Abstract
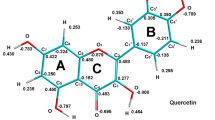
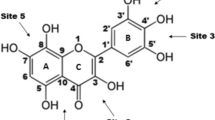
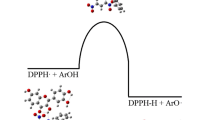
Kaempferol (3,5,7-trihydroxy-2-(4-hydroxyphenyl)-4H-chromen-4-one), one of the most bioactive plant flavonoids, was quantitatively investigated for its ability to bind iron and scavenge DPPH radicals. The DPPH reduction test showed kaempferol and its iron complex to be less potent antioxidants towards DPPH radicals compared to structurally similar flavone molecules quercetin, baicalein, fisetin, morin, and their iron complexes. The equilibrium geometries of free and complexed kaempferol were optimized with the M05-2X functionals and 6-311G(d,p) basis set.
Graphical abstract





Similar content being viewed by others
References
Cody V, Middleton E, Harborne JB (1986) Plant flavonoids in biology and medicine—biochemical, pharmacological and structure-activity relationships. Liss, New York, p 429
Rice-Evans C (2001) Curr Med Chem 8:797
Beard JL, Dawson H, Pinero DJ (1996) Nutr Rev 54:295
Calderón-Montaño JM, Burgos-Morón E, Pérez-Guerrero C, López-Lázaro M (2011) Mini-Rev Med Chem 11:298
Martell AE, Hancock RD (1996) Metal complexes in aqueous solutions. Plenum, New York
Dimitrić Marković JM, Marković ZS, Brdarić TP, Pavelkić VM, Jadranin MB (2011) Food Chem 129:1567
Dimitrić Marković JM, Marković ZS, Brdarić TP, Filipović ND (2011) Dalton Trans 40:4560
Fernandez TM, Mira LM, Florencio HM, Jennings KR (2002) J Inorg Biochem 92:105
Leopoldini M, Russo N, Toscano M (2011) Food Chem 125:288
Brand-Williams W, Cuvelier ME, Berset C (1995) LWT-Food Sci Technol 28:25
Wang L, Tu YC, Lian TW, Hung JT, Yen JH, Wu MJ (2006) J Agric Food Chem 54:9798
Dimitrić Marković JM, Marković ZS, Pašti IA, Brdarić TP, Popović-Bijelića A, Mojović M (2012) Dalton Trans 41:7295
Dangles O, Fargeix G, Dufour C (1999) J Chem Soc Perkin Trans 2:1387
Dangles O, Fargeix G, Dufour C (2000) J Chem Soc Perkin Trans 2:1653
Zhao Y, Truhlar DG (2008) Theor Chem Acc 120:215
Bauernschmitt R, Ahlrichs R (1996) Chem Phys Lett 256:454
O’Boyle NM, Vos JG (2005) GausSum 1.0, Dublin City University. http://gausssum.sourceforge.net. Accessed 1 Jan 2013
Zhurko GA, Zhurko DA (2008) Chemcraft graphical program for working with quantum chemistry results, Chemcraft 1.6, 2008. http://www.chemcraftprog.com. Accessed 1 Jan 2013
Frisch MJ, Trucks GW, Schlegel HB, Scuseria GE, Robb MA, Cheeseman JR, Zakrzewski VG, Montgomery JA Jr, Stratmann RE, Burant JC, Dapprich S, Millam JM, Daniels AD, Kudin KN, Strain MC, Farkas O, Tomasi J, Barone V, Cossi M, Cammi R, Mennucci B, Pomelli C, Adamo C, Clifford S, Ochterski J, Petersson GA, Ayala PY, Cui Q, Morokuma K, Malick AD, Rabuck KD, Raghavachari K, Foresman JB, Cioslowski J, Ortiz JV, Baboul AG, Stefanov BB, Liu G, Liashenko A, Piskorz P, Komaromi I, Gomperts R, Martin RL, Fox DJ, Keith T, Al-Laham MA, Peng CY, Nanayakkara A, Challacombe M, Gill PMW, Johnson B, Chen W, Wong MW, Andres JL, Gonzalez C, Head-Gordon M, Replogle ES, Pople JA (2009) Gaussian 09, Revision A.1-SMP. Gaussian, Wallingford, CT
Acknowledgments
The authors acknowledge financial support of the Ministry of Science of the Republic of Serbia, Grant No. 172015.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Dimitrić Marković, J.M., Amić, D., Lučić, B. et al. Oxidation of kaempferol and its iron(III) complex by DPPH radicals: spectroscopic and theoretical study. Monatsh Chem 145, 557–563 (2014). https://doi.org/10.1007/s00706-013-1135-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00706-013-1135-z