenetic networks in evolutionary studies. Mol Biol Evol 23:254–267
Hutchison DW, Templeton AR (1999) Correlation of pairwise genetic and geographic distance measures: inferring the relative influences of gene flow and drift on the distribution of genetic variability. Evolution 53:1898–1914
Ingvarsson PK (2001) Restoration of genetic variation lost—the genetic rescue hypothesis. Trends Ecol Evol 16:62–63
Jacquemyn H, Vandepitte K, Roldan-Ruiz I, Honnay O (2008) Rapid loss of genetic variation in a founding population of Primula elatior (Primulaceae) after colonization. Ann Bot 103:777–783
Keller LF, Jeffery KJ, Arcese P, Beaumont M (2001) Immigration and the ephemerality of a natural population bottleneck: evidence from molecular markers. Proc Roy Soc Biol Sci Ser B 268:1387–1394
Leberg PL (1992) Effects of population bottlenecks on genetic diversity as measured by allozyme electrophoresis. Evolution 46:477–494
López PG, Tremetsberger K, Stuessy TF, Gómez-Gónzalez S, Jiménez A, Baeza CM (2010) Patterns of genetic variation in colonizing plant species: Nassauvia lagascae (Asteraceae: Mutiseae) on Volcán Lonquimay, Chile. Amer J Bot 97:423–432
Maruyama T, Fuerst PA (1985) Population bottlenecks and nonequilibrium models in population genetics. II. Number of alleles in a small population that was formed by a recent bottleneck. Genetics 111:675–689
Mayr E (1942) Systematics and the origin of species. Columbia University Press, New York
McCauley DE, Raveill J, Antonovics J (1995) Local founding events as determinants of genetic structure in a plant metapopulation. Heredity 75:630–636
Merilä J, Björklund M, Baker A (1996) The successful founder: genetics of introduced Carduelis chloris (greenfinch) populations in New Zealand. Heredity 77:410–422
Miller NJ, Ciosi M, Sappington TW (2007) Genome scan of Diabrotica virgifera virgifera for genetic variation associated with crop rotation tolerance. J Appl Entomol 131:378–385
Mueller UG, Wolfenbarger LL (1999) AFLP genotyping and fingerprinting. Trends Ecol Evol 14:389–394
Nei M, Maruyama T, Chakraborty R (1975) The bottleneck effect and genetic variability in populations. Evolution 29:1–10
Pannell JR, Dorken ME (2006) Colonisation as a common denominator in plant metapopulations and range expansions: effects on genetic diversity and sexual systems. Landscape Ecol 21:837–848
Parrish TL (2002) Krakatau: genetic consequences of island colonization. PhD thesis, University of Utrecht, Netherlands
Philippi RA (1894) Plantas nuevas Chilenas. Anales Univ Chile 87:82
Polans NO, Allard RW (1989) An experimental evaluation of the recovery potential of ryegrass populations from genetic stress resulting from restriction of population size. Evolution 43:1320–1324
Provine WB (1989) Founder effects and genetic revolutions in microevolution and speciation: an historical perspective. In: Giddings LV, Kaneshiro KY, Anderson WW (eds) Genetics, speciation and the founder principle. Oxford University Press, Oxford, pp 43–76
R Development Core Team (2008) R: a language and environment for statistical computing. R Foundation for Statistical Computing, Vienna
Robichaux RH, Friar EA, Mount DW (1997) Molecular genetic consequences of a population bottleneck associated with reintroduction of the Mauna Kea silversword. Conservation Biol 11:1140–1146
Schlüter PM, Harris S (2006) Analysis of multilocus fingerprinting datasets containing missing data. Mol Ecol Notes 6:569–572
Slatkin M (1977) Gene flow and genetic drift in a species subject to frequent local extinctions. Theor Populat Biol 12:253–262
Sokal RR, Michener CD (1958) A statistical method for evaluating systematic relationships. Kansas Univ Sci Bull 38:1409–1438
Spiegelhalter DJ, Best NG, Carlin BP, van der Linde A (2002) Bayesian measures of model complexity and fit. J R Statist Soc B 64:583–639
Sun M (1996) Effects of population size, mating system, and evolutionary origin on genetic diversity in Spiranthes sinensis and S. hongkongensis. Conservation Biol 10:785–795
Swofford DL (2002) PAUP*: Phylogenetic Analyses Using Parsimony (*and other methods) 4.0 beta version. Sinauer Associates, Massachussetts
Templeton AR (1980) The theory of speciation via the founder principle. Genetics 94:1011–1038
Thornton IWB (1996) Krakatau: the destruction and reassembly of an island ecosystem. Harvard University Press, Massachussetts
Thornton IWB (2001) Colonization of an island volcano, Long Island, Papua New Guinea, and an emergent island, Motmot, in its Caldera Lake. I. General Introduction. J Biogeogr 28:1299–1310
Travis SE, Proffitt CE, Lowenfeld RC, Mitchell TW (2002) A comparative assessment of genetic diversity among differently-aged populations of Spartina alterniflora on restored versus natural wetlands. Restorat Ecol 10:37–42
Tremetsberger K, Stuessy TF, Samuel RM, Baeza CM, Fay M (2003) Genetics of colonization in Hypochaeris tenuifolia (Asteraceae, Lactuceae) on Volcán Lonquimay, Chile. Mol Ecol 12:2649–2659
Vos P, Hogers R, Bleeker M, Reijans M, van der Lee T, Hornes M, Frijters A, Pot J, Peleman J, Kuiper M, Zabeau M (1995) AFLP: a new technique for DNA fingerprinting. Nucl Acids Res 23:4407–4414
Wade MC, McCauley DE (1988) Extinction and recolonization: their effects on the genetic differentiation of local populations. Evolution 42:995–1005
Whittaker RJ, Fernández-Palacios JM (2008) Island biogeography: ecology, evolution and conservation, 2nd edn. Oxford University Press, Oxford
Wooten JA, Tolley-Jordan LR (2009) Validation of phylogenetic signals in amplified fragment length data: testing the utility and reliability in closely related taxa. BMC Res Notes 2:26
Yang S, Bishop JL, Webster MS (2008) Colonization genetics of an animal-dispersed plant (Vaccinium membranaceum) at Mount St. Helens, Washington. Mol Ecol 17:731–740
Acknowledgments
The authors thank: the Departamento de Botánica, Universidad de Concepción, for work, space and consultation of herbarium material (CONC) in Chile; the Corporación Nacional Forestal (CONAF) for permission to collect samples from Chilean National Parks; R. Hössinger and M. J. Parra for help with collecting; G. Kadlec for technical assistance; Dr. David Hulce at SoftGenetics for guidance on the appropriate use of GeneMarker for scoring of AFLPs and an anonymous reviewer for helpful comments on the manuscript. This project was funded by a grant from Fonds zur Förderung der Wissenschaftlichen Forschung (FWF grant P18446 to TFS). EG was supported by a UK Natural Environment Research Council Masters studentship via the University of York Biology Department.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Green, E.C., Tremetsberger, K., Jiménez, A. et al. Genetic diversity of pioneer populations: the case of Nassauvia argentea (Asteraceae: Mutisieae) on Volcán Lonquimay, Chile. Plant Syst Evol 298, 109–119 (2012). https://doi.org/10.1007/s00606-011-0527-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00606-011-0527-y