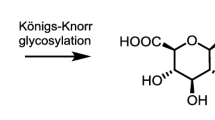
Abstract
Six 3-O- and 6-O-glucosides of morphine and codeine derivatives were synthesized by means of glucosylation with acetobromo-α-d-glucose. O-Glucosylation at C6 was carried out by the Koenigs-Knorr method, whereas the 3-O-glycoside of morphine was synthesized directly upon stirring morphine with acetobromo-α-d-glucose and aqueous sodium hydroxide in acetone. Complete 1H and 13C NMR assignments are presented for each synthesized compound based on one- and two-dimensional homo- and heteronuclear NMR techniques. Circular dichroism, ultraviolet absorbance, and high-resolution mass spectroscopy data ensure identification and structural characterization of the O-glucoside conjugates. The synthesized glucoside conjugates are potential analgesics; the presented spectral and chromatographic data are useful references for various analytical and metabolic studies including samples of biological origin.
Graphical Abstract





Similar content being viewed by others
References
Trescot A, Glaser SE, Hansen H, Benyamin R, Patel S, Manchikanti L (2008) Pain Phys 11:S181
Vallejo R, Barkin RL, Wang VC (2011) Pain Phys 14:E343
Smith HS (2011) Clin J Pain 27:824
Váradi A, Gergely A, Béni S, Jankovics P, Noszál B, Hosztafi S (2011) Eur J Pharm Sci 42:65
Mori M, Oguri K, Yoshimura H, Shimomura K, Kamata O, Ueki S (1972) Life Sci 11:525
Yoshimura H, Ida S, Oguri K, Tsukamoto H (1973) Biochem Pharmacol 22:1423
Pasternak GW, Bodnar RJ, Clark JA, Inturrisi CE (1987) Life Sci 41:2845
Paul D, Standifer KM, Inturrisi CE, Pasternak GW (1989) J Pharmacol Exp Ther 251:477
Chen XY, Zhao LM, Zhong DF (2003) Br J Clin Pharmacol 55:570
Matern H, Matern S (1987) Biochim Biophys Acta. Lipids Lipid Metab 921:1
Paibir SG, Soine WH, Thomas DF, Fisher RA (2004) Eur J Drug Metab Pharmacokinet 29:51
Shipkova M, Armstrong VW, Wieland E, Niedmann PD, Schütz E, Brenner-Weiß G, Voihsel M, Braun F, Oellerich M (1999) Br J Pharmacol 126:1075
Tang BK (1990) Pharmacol Ther 46:53
Tang BK, Kalow W, Grey AA (1978) Res Commun Chem Pathol Pharmacol 21:45
Tang BK, Kalow W, Grey AA (1979) Drug Metab Dispos 7:315
Tjornelund J, Hansen SH, Cornett C (1989) Xenobiotica 19:891
Biasutto L, Marotta E, Bradaschia A, Fallica M, Mattarei A, Garbisa S, Zoratti M, Paradisi C (2009) Bioorg Med Chem Lett 19:6721
Hirpara KV, Aggarwal P, Mukherjee AJ, Joshi NJ, Burman AC (2009) Anticancer Agent Med Chem 9:138
Zhao X, Tao X, Wei D, Song Q (2006) Eur J Med Chem 41:1352
Casparis P, Kuhni E, Leinzinger E (1949) Pharm Acta Helv 24:145
Kováč P, Rice KC (1995) Heterocycles 41:697
Lacy C, Sainsbury M (1995) Tetrahedron Lett 36:3949
Stachulski AV, Jenkins GV (1998) Nat Prod Rep 15:173
Stachulski AV, Scheinmann F, Ferguson JR, Law JL, Lumbard KW, Hopkins P, Patel N, Clarke S, Gloyne A, Joel SP (2003) Bioorg Med Chem Lett 13:1207
Arsequell G, Salvatella M, Valencia G, Fernández-Mayoralas A, Fontanella M, Venturi C, Jiménez-Barbero J, Marrón E, Rodríguez RE (2009) J Med Chem 52:2656
Berrang B, Twine CE, Hennessee GL, Carroll FI (1975) Synth Commun 5:231
Brown RT, Carter NE, Lumbard KW, Scheinmann F (1995) Tetrahedron Lett 36:8661
Yoshimura H, Oguri K, Tsukamoto H (1968) Chem Pharm Bull 16:2114
Mertz AAH (1993) Method for synthesizing glucuronides of 4,5-epoxy morphinanes. PCT Int Appl WO1993005057; March 18, 1993
Welsh LH (1954) J Org Chem 19:1409
Bognár R, Lévai A (1973) Acta Chim Acad Sci Hung 77:435
Bosch ME, Sánchez AR, Rojas FS, Ojeda CB (2007) J Pharm Biomed Anal 43:799
Barrett DA, Pawula M, Knaggs RD, Shaw PN (1998) Chromatographia 47:667
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Váradi, A., Lévai, D., Tóth, G. et al. Glucosides of morphine derivatives: synthesis and characterization. Monatsh Chem 144, 255–262 (2013). https://doi.org/10.1007/s00706-012-0868-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00706-012-0868-4