Abstract
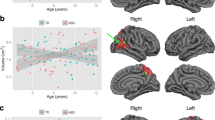
Autism spectrum disorder (ASD) is a lifelong neurodevelopmental condition that is accompanied by an atypical development of brain maturation. So far, brain development has mainly been studied during early childhood in ASD, and using measures of total or lobular brain volume. However, cortical volumetric measures are a product of two distinct biological neuroanatomical features, cortical thickness, and surface area, which most likely also have different neurodevelopmental trajectories in ASD. Here, we therefore examined age-related differences in cortical thickness and surface area in a cross-sectional sample of 77 male individuals with ASD ranging from 7 to 25 years of age, and 77 male neurotypical controls matched for age and FSIQ. Surface-based measures were analyzed using a general linear model (GLM) including linear, quadratic, and cubic age terms, as well as their interactions with the main effect of group. When controlling for the effects of age, individuals with ASD had spatially distributed reductions in cortical thickness relative to controls, particularly in fronto-temporal regions, and also showed significantly reduced surface area in the prefrontal cortex and the anterior temporal lobe. We also observed significant group × age interactions for both measures. However, while cortical thickness was best predicted by a quadratic age term, the neurodevelopmental trajectory for measures of surface area was mostly linear. Our findings suggest that ASD is accompanied by age-related and region-specific reductions in cortical thickness and surface area during childhood and early adulthood. Thus, differences in the neurodevelopmental trajectory of maturation for both measures need to be taken into account when interpreting between-group differences overall.





Similar content being viewed by others
References
Amaral DG, Schumann CM, Nordahl CW (2008) Neuroanatomy of autism. Trends Neurosci 31:137–145. doi:10.1016/j.tins.2007.12.005
Blakemore S-J (2008) The social brain in adolescence. Nat Rev Neurosci 9:267–277. doi:10.1038/nrn2353
Carper RA, Courchesne E (2005) Localized enlargement of the frontal cortex in early autism. Biol Psychiatry 57:126–133. doi:10.1016/j.biopsych.2004.11.005
Casanova MF, van Kooten IAJ, Switala AE et al (2006) Minicolumnar abnormalities in autism. Acta Neuropathol 112:287–303. doi:10.1007/s00401-006-0085-5
Castelli F, Frith C, Happé F, Frith U (2002) Autism, Asperger syndrome and brain mechanisms for the attribution of mental states to animated shapes. Brain 125:1839–1849
Courchesne E, Pierce K (2005) Brain overgrowth in autism during a critical time in development: implications for frontal pyramidal neuron and interneuron development and connectivity. Int J Dev Neurosci 23:153–170. doi:10.1016/j.ijdevneu.2005.01.003
Courchesne E, Mouton PR, Calhoun ME et al (2011) Neuron number and size in prefrontal cortex of children with autism. JAMA 306:2001–2010. doi:10.1001/jama.2011.1638
Dale AM, Fischl B, Sereno MI (1999) Cortical surface-based analysis. I. Segmentation and surface reconstruction. Neuroimage 9:179–194. doi:10.1006/nimg.1998.0395
Doyle-Thomas KAR, Kushki A, Duerden EG et al (2013) The effect of diagnosis, age, and symptom severity on cortical surface area in the cingulate cortex and insula in autism spectrum disorders. J Child Neurol 28:732–739. doi:10.1177/0883073812451496
Ecker C, Suckling J, Deoni SC et al (2012) Brain anatomy and its relationship to behavior in adults with autism spectrum disorder: a multicenter magnetic resonance imaging study. Arch Gen Psychiatry 69:195–209. doi:10.1001/archgenpsychiatry.2011.1251
Ecker C, Ginestet C, Feng Y et al (2013a) Brain surface anatomy in adults with autism: the relationship between surface area, cortical thickness, and autistic symptoms. JAMA Psychiatry 70:59–70. doi:10.1001/jamapsychiatry.2013.265
Ecker C, Ronan L, Feng Y et al (2013b) Intrinsic gray-matter connectivity of the brain in adults with autism spectrum disorder. Proc Natl Acad Sci USA. doi:10.1073/pnas.1221880110
Fischl B, Sereno MI, Dale AM (1999a) Cortical surface-based analysis. II: inflation, flattening, and a surface-based coordinate system. Neuroimage 9:195–207. doi:10.1006/nimg.1998.0396
Fischl B, Sereno MI, Tootell RB, Dale AM (1999b) High-resolution intersubject averaging and a coordinate system for the cortical surface. Hum Brain Mapp 8:272–284
Gaffrey MS, Kleinhans NM, Haist F et al (2007) Atypical [corrected] participation of visual cortex during word processing in autism: an fMRI study of semantic decision. Neuropsychologia 45:1672–1684. doi:10.1016/j.neuropsychologia.2007.01.008
Geschwind DH, Levitt P (2007) Autism spectrum disorders: developmental disconnection syndromes. Curr Opin Neurobiol 17:103–111. doi:10.1016/j.conb.2007.01.009
Giedd JN, Blumenthal J, Jeffries NO et al (1999) Brain development during childhood and adolescence: a longitudinal MRI study. Nat Neurosci 2:861–863. doi:10.1038/13158
Hardan AY, Muddasani S, Vemulapalli M et al (2006) An MRI study of increased cortical thickness in autism. Am J Psychiatry 163:1290–1292. doi:10.1176/appi.ajp.163.7.1290
Hardan AY, Libove RA, Keshavan MS et al (2009) A preliminary longitudinal magnetic resonance imaging study of brain volume and cortical thickness in autism. Biol Psychiatry 66:320–326. doi:10.1016/j.biopsych.2009.04.024
Hazlett HC, Poe MD, Gerig G et al (2011) Early brain overgrowth in autism associated with an increase in cortical surface area before age 2 years. Arch Gen Psychiatry 68:467–476. doi:10.1001/archgenpsychiatry.2011.39
Huttenlocher PR, Dabholkar AS (1997) Regional differences in synaptogenesis in human cerebral cortex. J Comp Neurol 387:167–178
Hyde KL, Samson F, Evans AC, Mottron L (2010) Neuroanatomical differences in brain areas implicated in perceptual and other core features of autism revealed by cortical thickness analysis and voxel-based morphometry. Hum Brain Mapp 31:556–566. doi:10.1002/hbm.20887
Langen M, Durston S, Staal WG et al (2007) Caudate nucleus is enlarged in high-functioning medication-naive subjects with autism. Biol Psychiatry 62:262–266. doi:10.1016/j.biopsych.2006.09.040
Langen M, Leemans A, Johnston P et al (2011) Fronto-striatal circuitry and inhibitory control in autism: findings from diffusion tensor imaging tractography. Cortex. doi:10.1016/j.cortex.2011.05.018
Lombardo MV, Barnes JL, Wheelwright SJ, Baron-Cohen S (2007) Self-referential cognition and empathy in autism. PLoS One 2:e883. doi:10.1371/journal.pone.0000883
Lombardo MV, Chakrabarti B, Bullmore ET et al (2010) Atypical neural self-representation in autism. Brain 133:611–624. doi:10.1093/brain/awp306
Lord C, Rutter M, Goode S et al (1989) Autism diagnostic observation schedule: a standardized observation of communicative and social behavior. J Autism Dev Disord 19:185–212
Lord C, Rutter M, Le Couteur A (1994) Autism diagnostic interview-revised: a revised version of a diagnostic interview for caregivers of individuals with possible pervasive developmental disorders. J Autism Dev Disord 24:659–685
Mak-Fan KM, Taylor MJ, Roberts W, Lerch JP (2011) Measures of cortical grey matter structure and development in children with autism spectrum disorder. J Autism Dev Disord. doi:10.1007/s10803-011-1261-6
McAlonan GM, Cheung V, Cheung C et al (2005) Mapping the brain in autism. A voxel-based MRI study of volumetric differences and intercorrelations in autism. Brain 128:268–276. doi:10.1093/brain/awh332
Miyata T, Kawaguchi A, Saito K et al (2004) Asymmetric production of surface-dividing and non-surface-dividing cortical progenitor cells. Development 131:3133–3145. doi:10.1242/dev.01173
Mundy P, Gwaltney M, Henderson H (2010) Self-referenced processing, neurodevelopment and joint attention in autism. Autism 14:408–429. doi:10.1177/1362361310366315
Noctor SC, Martínez-Cerdeño V, Ivic L, Kriegstein AR (2004) Cortical neurons arise in symmetric and asymmetric division zones and migrate through specific phases. Nat Neurosci 7:136–144. doi:10.1038/nn1172
Nordahl CW, Scholz R, Yang X et al (2012) Increased rate of amygdala growth in children aged 2 to 4 years with autism spectrum disorders: a longitudinal study. Arch Gen Psychiatry 69:53–61. doi:10.1001/archgenpsychiatry.2011.145
Panizzon MS, Fennema-Notestine C, Eyler LT et al (2009) Distinct genetic influences on cortical surface area and cortical thickness. Cereb Cortex 19:2728–2735. doi:10.1093/cercor/bhp026
Patterson K, Nestor PJ, Rogers TT (2007) Where do you know what you know? The representation of semantic knowledge in the human brain. Nat Rev Neurosci 8:976–987. doi:10.1038/nrn2277
Paus T (2005) Mapping brain maturation and cognitive development during adolescence. Trends Cogn Sci (Regul Ed) 9:60–68. doi:10.1016/j.tics.2004.12.008
Pessoa L (2008) On the relationship between emotion and cognition. Nat Rev Neurosci 9:148–158. doi:10.1038/nrn2317
Pontious A, Kowalczyk T, Englund C, Hevner RF (2008) Role of intermediate progenitor cells in cerebral cortex development. Dev Neurosci 30:24–32. doi:10.1159/000109848
Rakic P (1995) A small step for the cell, a giant leap for mankind: a hypothesis of neocortical expansion during evolution. Trends Neurosci 18:383–388
Raznahan A, Toro R, Daly E et al (2010) Cortical anatomy in autism spectrum disorder: an in vivo MRI study on the effect of age. Cereb Cortex 20:1332–1340. doi:10.1093/cercor/bhp198
Raznahan A, Shaw P, Lalonde F et al (2011) How does your cortex grow? J Neurosci 31:7174–7177. doi:10.1523/JNEUROSCI.0054-11.2011
Redcay E, Courchesne E (2008) Deviant functional magnetic resonance imaging patterns of brain activity to speech in 2-3-year-old children with autism spectrum disorder. Biol Psychiatry 64:589–598. doi:10.1016/j.biopsych.2008.05.020
Scheel C, Rotarska-Jagiela A, Schilbach L et al (2011) Imaging derived cortical thickness reduction in high-functioning autism: key regions and temporal slope. Neuroimage 58:391–400. doi:10.1016/j.neuroimage.2011.06.040
Schumann CM, Barnes CC, Lord C, Courchesne E (2009) Amygdala enlargement in toddlers with autism related to severity of social and communication impairments. Biol Psychiatry 66:942–949. doi:10.1016/j.biopsych.2009.07.007
Schumann CM, Bloss CS, Barnes CC et al (2010) Longitudinal magnetic resonance imaging study of cortical development through early childhood in autism. J Neurosci 30:4419–4427. doi:10.1523/JNEUROSCI.5714-09.2010
Ségonne F, Dale AM, Busa E et al (2004) A hybrid approach to the skull stripping problem in MRI. Neuroimage 22:1060–1075. doi:10.1016/j.neuroimage.2004.03.032
Shaw P, Lerch J, Greenstein D et al (2006) Longitudinal mapping of cortical thickness and clinical outcome in children and adolescents with attention-deficit/hyperactivity disorder. Arch Gen Psychiatry 63:540–549. doi:10.1001/archpsyc.63.5.540
Silani G, Bird G, Brindley R et al (2008) Levels of emotional awareness and autism: an fMRI study. Soc Neurosci 3:97–112. doi:10.1080/17470910701577020
Waiter GD, Williams JHG, Murray AD et al (2004) A voxel-based investigation of brain structure in male adolescents with autistic spectrum disorder. Neuroimage 22:619–625. doi:10.1016/j.neuroimage.2004.02.029
Wallace GL, Dankner N, Kenworthy L et al (2010) Age-related temporal and parietal cortical thinning in autism spectrum disorders. Brain 133:3745–3754. doi:10.1093/brain/awq279
Wallace GL, Robustelli B, Dankner N et al (2013) Increased gyrification, but comparable surface area in adolescents with autism spectrum disorders. Brain 136:1956–1967. doi:10.1093/brain/awt106
Wang K, Zhang H, Ma D et al (2009) Common genetic variants on 5p14.1 associate with autism spectrum disorders. Nature 459:528–533. doi:10.1038/nature07999
Wechsler D (1999) Wechsler abbreviated scale of intelligence (WASI). Harcourt Assessment, San Antonio
Wing L (1997) The autistic spectrum. Lancet 350:1761–1766. doi:10.1016/S0140-6736(97)09218-0
Winkler AM, Sabuncu MR, Yeo BTT et al (2012) Measuring and comparing brain cortical surface area and other areal quantities. Neuroimage. doi:10.1016/j.neuroimage.2012.03.026
Worsley KJ, Andermann M, Koulis T et al (1999) Detecting changes in nonisotropic images. Hum Brain Mapp 8:98–101
Acknowledgments
This work was supported (1) by the Medical Research Council UK (G0400061 and G0800298), (2) by the Dr. Mortimer and Theresa Sackler Foundation, (3) by the EU-AIMS project (European Autism Interventions—a Multicentre Study for developing New Medications) receiving support from the Innovative Medicines Initiative Joint Undertaking under grant agreement no. 115300, which includes financial contributions from the EU Seventh Framework Programme (FP7/2007-2013), (4) by the NIHR Biomedical Research Centre for Mental Health at King’s College London, Institute of Psychiatry, and (5) by the South London & Maudsley NHS Foundation Trust. We are also grateful to those who agreed to be scanned and who gave their time so generously to this study.
Author information
Authors and Affiliations
Corresponding author
Additional information
C. Ecker and A. Shahidiani contributed equally to the manuscript.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Ecker, C., Shahidiani, A., Feng, Y. et al. The effect of age, diagnosis, and their interaction on vertex-based measures of cortical thickness and surface area in autism spectrum disorder. J Neural Transm 121, 1157–1170 (2014). https://doi.org/10.1007/s00702-014-1207-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00702-014-1207-1