Abstract
Density functional theory (DFT) calculations at the B3LYP/6-31G* level for isobenzofuran 1 and eleven benzannulated derivatives of types 2 and 3 have been performed in order to compare their relative reactivities as dienes in Diels–Alder reactions. The transition state (TS) energies for their reactions with ethylene have been determined and shown to form a linear correlation between activation energies and structure count (SC) ratios. TS energies as a method for comparison of diene reactivities can be applied to IBFs bearing substituents on the ring as well as those containing heteroatoms, for which the SC ratio method failed. Different measures of aromaticity of benzannulated IBFs indicated a decrease in aromaticity going from 4 to 14, which is also reflected in their reactivity as a dienes in Diels–Alder reaction.
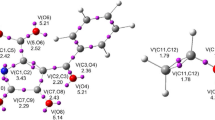
Figure Isobenzofuran 1 and benzannulated isobenzofurans 2 and 3











Similar content being viewed by others
References
Friedrichsen W (1999) Adv Heterocycl Chem 73:1–96
Peters O, Friedrichsen W (1995) Trends Heterocycl Chem 4:217–228
Wiersum UE (1981) Aldrichimica Acta 14:53–58
Friedrichsen W (1980) Adv Heterocycl Chem 26:135–241
Haddadin MJ (1978) Heterocycles 9:865–901
See for instance Warrener RN, Wang S, Butler DN, Russell RA (1997) Synlett 44–46
Warrener RN, Wang S, Russell RA, Gunter MJ (1997) Synlett 47–50
Warrener RN, Wang S, Butler DN, Russell RA (1997) Tetrahedron 53:3975–3990
Smith JG, Dibble PW, Sandborn RE (1983) J Chem Soc Chem Commun 1197–1198
Dibble PW, Rodrigo R (1988) Org Mass Spectrom 23:743–750
Moursounidis J, Wege D (1988) Aust J Chem 41:235–249
Tu NPW, Yip JC, Dibble PW (1996) Synthesis 77–81
Juršić BS (1995) J Chem Soc Perkin Trans 2 1217–1222
Juršić BS (1997) Tetrahedron 53:13285 −13294
Juršić BS (1996) J Heterocycl Chem 33:1079–1081
Frisch MJ, Trucks GW, Schlegel HB, Scuseria GE, Robb MA, Cheeseman JR, Zakrzewski VG, Montgomery Jr. JA, Stratmann RE, Burant JC, Dapprich S, Millam JM, Daniels AD, Kudin KN, Strain MC, Farkas O, Tomasi J, Barone V, Cossi M, Cammi R, Mennucci B, Pomelli C, Adamo C, Clifford S, Ochterski J, Petersson GA, Ayala PY, Cui Q, Morokuma K, Malick DK, Rabuck AD, Raghavachari K, Foresman JB, Cioslowski J, Ortiz JV, Stefanov BB, Liu G, Liashenko A, Piskorz P, Komaromi I, Gomperts R, Martin RL, Fox DJ, Keith T, Al-Laham MA, Peng CY, Nanayakkara A, Gonzalez C, Challacombe M, Gill PMW, Johnson B, Chen W, Wong MW, Andres JL, Gonzalez C, Head-Gordon M, Replogle ES, Pople JA (1998) Gaussian 98, Revision A.5. Gaussian, Pittsburgh, Pa.
Becke AD (1993) J Chem Phys 98:1372–1377
Lee C, Yang W, Parr RG (1988) Phys Rev B 37:785–789
Herndon WC (1975) J Org Chem 40:3583–3586
Pearson RG (1989) J Org Chem 54:1432–1435
Zhou Z, Parr RG (1989) J Am Chem Soc 111:7371–7379
Bean GP (1998) J Org Chem 63:2497–2506
Katritzky AR, Barczynski P, Musumarra G, Pisano D, Szafran M (1989) J Am Chem Soc 111:7–15
Scott AP, Agranat I, Biedermann PU, Riggs NV, Radom L (1997) J Org Chem 62:2026–2038
Juršić BS (1999) Theochem 468:171–179
Manoharan M, De Proft F, Geerlings P (2000) J Chem Soc Perkin 2 8:1767–1773
Subramanian G, Schleyer PvR, Jiao H (1996) Angew Chem, Int Ed Engl 35:2638–2641
Juršić BS (1998) Theochem 427:165–169
Gugelchuk MM, Chan PCM, Sprules TJ (1994) J Org Chem 59:7723–7731
Hammond GS (1955) J Am Chem Soc 77:334–338
Acknowledgments
We thank the Australian Research Council (ARC) for funding.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Margetić, D., Warrener, R.N. & Dibble, P.W. Diels–Alder reactivity of benzannulated isobenzofurans as assessed by density functional theory. J Mol Model 10, 87–93 (2004). https://doi.org/10.1007/s00894-003-0143-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00894-003-0143-z