Abstract
Background
This phase I study was performed to determine the maximum tolerated dose (MTD), recommended dose (RD), and dose-limiting toxicities (DLTs) of oxaliplatin combined with preoperative chemoradiotherapy with S-1, oxaliplatin, and bevacizumab in locally advanced rectal cancer.
Methods
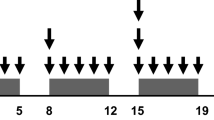
Eligible patients had a newly diagnosed clinical stage T1–4 N0–3 M0 rectal adenocarcinoma within 12 cm of the anal verge suitable for curative resection. Conformal radiation therapy was given (4 fields, 2 Gy daily fractions, 5 days/week, total dose 40 Gy) with concurrent S-1 (80 mg/m2/day orally, days 1–5, 8–12, 15–19, and 22–26), bevacizumab (90 min continuous intravenous infusion at 5 mg/kg, days 1 and 15), and oxaliplatin (120 min continuous intravenous infusion, days 1, 8, 15, and 22). The initial oxaliplatin dose (40 mg/m2/day) was gradually increased to determine the MTD and RD. Surgery was performed 6 weeks after completion of preoperative chemoradiotherapy.
Results
11 patients were enrolled. The MTD of oxaliplatin was considered to be 60 mg/m2, because three of five patients developed DLTs such as diarrhea and hives. The recommended dose of oxaliplatin was set at 50 mg/m2. Of the patients who received oxaliplatin at ≤ RD, 5 (83.3 %) had a clinical response [four pathological responses and one pathological complete response (Grade 3)].
Conclusions
With this new regimen, the MTD of oxaliplatin was 60 mg/m2, and the RD for phase II studies was 50 mg/m2. This new regimen appears to provide worthwhile outcomes for locally advanced rectal cancer and merits a phase II study.

Similar content being viewed by others
References
Enker WE, Thaler HT, Cranor ML et al (1995) Total mesorectal excision in the operative treatment of carcinoma of the rectum. J Am Coll Surg 181:335–346
Quirke P, Durdey P, Dixon MF et al (1986) Local recurrence of rectal adenocarcinoma due to inadequate surgical resection. Histopathological study of lateral tumour spread and surgical excision. Lancet 2:996–999
Van Gijn W, Marijnen CA, Naqtegaal ID et al (2011) Preoperative radiotherapy combined with total mesorectal excision for resectable rectal cancer: 12-year follow-up of the multicentre, randomised controlled TME trial. Lancet Oncol 12:575–582
Bosset JF, Collette L, Calais G et al (2006) Chemotherapy with preoperative radiotherapy in rectal carcinoma. N Engl J Med 355:1114–1123
Gerad JP, Conroy T, Bonnetain F et al (2006) Preoperative radiotherapy with or without concurrent fluorouracil and leucovorin in T3-4 rectal cancers: results of FFCD9203. J Clin Oncol 24:4620–4625
Sauer R, Becker H, Hohenberger W et al (2004) Preoperative versus postoperative chemoradiotherapy for rectal cancer. N Engl J Med 351:1731–1740
Gavioli M, Luppi G, Losi L et al (2005) Incidence and clinical impact of sterilized disease and minimal residual disease after preoperative radiochemotherapy for rectal cancer. Dis Colon Rectum 48:1851–1857
Stipa F, Chessin DB, Shia J et al (2006) A pathologic complete response of rectal cancer to preoperative combined-modality therapy results in improved oncological outcome compared with those who achieve no downstaging on the basis of preoperative endorectal ultrasonography. Ann Surg Oncol 13:1047–1053
Aschele C, Cionini L, Lonardi S et al (2011) Primary tumor response to preoperative chemoradiation with or without oxaliplatin in locally advanced rectal cancer: pathologic results of the STAR-01 randomized phase III trial. J Clin Oncol 29:2773–2780
Rullier A, Gourgou-Bourgade S, Jarlier M et al (2013) Predictive factors of positive circumferential resection margin after radiochemotherapy for rectal cancer: the French randomised trial ACCORD12/0405 PRODIGE 2. Eur J Cancer 49:82–89
Rödel C, Liersch T, Becker H, et al (2014) Preoperative chemoradiotherapy and postoperative chemotherapy with fluorouracil and oxaliplatin versus fluorouracil alone in locally advanced rectal cancer: initial results of the German CAO/ARO/AIO-04 randomised phase 3 trial. ASCO Annual Meeting
An X, Lin X, Wang FH et al (2013) Short term results of neoadjuvant chemoradiotherapy with fluoropyrimidine alone or in combination with oxaliplatin in locally advanced rectal cancer: a meta analysis. Eur J Cancer 49:843–851
Kennecke H, Berry S, Wong R et al (2012) Pre-operative bevacizumab, capecitabine, oxaliplatin and radiation among patients with locally advanced or low rectal cancer: a phase II trial. Eur J Cancer 48:37–45
Landry JC, Feng Y, Cohen SJ et al (2013) Phase 2 study of preoperative radiation with concurrent capecitabine, oxaliplatin, and bevacizumab followed by surgery and postoperative 5-fluorouracil, leucovorin, oxaliplatin (FOLFOX), and bevacizumab in patients with locally advanced rectal cancer: eCOG 3204. Cancer 119:1521–1527
Sato T, Kokuba Y, Koizumi W et al (2007) Phase I trial of neoadjuvant preoperative chemotherapy with S-1 and irinotecan plus radiation in patients with locally advanced rectal cancer. Int J Radiat Oncol Biol Phys 69:1442–1447
Wong SJ, Winter K, Meropol NJ et al (2012) Radiation Therapy Oncology Group 0247: a randomized Phase II study of neoadjuvant capecitabine and irinotecan or capecitabine and oxaliplatin with concurrent radiotherapy for patients with locally advanced rectal cancer. Int J Radiat Oncol Biol Phys 82:1367–1375
Hong YS, Park YS, Lim HY et al (2012) S-1 plus oxaliplatin versus capecitabine plus oxaliplatin for first-line treatment of patients with metastatic colorectal cancer: a randomised, non-inferiority phase 3 trial. Lancet Oncol 13:1125–1132
Acknowledgments
This study was supported by a grant-in-aid from a cooperative cancer research project between TAIHO Pharmaceutical Co., Ltd., Yakult Co., Ltd. and Chugai Co., Ltd..
Conflict of interest
Drs. Sato, Kurita, Iwata, Yoshikawa, Higashigima, Chikakio, Kashihara, Takasu, Mastumoto, Eto declare no conflict of interest or financial ties. Dr. Shimada declares the following conflict of interest: financial grants from a cooperative cancer research project between TAIHO Pharmaceutical Co., Ltd., Yakult Co., Ltd. and Chugai Co., Ltd..
Author information
Authors and Affiliations
Corresponding authors
About this article
Cite this article
Sato, H., Shimada, M., Kurita, N. et al. Phase I trial of neoadjuvant preoperative chemotherapy with S-1, oxaliplatin, and bevacizumab plus radiation in patients with locally advanced rectal cancer. Int J Clin Oncol 20, 543–548 (2015). https://doi.org/10.1007/s10147-014-0733-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10147-014-0733-z