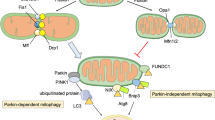
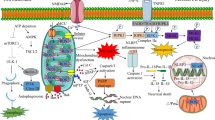
Abstract
Stroke is one of the main causes of mortality and disability in most countries of the world. The only way of managing patients with ischemic stroke is the use of intravenous tissue plasminogen activator and endovascular thrombectomy. However, very few patients receive these treatments as the therapeutic time window is narrow after an ischemic stroke. The paucity of stroke management approaches can only be addressed by identifying new possible therapeutic targets. Mitochondria have been a rare target in the clinical management of stroke. Previous studies have only investigated the bioenergetics and apoptotic roles of this organelle; however, the mitochondrion is now considered as a key organelle that participates in many cellular and molecular functions. This review discusses the mitochondrial mechanisms in cerebral ischemia such as its role in reactive oxygen species (ROS) generation, apoptosis, and electron transport chain dysfunction. Understanding the mechanisms of mitochondria in neural cell death during ischemic stroke might help to design new therapeutic targets for ischemic stroke as well as other neurological diseases.

Similar content being viewed by others
References
Ahangar AA, Saadat P, Heidari B, Taheri ST, Alijanpour S (2017) Sex difference in types and distribution of risk factors in ischemic and hemorrhagic stroke. Int J Stroke:1747493017724626. https://doi.org/10.1177/1747493017724626
Alam MR, Baetz D, Ovize M (2015) Cyclophilin D and myocardial ischemia-reperfusion injury: a fresh perspective. J Mol Cell Cardiol 78:80–89. https://doi.org/10.1016/j.yjmcc.2014.09.026
Amigo I et al (2016) Mitochondrial form, function and signalling in aging. Biochem J 473:3421–3449. https://doi.org/10.1042/bcj20160451
Andrabi SS, Parvez S, Tabassum H (2015) Melatonin and ischemic stroke: mechanistic roles and action. Adv Pharmacol Sci 2015:384750. https://doi.org/10.1155/2015/384750
Andrabi SS, Parvez S, Tabassum H (2017) Neurosteroids and ischemic stroke: progesterone a promising agent in reducing the brain injury in ischemic stroke. J Environ Pathol Toxicol Oncol 36:191–205. https://doi.org/10.1615/JEnvironPatholToxicolOncol.2017017156
Andrabi SS, Ali M, Tabassum H, Parveen S, Parvez S (2019) Pramipexole prevents ischemic cell death via mitochondrial pathways in ischemic stroke. Dis Model Mech. https://doi.org/10.1242/dmm.033860
Andrienko TN, Pasdois P, Pereira GC, Ovens MJ, Halestrap AP (2017) The role of succinate and ROS in reperfusion injury - a critical appraisal. J Mol Cell Cardiol 110:1–14. https://doi.org/10.1016/j.yjmcc.2017.06.016
Archer DP, Walker AM, McCann SK, Moser JJ, Appireddy RM (2017) Anesthetic neuroprotection in experimental stroke in rodents: a systematic review and meta-analysis. Anesthesiology 126:653–665. https://doi.org/10.1097/aln.0000000000001534
Azad P, Haddad GG (2013) Genetic animal models of preconditioning. Transl Stroke Res 4:51–55. https://doi.org/10.1007/s12975-012-0218-1
Baines CP, Kaiser RA, Sheiko T, Craigen WJ, Molkentin JD (2007) Voltage-dependent anion channels are dispensable for mitochondrial-dependent cell death. Nat Cell Biol 9:550–555. https://doi.org/10.1038/ncb1575
Bejot Y, Bailly H, Durier J, Giroud M (2016) Epidemiology of stroke in Europe and trends for the 21st century Presse medicale (Paris, France : 1983) 45:e391–e398. https://doi.org/10.1016/j.lpm.2016.10.003
Bakthavachalam P, Shanmugam PS (2017) Mitochondrial dysfunction - silent killer in cerebral ischemia. J Neurol Sci 375:417–423. https://doi.org/10.1016/j.jns.2017.02.043
Banerjee A, Larsen RS, Philpot BD, Paulsen O (2016) Roles of rresynaptic NMDA receptors in neurotransmission and plasticity. Trends Neurosci 39:26–39. https://doi.org/10.1016/j.tins.2015.11.001
Bernardi P (2013) The mitochondrial permeability transition pore: a mystery solved? Front Physiol 4:95. https://doi.org/10.3389/fphys.2013.00095
Bettler B, Fakler B (2017) Ionotropic AMPA-type glutamate and metabotropic GABAB receptors: determining cellular physiology by proteomes. Curr Opin Neurobiol 45:16–23. https://doi.org/10.1016/j.conb.2017.02.011
Brassai A, Suvanjeiev RG, Ban EG, Lakatos M (2015) Role of synaptic and nonsynaptic glutamate receptors in ischaemia induced neurotoxicity. Brain Res Bull 112:1–6. https://doi.org/10.1016/j.brainresbull.2014.12.007
Brenner C, Moulin M (2012) Physiological roles of the permeability transition pore. Circ Res 111:1237–1247. https://doi.org/10.1161/circresaha.112.265942
Buoncervello M, Marconi M, Care A, Piscopo P, Malorni W, Matarrese P (2017) Preclinical models in the study of sex differences. Clin Sci (London, England : 1979) 131:449–469. https://doi.org/10.1042/cs20160847
Borutaite V (2010) Mitochondria as decision-makers in cell death Environmental and molecular mutagenesis 51:406–416. https://doi.org/10.1002/em.20564
Chang P, Prabhakaran S (2017) Recent advances in the management of acute ischemic stroke. F1000Research 6. https://doi.org/10.12688/f1000research.9191.1
Cho BB, Toledo-Pereyra LH (2008) Caspase-independent programmed cell death following ischemic stroke. J Investig Surg 21:141–147. https://doi.org/10.1080/08941930802029945
Drose S, Stepanova A, Galkin A (2016) Ischemic A/D transition of mitochondrial complex I and its role in ROS generation. Biochim Biophys Acta 1857:946–957. https://doi.org/10.1016/j.bbabio.2015.12.013
El Amki M, Clavier T, Perzo N, Bernard R, Guichet PO, Castel H (2015) Hypothalamic, thalamic and hippocampal lesions in the mouse MCAO model: potential involvement of deep cerebral arteries? J Neurosci Methods 254:80–85. https://doi.org/10.1016/j.jneumeth.2015.07.008
Ertracht O, Malka A, Atar S, Binah O (2014) The mitochondria as a target for cardioprotection in acute myocardial ischemia. Pharmacol Ther 142:33–40. https://doi.org/10.1016/j.pharmthera.2013.11.003
Fan J, Dawson TM, Dawson VL (2017) Cell Death mechanisms of neurodegeneration. Adv Neurobiol 15:403–425. https://doi.org/10.1007/978-3-319-57193-5_16
Fidaleo M, Cavallucci V, Pani G (2017) Nutrients, neurogenesis and brain ageing: from disease mechanisms to therapeutic opportunities. Biochem Pharmacol. https://doi.org/10.1016/j.bcp.2017.05.016
Gollihue JL, Rabchevsky AG (2017) Prospects for therapeutic mitochondrial transplantation. Mitochondrion. https://doi.org/10.1016/j.mito.2017.05.007
Granger DN, Kvietys PR (2015) Reperfusion injury and reactive oxygen species: the evolution of a concept. Redox Biol 6:524–551. https://doi.org/10.1016/j.redox.2015.08.020
Green DR, Llambi F (2015) Cell death signaling. Cold Spring Harb Perspect Biol 7. https://doi.org/10.1101/cshperspect.a0060807
Grimm A, Eckert A (2017) Brain aging and neurodegeneration: from a mitochondrial point of view. J Neurochem. https://doi.org/10.1111/jnc.14037
He J, Ford HC, Carroll J, Ding S, Fearnley IM, Walker JE (2017) Persistence of the mitochondrial permeability transition in the absence of subunit c of human ATP synthase. Proc Natl Acad Sci U S A 114:3409–3414. https://doi.org/10.1073/pnas.1702357114
Hurst S, Hoek J, Sheu SS (2017) Mitochondrial Ca2+ and regulation of the permeability transition pore. J Bioenerg Biomembr 49:27–47. https://doi.org/10.1007/s10863-016-9672-x
Izzo V, Bravo-San Pedro JM, Sica V, Kroemer G, Galluzzi L (2016) Mitochondrial Permeability transition: new findings and persisting uncertainties. Trends Cell Biol 26:655–667. https://doi.org/10.1016/j.tcb.2016.04.006
Jordan J, de Groot PW, Galindo MF (2011) Mitochondria: the headquarters in ischemia-induced neuronal death. Cent Nerv Syst Agents Med Chem 11:98–106
Julien O, Wells JA (2017) Caspases and their substrates. Cell Death Differ. https://doi.org/10.1038/cdd.2017.44
Karch J, Molkentin JD (2014) Identifying the components of the elusive mitochondrial permeability transition pore. Proc Natl Acad Sci U S A 111:10396–10397. https://doi.org/10.1073/pnas.1410104111
Karsy M, Brock A, Guan J, Taussky P, Kalani MY, Park MS (2017) Neuroprotective strategies and the underlying molecular basis of cerebrovascular stroke Neurosurgical focus 42:E3. https://doi.org/10.3171/2017.1.Focus16522
Khoshnam SE, Winlow W, Farzaneh M, Farbood Y, Moghaddam HF (2017) Pathogenic mechanisms following ischemic stroke. Neurol Sci. https://doi.org/10.1007/s10072-017-2938-1
Kiselyov K, Muallem S (2016) ROS and intracellular ion channels Cell calcium 60:108–114. https://doi.org/10.1016/j.ceca.2016.03.004
Korge P, John SA, Calmettes G, Weiss JN (2017) Reactive oxygen species production induced by pore opening in cardiac mitochondria: the role of complex II. J Biol Chem. https://doi.org/10.1074/jbc.M116.768325
Ma Y, Zechariah A, Qu Y, Hermann DM (2012) Effects of vascular endothelial growth factor in ischemic stroke. J Neurosci Res 90:1873–1882. https://doi.org/10.1002/jnr.23088
Ma D, Feng L, Deng F, Feng JC (2017a) Overview of Experimental and Clinical Findings regarding the Neuroprotective Effects of Cerebral Ischemic Postconditioning. Biomed Res Int 2017:6891645. https://doi.org/10.1155/2017/6891645
Ma MW, Wang J, Zhang Q, Wang R, Dhandapani KM, Vadlamudi RK, Brann DW (2017b) NADPH oxidase in brain injury and neurodegenerative disorders. Mol Neurodegener 12:7. https://doi.org/10.1186/s13024-017-0150-7
Maldonado EN, Lemasters JJ (2014) ATP/ADP ratio, the missed connection between mitochondria and the Warburg effect. Mitochondrion 19(Pt a):78–84. https://doi.org/10.1016/j.mito.2014.09.002
Mozaffarian D et al (2016) Executive summary: heart disease and stroke statistics--2016 update: a report from the American Heart Association. Circulation 133:447–454. https://doi.org/10.1161/cir.0000000000000366
Neumann JT et al (2013) Association of MR-proadrenomedullin with cardiovascular risk factors and subclinical cardiovascular disease Atherosclerosis 228:451–459. https://doi.org/10.1016/j.atherosclerosis.2013.03.006
Osman MM, Lulic D, Glover L, Stahl CE, Lau T, van Loveren H, Borlongan CV (2011) Cyclosporine-A as a neuroprotective agent against stroke: its translation from laboratory research to clinical application. Neuropeptides 45:359–368. https://doi.org/10.1016/j.npep.2011.04.002
Perez MJ, Quintanilla RA (2017) Development or disease: duality of the mitochondrial permeability transition pore. Dev Biol 426:1–7. https://doi.org/10.1016/j.ydbio.2017.04.018
Piotrowska A, Bartnik E (2014) The role of reactive oxygen species and mitochondria in aging. Postepy Biochem 60:240–247
Ponnalagu D, Singh H (2017) Anion channels of mitochondria. Handb Exp Pharmacol 240:71–101. https://doi.org/10.1007/164_2016_39
Puyal J, Ginet V, Clarke PG (2013) Multiple interacting cell death mechanisms in the mediation of excitotoxicity and ischemic brain damage: a challenge for neuroprotection. Prog Neurobiol 105:24–48. https://doi.org/10.1016/j.pneurobio.2013.03.002
Rastogi R, Geng X, Li F, Ding Y (2016) NOX activation by subunit interaction and underlying mechanisms in disease. Front Cell Neurosci 10:301. https://doi.org/10.3389/fncel.2016.00301
Rehni AK, Liu A, Perez-Pinzon MA, Dave KR (2017) Diabetic aggravation of stroke and animal models. Exp Neurol 292:63–79. https://doi.org/10.1016/j.expneurol.2017.03.004
Rekuviene E, Ivanoviene L, Borutaite V, Morkuniene R (2017) Rotenone decreases ischemia-induced injury by inhibiting mitochondrial permeability transition in mature brains. Neurosci Lett 653:45–50. https://doi.org/10.1016/j.neulet.2017.05.028
Rodriguez-Lara SQ, Cardona-Munoz EG, Ramirez-Lizardo EJ, Totsuka-Sutto SE, Castillo-Romero A, Garcia-Cobian TA, Garcia-Benavides L (2016) Alternative interventions to prevent oxidative damage following ischemia/reperfusion. Oxidative Med Cell Longev 2016:7190943. https://doi.org/10.1155/2016/7190943
Roque C, Baltazar G (2017) Impact of astrocytes on the injury induced by in vitro ischemia. Cell Mol Neurobiol. https://doi.org/10.1007/s10571-017-0483-3
Rousselet E, Kriz J, Seidah NG (2012) Mouse model of intraluminal MCAO: cerebral infarct evaluation by cresyl violet staining. J Visualized Exp. https://doi.org/10.3791/4038
Schmidt A, Minnerup J (2016) Promoting recovery from ischemic stroke. Expert Rev Neurother 16:173–186. https://doi.org/10.1586/14737175.2016.1134324
Shakeri R, Kheirollahi A, Davoodi J (2017) Apaf-1: regulation and function in cell death. Biochimie 135:111–125. https://doi.org/10.1016/j.biochi.2017.02.001
Siket MS (2016) Treatment of Acute Ischemic Stroke Emergency medicine clinics of North America 34:861–882. https://doi.org/10.1016/j.emc.2016.06.009
Sims NR, Yew WP (2017) Reactive astrogliosis in stroke: Contributions of astrocytes to recovery of neurological function Neurochemistry international 107:88–103. https://doi.org/10.1016/j.neuint.2016.12.016
Skelding KA, Arellano JM, Powis DA, Rostas JA (2014) Excitotoxic stimulation of brain microslices as an in vitro model of stroke. J Visualized Exp:e51291. https://doi.org/10.3791/51291
Sommer CJ (2017) Ischemic stroke: experimental models and reality. Acta Neuropathol 133:245–261. https://doi.org/10.1007/s00401-017-1667-0
Song FE, Huang JL, Lin SH, Wang S, Ma GF, Tong XP (2017) Roles of NG2-glia in ischemic stroke. CNS Neurosci Ther 23:547–553. https://doi.org/10.1111/cns.12690
Takatsuru Y, Nabekura J, Koibuchi N (2014) Contribution of neuronal and glial circuit in intact hemisphere for functional remodeling after focal ischemia Neuroscience research 78:38–44. https://doi.org/10.1016/j.neures.2013.07.004
Tang YC, Tian HX, Yi T, Chen HB (2016) The critical roles of mitophagy in cerebral ischemia. Protein Cell 7:699–713. https://doi.org/10.1007/s13238-016-0307-0
Taylor RA, Sansing LH (2013) Microglial responses after ischemic stroke and intracerebral hemorrhage. Clin Dev Immunol 2013:746068. https://doi.org/10.1155/2013/746068
Thakur KT et al (2016) Neurological Disorders. In: Patel V, Chisholm D, Dua T, Laxminarayan R, Medina-Mora ME (eds) Mental, neurological, and substance use disorders: disease control priorities, vol 4, Third edn. 2016 International Bank for Reconstruction and Development / The World Bank, Washington DC. https://doi.org/10.1596/978-1-4648-0426-7_ch5
Thompson JW, Narayanan SV, Perez-Pinzon MA (2012) Redox signaling pathways involved in neuronal ischemic preconditioning. Curr Neuropharmacol 10:354–369. https://doi.org/10.2174/157015912804143577
Thornton C, Hagberg H (2015) Role of mitochondria in apoptotic and necroptotic cell death in the developing brain Clinica chimica acta. IntJ Clinical Chem 451:35–38. https://doi.org/10.1016/j.cca.2015.01.026
Thrift AG, Cadilhac DA, Thayabaranathan T, Howard G, Howard VJ, Rothwell PM, Donnan GA (2014) Global stroke statistics. Int J Stroke 9:6–18. https://doi.org/10.1111/ijs.12245
Vakifahmetoglu-Norberg H, Ouchida AT, Norberg E (2017) The role of mitochondria in metabolism and cell death. Biochem Biophys Res Commun 482:426–431. https://doi.org/10.1016/j.bbrc.2016.11.088
Vandenberg RJ, Ryan RM (2013) Mechanisms of glutamate transport. Physiol Rev 93:1621–1657. https://doi.org/10.1152/physrev.00007.2013
Wasay M, Khatri IA, Kaul S (2014) Stroke in South Asian countries. Nat Rev Neurol 10:135–143. https://doi.org/10.1038/nrneurol.2014.13
Wijermars LG, Schaapherder AF, Kostidis S, Wust RC, Lindeman JH (2016) Succinate accumulation and ischemia-reperfusion injury: of mice but not men, a study in renal ischemia-reperfusion. Am J Transplant Off J Am Soc Transplant Am Soc Transplant Surg 16:2741–2746. https://doi.org/10.1111/ajt.13793
Yagami T, Koma H, Yamamoto Y (2016) Pathophysiological roles of cyclooxygenases and prostaglandins in the central nervous system. Mol Neurobiol 53:4754–4771. https://doi.org/10.1007/s12035-015-9355-3
Yang CH, Yen TL, Hsu CY, Thomas PA, Sheu JR, Jayakumar T (2017) Multi-targeting andrographolide, a novel NF-kappaB inhibitor, as a potential therapeutic agent for stroke. Int J Mol Sci 18. https://doi.org/10.3390/ijms18081638
Yuan J, Najafov A, Py BF (2016) Roles of caspases in necrotic cell death. Cell 167:1693–1704. https://doi.org/10.1016/j.cell.2016.11.047
Funding
This study is funded by the DST-PURSE program through grant (No. SR/PURSE Phase 2/39 (C)) and SERB-EMR grant (2016/001070/HS).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Handling Editor: Klaudia Brix
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Andrabi, S.S., Parvez, S. & Tabassum, H. Ischemic stroke and mitochondria: mechanisms and targets. Protoplasma 257, 335–343 (2020). https://doi.org/10.1007/s00709-019-01439-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00709-019-01439-2