Abstract
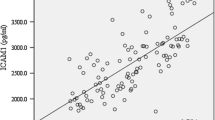
There are limited studies evaluating the fibrinogen levels in patients with migraine. It remains unknown whether the levels of the haematological marker of thromboembolism, d-dimer, and the levels of galectin-3, which plays an important role in inflammation as a proinflammatory mediator, change during the attacks in patients with migraine. The present study aims to compare galectin-3, fibrinogen and d-dimer levels in patients with migraine during the attacks and interictal periods, and to compare galectin-3, fibrinogen and d-dimer levels between patients with migraine and healthy controls to investigate the role of these parameters in the pathogenesis of migraine. Fifty-nine patients with migraine and 30 age-gender matched healthy control subjects were enrolled in the study. Blood galectin-3, fibrinogen and d-dimer levels were measured in patients with migraine. Patients with migraine had higher levels of galectin-3, fibrinogen and d-dimer compared to the healthy controls (p < 0.05). No statistically significant difference was found between galectin-3 and fibrinogen levels during the attacks and interictal period in the migraine group (p > 0.05). Migraine patients had higher d-dimer levels during the attacks compared to the patients in the interictal period in the migraine group (p = 0.05). In conclusion, we found increased levels of fibrinogen, d-dimer and galectin-3 in patients with migraine compared to the healthy control group. Furthermore, we showed increased galectin-3 levels in patients with migraine, and higher d-dimer levels during migraine attacks compared to the interictal periods for the first time. These findings may be associated with the hypercoagulability and neurogenic inflammation during migraine headaches.
Similar content being viewed by others
References
Ferrara M, Capozzi L, Bertocco F, Ferrara D, Russo R (2012) Thrombophilic gene mutations in children with migraine. Hematology 17:115–117. doi:10.1179/102453312X13221316478010
Martinez-Sanchez P, Martinez–Martinez M, Fuentes B, Cuesta MV, Cuellar-Gamboa L, Idrovo-Freire L, Fernandez-Dominguez J, Diez-Tejedor E (2011) Migraine and hypercoagulable states in ischemic stroke. Cephalalgia: Int J Headache 31:1609–1617. doi:10.1177/0333102411427599
Schurks M, Rist PM, Bigal ME, Buring JE, Lipton RB, Kurth T (2009) Migraine and cardiovascular disease: systematic review and meta-analysis. BMJ 339:b3914. doi:10.1136/bmj.b3914
Isobe C, Terayama Y (2010) A remarkable increase in total homocysteine concentrations in the CSF of migraine patients with aura. Headache 50:1561–1569. doi:10.1111/j.1526-4610.2010.01713.x
Termine C, Trotti R, Ondei P, Gamba G, Montani N, Gamba A, De Simone M, Marni E, Balottin U (2010) Mitral valve prolapse and abnormalities of haemostasis in children and adolescents with migraine with aura and other idiopathic headaches: a pilot study. Acta Neurol Scand 122:91–96. doi:10.1111/j.1600-0404.2009.01268.x
Walkowiak B, Kozubski W, Pawlowska Z, Prusinski A, Cierniewski CS (1989) Expression of fibrinogen receptors in platelets of migraine patients—correlation with platelet GPIIb content and plasma cholesterol. Thromb Haemost 61:419–422
Stoppa-Vaucher S, Dirlewanger MA, Meier CA, de Moerloose P, Reber G, Roux-Lombard P, Combescure C, Saudan S, Schwitzgebel VM (2012) Inflammatory and prothrombotic states in obese children of European descent. Obesity (Silver Spring) 20:1662–1668. doi:10.1038/oby.2012.85
Hirano K, Takashima S, Dougu N, Taguchi Y, Nukui T, Konishi H, Toyoda S, Kitajima I, Tanaka K (2012) Study of hemostatic biomarkers in acute ischemic stroke by clinical subtype. J Stroke Cerebrovasc Dis: Off J Natl Stroke Assoc 21:404–410. doi:10.1016/j.jstrokecerebrovasdis.2011.08.013
Kurth T, Ridker PM, Buring JE (2008) Migraine and biomarkers of cardiovascular disease in women. Cephalalgia: Int J Headache 28:49–56. doi:10.1111/j.1468-2982.2007.01467.x
Shin T (2013) The pleiotropic effects of galectin-3 in neuroinflammation: a review. Acta Histochem. doi:10.1016/j.acthis.2012.11.010
Fernandez GC, Ilarregui JM, Rubel CJ, Toscano MA, Gomez SA, Beigier Bompadre M, Isturiz MA, Rabinovich GA, Palermo MS (2005) Galectin-3 and soluble fibrinogen act in concert to modulate neutrophil activation and survival: involvement of alternative MAPK pathways. Glycobiology 15:519–527. doi:10.1093/glycob/cwi026
Olesen J (2005) The international classification of headache disorders, 2nd edition: application to practice. Funct Neurol 20:61–68
Takagi H, Manabe H, Kawai N, Goto S, Umemoto T (2009) Plasma fibrinogen and d-dimer concentrations are associated with the presence of abdominal aortic aneurysm: a systematic review and meta-analysis. Eur J Vasc Endovasc Surg: Off J Eur Soc Vasc Surg 38:273–277. doi:10.1016/j.ejvs.2009.05.013
de Almeida VV, Silva-Herdade A, Calado A, Rosario HS, Saldanha C (2012) Fibrinogen modulates leukocyte recruitment in vivo during the acute inflammatory response. Clin Hemorheol Microcirc. doi:10.3233/CH-121660
Davalos D, Akassoglou K (2012) Fibrinogen as a key regulator of inflammation in disease. Semin Immunopathol 34:43–62. doi:10.1007/s00281-011-0290-8
Farrell DH (2012) gamma’ Fibrinogen as a novel marker of thrombotic disease. Clin Chem Lab Med: CCLM/FESCC 50:1903–1909. doi:10.1515/cclm-2012-0005
Rudnicka AR, Mt-Isa S, Meade TW (2006) Associations of plasma fibrinogen and factor VII clotting activity with coronary heart disease and stroke: prospective cohort study from the screening phase of the Thrombosis Prevention Trial. J Thromb Haemostasis : JTH 4:2405–2410. doi:10.1111/j.1538-7836.2006.02221.x
Wannamethee SG, Whincup PH, Lennon L, Rumley A, Lowe GD (2012) Fibrin d-dimer, tissue-type plasminogen activator, von Willebrand factor, and risk of incident stroke in older men. Stroke J Cerebral Circ 43:1206–1211. doi:10.1161/STROKEAHA.111.636373
Tripodi A (2011) d-Dimer testing in laboratory practice. Clin Chem 57:1256–1262. doi:10.1373/clinchem.2011.166249
Zhou D, Yang PY, Zhou B, Rui YC (2007) Fibrin d-dimer fragments enhance inflammatory responses in macrophages: role in advancing atherosclerosis. Clin Exp Pharmacol Physiol 34:185–190. doi:10.1111/j.1440-1681.2007.04570.x
Bianchi A, Pitari G, Amenta V, Giuliano F, Gallina M, Costa R, Ferlito S (1996) Endothelial, haemostatic and haemorheological modifications in migraineurs. Artery 22:93–100
Yilmaz N, Aydin O, Yegin A, Tiltak A, Eren E (2011) Increased levels of total oxidant status and decreased activity of arylesterase in migraineurs. Clin Biochem 44:832–837. doi:10.1016/j.clinbiochem.2011.04.015
Walkowiak B, Kozubski W, Prusinski A, Cierniewski CS (1989) The increased contents of beta-thromboglobulin and fibrinogen in migraine platelets indicate an abnormality in thrombopoiesis. Headache 29:257
Wesley UV, Vemuganti R, Ayvaci ER, Dempsey RJ (2013) Galectin-3 enhances angiogenic and migratory potential of microglial cells via modulation of integrin linked kinase signaling. Brain Res 1496:1–9. doi:10.1016/j.brainres.2012.12.008
Satoh K, Niwa M, Binh NH, Nakashima M, Kobayashi K, Takamatsu M, Hara A (2011) Increase of galectin-3 expression in microglia by hyperthermia in delayed neuronal death of hippocampal CA1 following transient forebrain ischemia. Neurosci Lett 504:199–203. doi:10.1016/j.neulet.2011.09.015
Doverhag C, Hedtjarn M, Poirier F, Mallard C, Hagberg H, Karlsson A, Savman K (2010) Galectin-3 contributes to neonatal hypoxic-ischemic brain injury. Neurobiol Dis 38:36–46. doi:10.1016/j.nbd.2009.12.024
Yan YP, Lang BT, Vemuganti R, Dempsey RJ (2009) Galectin-3 mediates post-ischemic tissue remodeling. Brain Res 1288:116–124. doi:10.1016/j.brainres.2009.06.073
Jeon SB, Yoon HJ, Chang CY, Koh HS, Jeon SH, Park EJ (2010) Galectin-3 exerts cytokine-like regulatory actions through the JAK-STAT pathway. J Immunol 185:7037–7046. doi:10.4049/jimmunol.1000154
Williamson DJ, Hargreaves RJ (2001) Neurogenic inflammation in the context of migraine. Microsc Res Tech 53:167–178. doi:10.1002/jemt.1081
Chen HY, Sharma BB, Yu L, Zuberi R, Weng IC, Kawakami Y, Kawakami T, Hsu DK, Liu FT (2006) Role of galectin-3 in mast cell functions: galectin-3-deficient mast cells exhibit impaired mediator release and defective JNK expression. J Immunol 177:4991–4997
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Yucel, Y., Tanriverdi, H., Arıkanoglu, A. et al. Increased fibrinogen, d-dimer and galectin-3 levels in patients with migraine. Neurol Sci 35, 545–549 (2014). https://doi.org/10.1007/s10072-013-1542-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10072-013-1542-2