Abstract
Objectives
Iguratimod (IGU) is a conventional synthetic disease-modifying drug that has been approved based on its additive effects with methotrexate (MTX) for the treatment of rheumatoid arthritis (RA). The objective of the study is to establish the effectiveness of IGU with versus IGU without MTX irrespective of whether MTX is well tolerated or not by the patients.
Methods
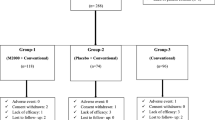
Disease activity scores in 177 RA patients treated using IGU were retrospectively evaluated at baseline and after 4, 12, and 24 weeks, and adverse events (AEs) were noted.
Results
IGU reduced the disease activity parameters, disease activity score (DAS)-ESR, DAS-CRP, the simplified disease activity index (SDAI), and clinical disease activity index (CDAI) in the concomitant MTX and non-MTX, female and male, and young and elderly patient groups after 24 weeks. Multivariate analysis demonstrated that IGU was more effective with concomitant MTX and in elderly and male patients. Severe AEs were observed only in the elderly group: two cases of pneumonia, 1 of pneumocystis pneumonia, 1 of heart failure, and 1 of salivary gland adenoma.
Conclusions
IGU is effective for RA, especially with concomitant MTX, and in elderly and male patients.
Key Points • Iguratimod is effective for RA, especially with concomitant MTX, and in elderly and male patients. • Since all serious adverse events were in the elderly group in this study, sufficient monitoring for adverse events, especially for elderly RA patients, is needed during iguratimod therapy. |



Similar content being viewed by others
References
Tanaka K, Aikawa Y, Kawasaki H, Asaoka K, Inaba T, Yoshida C (1992) Pharmacological studies on 3-formylamino-7-methylsulfonylamino-6-phenoxy-4H-1-benzopyran-4-one (T-614), a novel antiinflammatory agent. 4th communication: inhibitory effect on the production of interleukin-1 and interleukin-6. Aust J Pharm 15(11):649–655
Tanaka K, Makino S, Shimotori T, Aikawa Y, Inaba T, Yoshida C (1992) Pharmacological studies of the new antiinflammatory agent 3-formylamino-7-methylsulfonylamino-6-phenoxy-4′-1-benzopyran-4-o ne. 2nd communication: effect on the arachidonic acid cascades. Arzneimittelforschung 42(7):945–950
Tanaka K, Shimotori T, Makino S, Aikawa Y, Inaba T, Yoshida C, Takano S (1992) Pharmacological studies of the new antiinflammatory agent 3-formylamino-7-methylsulfonylamino-6-phenoxy-4H-1-benzopyran-4-o ne. 1st communication: antiinflammatory, analgesic and other related properties. Arzneimittelforschung 42(7):935–944
Tanaka K, Shimotori T, Makino S, Eguchi M, Asaoka K, Kitamura R, Yoshida C (1992) Pharmacological studies on 3-formylamino-7-methylsulfonylamino-6-phenoxy-4H-1-benzopyran-4-one (T-614), a novel antiinflammatory agent. 3rd communication: the involvement of bradykinin in its analgesic actions. Aust J Pharm 15(11):641–647
Song J, Liu H, Zhu Q, Miao Y, Wang F, Yang F, Cheng W, Xi Y, Niu X, He D, Chen G (2018) T-614 promotes Osteoblastic cell differentiation by increasing Dlx5 expression and regulating the activation of p38 and NF-kappaB. Biomed Res Int 2018:4901591–4901598. https://doi.org/10.1155/2018/4901591
Aikawa Y, Yamamoto M, Yamamoto T, Morimoto K, Tanaka K (2002) An anti-rheumatic agent T-614 inhibits NF-kappaB activation in LPS- and TNF-alpha-stimulated THP-1 cells without interfering with IkappaBalpha degradation. Inflamm Res 51(4):188–194
Kawakami A, Tsuboi M, Urayama S, Matsuoka N, Yamasaki S, Hida A, Aoyagi T, Furuichi I, Nakashima T, Migita K, Kawabe Y, Nakashima M, Origuchi T, Eguchi K (1999) Inhibitory effect of a new anti-rheumatic drug T-614 on costimulatory molecule expression, cytokine production, and antigen presentation by synovial cells. J Lab Clin Med 133(6):566–574
Kohno M, Aikawa Y, Tsubouchi Y, Hashiramoto A, Yamada R, Kawahito Y, Inoue K, Kusaka Y, Kondo M, Sano H (2001) Inhibitory effect of T-614 on tumor necrosis factor-alpha induced cytokine production and nuclear factor-kappaB activation in cultured human synovial cells. J Rheumatol 28(12):2591–2596
Tanaka K, Yamamoto T, Aikawa Y, Kizawa K, Muramoto K, Matsuno H, Muraguchi A (2003) Inhibitory effects of an anti-rheumatic agent T-614 on immunoglobulin production by cultured B cells and rheumatoid synovial tissues engrafted into SCID mice. Rheumatology (Oxford) 42(11):1365–1371. https://doi.org/10.1093/rheumatology/keg381
Hara M, Abe T, Sugawara S, Mizushima Y, Hoshi K, Irimajiri S, Hashimoto H, Yoshino S, Matsui N, Nobunaga M, Nakano S (2007) Efficacy and safety of iguratimod compared with placebo and salazosulfapyridine in active rheumatoid arthritis: a controlled, multicenter, double-blind, parallel-group study. Mod Rheumatol 17(1):1–9. https://doi.org/10.1007/s10165-006-0542-y
Nozaki Y, Inoue A, Kinoshita K, Funauchi M, Matsumura I (2020) Efficacy of iguratimod vs. salazosulfapyridine as the first-line csDMARD for rheumatoid arthritis. Mod Rheumatol 30(2):249–258. https://doi.org/10.1080/14397595.2019.1572267
Hara M, Ishiguro N, Katayama K, Kondo M, Sumida T, Mimori T, Soen S, Nagai K, Yamaguchi T, Yamamoto K, Iguratimod-Clinical Study G (2014) Safety and efficacy of combination therapy of iguratimod with methotrexate for patients with active rheumatoid arthritis with an inadequate response to methotrexate: an open-label extension of a randomized, double-blind, placebo-controlled trial. Mod Rheumatol 24(3):410–418. https://doi.org/10.3109/14397595.2013.843756
Singh JA, Saag KG, Bridges SL Jr, Akl EA, Bannuru RR, Sullivan MC, Vaysbrot E, McNaughton C, Osani M, Shmerling RH, Curtis JR, Furst DE, Parks D, Kavanaugh A, O'Dell J, King C, Leong A, Matteson EL, Schousboe JT, Drevlow B, Ginsberg S, Grober J, St Clair EW, Tindall E, Miller AS, McAlindon T (2016) 2015 American College of Rheumatology Guideline for the treatment of rheumatoid arthritis. Arthritis Rheum 68(1):1–26. https://doi.org/10.1002/art.39480
Smolen JS, Landewe R, Bijlsma J, Burmester G, Chatzidionysiou K, Dougados M, Nam J, Ramiro S, Voshaar M, van Vollenhoven R, Aletaha D, Aringer M, Boers M, Buckley CD, Buttgereit F, Bykerk V, Cardiel M, Combe B, Cutolo M, van Eijk-Hustings Y, Emery P, Finckh A, Gabay C, Gomez-Reino J, Gossec L, Gottenberg JE, Hazes JM, Huizinga T, Jani M, Karateev D, Kouloumas M, Kvien T, Li Z, Mariette X, McInnes I, Mysler E, Nash P, Pavelka K, Poor G, Richez C, van Riel P, Rubbert-Roth A, Saag K, da Silva J, Stamm T, Takeuchi T, Westhovens R, de Wit M, van der Heijde D (2017) EULAR recommendations for the management of rheumatoid arthritis with synthetic and biological disease-modifying antirheumatic drugs: 2016 update. Ann Rheum Dis 76:960–977. https://doi.org/10.1136/annrheumdis-2016-210715
Atsumi T, Yamamoto K, Takeuchi T, Yamanaka H, Ishiguro N, Tanaka Y, Eguchi K, Watanabe A, Origasa H, Yasuda S, Yamanishi Y, Kita Y, Matsubara T, Iwamoto M, Shoji T, Okada T, van der Heijde D, Miyasaka N, Koike T (2016) The first double-blind, randomised, parallel-group certolizumab pegol study in methotrexate-naive early rheumatoid arthritis patients with poor prognostic factors, C-OPERA, shows inhibition of radiographic progression. Ann Rheum Dis 75(1):75–83. https://doi.org/10.1136/annrheumdis-2015-207511
Arnett FC, Edworthy SM, Bloch DA, McShane DJ, Fries JF, Cooper NS, Healey LA, Kaplan SR, Liang MH, Luthra HS et al (1988) The American Rheumatism Association 1987 revised criteria for the classification of rheumatoid arthritis. Arthritis Rheum 31(3):315–324
Aletaha D, Neogi T, Silman AJ, Funovits J, Felson DT, Bingham CO 3rd, Birnbaum NS, Burmester GR, Bykerk VP, Cohen MD, Combe B, Costenbader KH, Dougados M, Emery P, Ferraccioli G, Hazes JM, Hobbs K, Huizinga TW, Kavanaugh A, Kay J, Kvien TK, Laing T, Mease P, Menard HA, Moreland LW, Naden RL, Pincus T, Smolen JS, Stanislawska-Biernat E, Symmons D, Tak PP, Upchurch KS, Vencovsky J, Wolfe F, Hawker G (2010) 2010 rheumatoid arthritis classification criteria: an American College of Rheumatology/European League Against Rheumatism collaborative initiative. Ann Rheum Dis 69(9):1580–1588. https://doi.org/10.1136/ard.2010.138461
Aletaha D, Nell VP, Stamm T, Uffmann M, Pflugbeil S, Machold K, Smolen JS (2005) Acute phase reactants add little to composite disease activity indices for rheumatoid arthritis: validation of a clinical activity score. Arthritis Res Ther 7(4):R796–R806. https://doi.org/10.1186/ar1740
Rubin DB, Schenker N (1991) Multiple imputation in health-care databases: an overview and some applications. Stat Med 10(4):585–598. https://doi.org/10.1002/sim.4780100410
Steinbrocker O, Traeger CH, Batterman RC (1949) Therapeutic criteria in rheumatoid arthritis. J Am Med Assoc 140(8):659–662
Genestier L, Paillot R, Fournel S, Ferraro C, Miossec P, Revillard JP (1998) Immunosuppressive properties of methotrexate: apoptosis and clonal deletion of activated peripheral T cells. J Clin Invest 102(2):322–328. https://doi.org/10.1172/JCI2676
Rosenthal GJ, Germolec DR, Lamm KR, Ackermann MF, Luster MI (1987) Comparative effects on the immune system of methotrexate and trimetrexate. Int J Immunopharmacol 9(7):793–801. https://doi.org/10.1016/0192-0561(87)90075-0
Weinblatt ME, Maier AL (1989) Treatment of rheumatoid arthritis. Arthritis Care Res 2(3):S23–S32. https://doi.org/10.1002/anr.1790020311
Ishiguro N, Shibata K, Yoshimura A, Ikeuchi S, Ishii M (2019) Investigation of the predictors of the response to iguratimod therapy: a post-hoc analysis of post-marketing surveillance study. Mod Rheumatol:1–7. https://doi.org/10.1080/14397595.2019.1649110
Mimori T, Harigai M, Atsumi T, Fujii T, Kuwana M, Matsuno H, Momohara S, Takei S, Tamura N, Takasaki Y, Ikeuchi S, Kushimoto S, Koike T (2017) Safety and effectiveness of 24-week treatment with iguratimod, a new oral disease-modifying antirheumatic drug, for patients with rheumatoid arthritis: interim analysis of a post-marketing surveillance study of 2679 patients in Japan. Mod Rheumatol 27(5):755–765. https://doi.org/10.1080/14397595.2016.1265695
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
SM received lectures fees from Janssen Pharmaceutical K.K.; Bristol-Myers Squibb K.K.; Chugai Pharmaceutical Co.; Mitsubishi-Tanabe Pharma Co.; Novartis Pharma K.K.; and Ono Pharmaceutical Co. TN received grant/research support from Chugai Pharmaceutical Co.; Eisai Co., Ltd.; Takeda Pharmaceutical Co., Ltd.; Teijin Pharma Ltd.; Eli Lilly Japan K.K.; Bristol-Myers Squibb K.K.; AbbVie GK.; Ono Pharmaceutical Co., Ltd.; Novartis Pharma K.K.; Asahikasei Pharma Corp.; Mitsubishi-Tanabe Pharma Co.; Astellas Pharma Inc.; Ayumi Pharmaceutical Corporation; Pfizer Japan Inc.; Daiichi Sankyo Co., Ltd.; Shionogi & Co., Ltd.; Sanofi K.K.; Nippon Kayaku Co., Ltd.; Yutoku Pharmaceutical Ind. Co., Ltd.; and Actelion Pharmaceutical Japan Ltd.; consultant fees from UCB Japan Co., Ltd.; Eisai Co., Ltd.; Ono Pharmaceutical Co., Ltd.; Gilead Science Inc.; and Chugai Pharmaceutical Co.; and speaker fees from Mitsubishi-Tanabe Pharma Co.; Chugai Pharmaceutical Co.; Eisai Co., Ltd.; Astellas Pharma Inc.; Janssen Pharmaceutical K.K.; Ayumi Pharmaceutical Corporation; Pfizer Japan Inc.; Asahikasei Pharma Corp.; Sanofi K.K., Daiichi Sankyo Co., Ltd.; Otsuka Pharmaceutical Co., Ltd.; and AbbVie GK. HY received consulting fees, speaking fees, and/or honoraria from AbbVie GK.; Asahikasei Pharma Corp.; Astellas Pharma Inc.; Daiichi Sankyo Co., Ltd.; Eisai Co., Ltd.; Kissei Pharmaceutical Co., Ltd.; Takeda Pharmaceutical Co., Ltd.; Mitsubishi-Tanabe Pharma Co.; Chugai Pharmaceutical Co.; Novartis Pharma K.K.; Eli Lilly Japan K.K.; Pfizer Japan Inc.; Janssen Pharmaceutical K.K.; Sanofi K.K.; Teijin Pharma Ltd.; Boehringer Ingelheim International GmbH; and Bayer Yakuhin, Ltd.; and research grants from Mitsubishi-Tanabe Pharma Co.; Takeda Pharmaceutical Co.; Daiichi Sankyo Co., Ltd.; Chugai Pharmaceutical Co.; Bristol-Myers Squibb K.K.; MSD K.K.; and Astellas Pharma Inc. HK, YS, and SY have no conflicts of interest to declare.
Ethical standards
We declare that this article has not been published and is not under consideration for publication elsewhere, and that there is no prior publication or the submission elsewhere of any part of the work. All the authors read and approved the final manuscript. This study was approved by the Ethics Committee for Clinical Investigation of the Fujita Health University School of Medicine (no. HM19-193) and the Yokkaichi Hazu Medical Center (no. 108) and was undertaken following the Principles of the Declaration of Helsinki amended during the 64th WMA General Assembly, Fortaleza, Brazil 2013.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Mizutani, S., Kodera, H., Sato, Y. et al. Clinical effectiveness of iguratimod based on real-world data of patients with rheumatoid arthritis. Clin Rheumatol 40, 123–132 (2021). https://doi.org/10.1007/s10067-020-05208-y
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10067-020-05208-y