Abstract
Background
Improving outcomes in locally advanced esophageal/GEJ squamous cell cancer (SCC) is an unmet need. We investigated the addition of oral metronomic chemotherapy (OMC) following definitive chemoradiotherapy (CRT).
Materials and methods
This was a randomized open-label integrated phase II/III study in patients with SCC of esophagus/GEJ following definitive CRT who had no radiologic evidence of progression, and no endoscopically detected disease. Randomization was 1:1 to OMC (celecoxib 200 mg twice daily and methotrexate 15 mg/m2 weekly) for 12 months or observation. The primary endpoint for the phase II portion was progression-free survival (PFS); secondary endpoints were overall survival (OS) and toxicity. P ≤ 0.2 for PFS was required to proceed to phase III.
Results
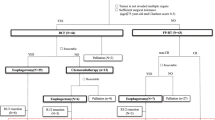
Between Jan 2016 and Dec 2019, we enrolled 151 patients for the phase II portion, 75 to OMC and 76 to observation. The tumor originated in the upper thoracic esophagus in 79% patients. Concurrent CRT consisted of median 63 Gy in a median of 35 fractions; concurrent chemotherapy was weekly paclitaxel + carboplatin in 91%. OMC was started at a median of 2.6 months (IQR 2.3–2.8) from CRT completion. Grade 3 or higher toxicities occurred in 18 patients (24%) in the OMC arm and 9 (12%) in the observation arm; P = 0.071. Median PFS was 25 months (95% CI, 17–58) in the OMC arm and was not attained [NA] (95% CI, 25–NA) in the observation arm; HR, 1.51, 95% CI, 1–2; P = 0.073. Median OS was 36 months (95% CI, 23–NA) in the OMC arm, and not attained (95% CI, NA–NA) in the observation arm; HR, 1.77; 95% CI, 1–2.9; P = 0.023.
Conclusion
Oral metronomic methotrexate and celecoxib in patients who have not progressed radiologically and have no endoscopic evidence of disease following radical CRT for locally advanced esophageal/GEJ SCC does not improve outcomes and may lower survival. [Funded by the TMC-Research Administration Council (TRAC); CHROME study (CHemoRadiotherapy followed by Oral Metronomic therapy in Esophageal cancer); ctri.nic.in number: CTRI/2015/09/006204].
Trial registration number
CTRI/2015/09/006204.





Similar content being viewed by others
Data availability
The data collected for the study, including deidentified individual participant data and a data dictionary defining each field in the set, will be made available to others, on reasonable request. Additionally, the study protocol, statistical analysis plan and the informed consent form will be made available, on reasonable request. These data will be made available, on reasonable request, starting from the time of publication. The data are available on reasonable request from Dr. Vanita Noronha (vanita.noronha@gmail.com) or Dr. Kumar Prabhash (kumarprabhashtmh@gmail.com). Data will be made available, on reasonable request for additional analyses or other scientific purposes, with investigator support and with a signed data access agreement.
References
Rustgi A, El-Serag HB. Esophageal carcinoma. N Engl J Med. 2015;372(15):1472–3.
Rackley T, Leong T, Foo M, Crosby T. Definitive chemoradiotherapy for oesophageal cancer—a promising start on an exciting journey. Clin Oncol (R Coll Radiol). 2014;26(9):533–40.
Herskovic A, Martz K, al-Sarraf M, et al. Combined chemotherapy and radiotherapy compared with radiotherapy alone in patients with cancer of the esophagus. N Engl J Med. 1992;326(24):1593–8.
al-Sarraf M, Martz K, Herskovic A, et al. Progress report of combined chemoradiotherapy versus radiotherapy alone in patients with esophageal cancer: an intergroup study. J Clin Oncol. 1997;15(1):277–84.
Minsky BD, Pajak TF, Ginsberg RJ, et al. INT 0123 (Radiation Therapy Oncology Group 94–05) phase III trial of combined-modality therapy for esophageal cancer: high-dose versus standard-dose radiation therapy. J Clin Oncol. 2002;20(5):1167–74.
Hulshof MCCM, Geijsen ED, Rozema T, et al. Randomized study on dose escalation in definitive chemoradiation for patients with locally advanced esophageal cancer (ARTDECO Study). J Clin Oncol. 2021;39:2816–24.
Minsky BD, Neuberg D, Kelsen DP, et al. Final report of Intergroup Trial 0122 (ECOG PE-289, RTOG 90–12): Phase II trial of neoadjuvant chemotherapy plus concurrent chemotherapy and high-dose radiation for squamous cell carcinoma of the esophagus. Int J Radiat Oncol Biol Phys. 1999;43(3):517–23.
Crosby T, Hurt CN, Falk S, et al. Chemoradiotherapy with or without cetuximab in patients with oesophageal cancer (SCOPE1): a multicentre, phase 2/3 randomised trial. Lancet Oncol. 2013;14(7):627–37.
Suntharalingam M, Winter K, Ilson D, et al. Effect of the addition of cetuximab to paclitaxel, cisplatin, and radiation therapy for patients with esophageal cancer: the NRG oncology RTOG 0436 phase 3 randomized clinical trial [published correction appears in JAMA Oncol. 2017;3:1589]. JAMA Oncol. 2017;3(11):1520–8.
Wu SX, Li XY, Xu HY, et al. Effect of consolidation chemotherapy following definitive chemoradiotherapy in patients with esophageal squamous cell cancer. Sci Rep. 2017;7(1):16870.
Kelly RJ. Emerging multimodality approaches to treat localized esophageal cancer. J Natl Compr Canc Netw. 2019;17(8):1009–14.
Pennathur A, Gibson MK, Jobe BA, Luketich JD. Oesophageal carcinoma. Lancet. 2013;381(9864):400–12.
Samarasam I. Esophageal cancer in India: Current status and future perspectives. Int J Adv Med Health Res. 2017;4(1):5–10.
Integrated genomic characterization of oesophageal carcinoma. Nature. 2017;541(7636):169–75.
Noronha V, Krishna MV, Patil V, Joshi A, Banavali SD, Prabhash K. Metronomic therapy: chemotherapy revisited. Indian J Cancer. 2013;50(2):142–8.
Patil VM, Noronha V, Joshi A, et al. Phase I/II study of palliative triple metronomic chemotherapy in platinum-refractory/early-failure oral cancer. J Clin Oncol. 2019;37(32):3032–41.
Patil V, Noronha V, Dhumal SB, et al. Low-cost OMC versus intravenous cisplatin in patients with recurrent, metastatic, inoperable head and neck carcinoma: an open-label, parallel-group, non-inferiority, randomised, phase 3 trial. Lancet Glob Health. 2020;8(9):e1213–22.
Pai PS, Vaidya AD, Prabhash K, Banavali SD. Oral metronomic scheduling of anticancer therapy-based treatment compared to existing standard of care in locally advanced oral squamous cell cancers: a matched-pair analysis. Indian J Cancer. 2013;50(2):135–41.
Chen JH, Huang WY, Ho CL, Chao TY, Lee JC. Evaluation of oral tegafur-uracil as metronomic therapy following concurrent chemoradiotherapy in patients with non-distant metastatic TNM stage IV nasopharyngeal carcinoma. Head Neck. 2019;41(11):3775–82.
Noronha V, Patil VM, Joshi A, Chougule A, Banavali S, Prabhash K. Potential role of metronomic chemotherapy in the treatment of esophageal and gastroesophageal cancer. Cancer Lett. 2017;400:267–75.
Noronha V, Prabhash K, Joshi A, et al. Clinical outcome in definitive concurrent chemoradiation with weekly paclitaxel and carboplatin for locally advanced esophageal and junctional cancer. Oncol Res. 2016;23(4):183–95.
Chakraborty S. A step-wise guide to performing survival analysis. Cancer Res Stat Treat. 2018;1(1):41–5.
Dessai S, Simha V, Patil V. Stepwise cox regression analysis in SPSS. Cancer Res Stat Treat. 2018;1(2):167–70.
Dessai S, Patil V. Testing and interpreting assumptions of COX regression analysis. Cancer Res Stat Treat. 2019;2(1):108–11.
Chen Y, Ye J, Zhu Z, et al. Comparing paclitaxel plus fluorouracil versus cisplatin plus fluorouracil in chemoradiotherapy for locally advanced esophageal squamous cell cancer: a randomized, multicenter, phase III clinical trial. J Clin Oncol. 2019;37(20):1695–703.
Yang GY, Wagner TD, Jobe BA, Thomas CR. The role of positron emission tomography in esophageal cancer. Gastrointest Cancer Res. 2008;2(1):3–9.
Dora TK, Deshmukh J, Chatterjee A, et al. Conformal radiation therapy versus volumetric arc therapy in high dose concurrent chemoradiotherapy for carcinoma esophagus: a retrospective analysis. Cancer Res Stat Treat. 2021;4:456–65.
Talapatra K. Radiation therapy in esophageal cancer: shifting paradigms. Cancer Res Stat Treat. 2021;4:538–40.
Lu Z, Fang Y, Liu C, Zhang X, Xin X, He Y, Cao Y, Jiao X, Sun T, Pang Y, Wang Y, Zhou J, Qi C, Gong J, Wang X, Li J, Tang L, Shen L. Early interdisciplinary supportive care in patients with previously untreated metastatic esophagogastric cancer: a phase III randomized controlled trial. J Clin Oncol. 2021;39(7):748–56.
Antonia SJ, Villegas A, Daniel D, et al. Overall survival with durvalumab after chemoradiotherapy in stage III NSCLC. N Engl J Med. 2018;379:2342–50.
Lee NY, Ferris RL, Psyrri A, et al. Avelumab plus standard-of-care chemoradiotherapy versus chemoradiotherapy alone in patients with locally advanced squamous cell carcinoma of the head and neck: a randomised, double-blind, placebo-controlled, multicentre, phase 3 trial. Lancet Oncol. 2021;22(4):450–62.
Kelly RJ, Ajani JA, Kuzdzal J, et al. CheckMate 577 investigators. Adjuvant nivolumab in resected esophageal or gastroesophageal junction cancer. N Engl J Med. 2021;384:1191–203.
Smyth EC, Knoedler MK, Nilsson M, et al. EORTC 1707 VESTIGE: Adjuvant immunotherapy in patients with resected gastric cancer following preoperative chemotherapy with high risk for recurrence (ypN+ and/or R1): An open-label randomized controlled phase II study. J Clin Oncol. 2020;38:no.4_suppl.
Available online at: https://clinicaltrials.gov/ct2/show/NCT02520453. Accessed 16 Nov 2020.
Hsu C-H, Guo J-C, Huang T-C, et al. Phase II study of pembrolizumab after chemoradiotherapy (CRT) as adjuvant therapy for locally advanced esophageal squamous cell carcinoma (LA-ESCC) patients at high risk of recurrence following preoperative CRT plus surgery. J Clin Oncol 2021; 39:no. 3_suppl.
Le Chevalier T. Adjuvant chemotherapy for resectable non-small-cell lung cancer: where is it going? Ann Oncol. 2010;21(Suppl 7):vii196-198.
Solomon SD, McMurray JJ, Pfeffer MA, et al. Adenoma Prevention with Celecoxib (APC) Study Investigators. Cardiovascular risk associated with celecoxib in a clinical trial for colorectal adenoma prevention. N Engl J Med. 2005;352:1071–80.
Wang X, Palaskas NL, Yusuf SW, et al. Incidence and onset of severe cardiac events after radiotherapy for esophageal cancer. J Thorac Oncol. 2020;15:1682–90.
Sasaki Y, Kato K. Chemoradiotherapy for esophageal squamous cell cancer. Jpn J Clin Oncol. 2016;46:805–10.
Bisogno G, De Salvo GL, Bergeron C, et al. Vinorelbine and continuous low-dose cyclophosphamide as maintenance chemotherapy in patients with high-risk rhabdomyosarcoma (RMS 2005): a multicentre, open-label, randomised, phase 3 trial. Lancet Oncol. 2019;20:1566–75.
Chen YP, Liu X, Zhou Q, et al. Metronomic capecitabine as adjuvant therapy in locoregionally advanced nasopharyngeal carcinoma: a multicentre, open-label, parallel-group, randomised, controlled, phase 3 trial. Lancet. 2021;398(10297):303–13.
Acknowledgements
We greatly appreciate the statistical analysis help provided by Ms. Smruti Mokal, Mr. Sanjay Talole, and Ms. Rohini Hawaldar.
Funding
This work was supported by the Tata Memorial Center Research Administration Council (TRAC) (grant number not applicable).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Ethical Statement
The study was approved by the Institutional Ethics Committee (IEC) of the Tata Memorial Center (Project number 155, approved by IEC-III).
Conflict of interest
Unrelated to the current study, Dr. Vanita Noronha has received research funding from Amgen, Sanofi India Ltd., Dr. Reddy’s Laboratories Inc., Intas Pharmaceuticals and Astra Zeneca Pharma India Ltd. (all research grants paid to the institution). Unrelated to the current study, Dr. Kumar Prabhash has received research funding from Dr. Reddy’s Laboratories Inc., Fresenius Kabi India Pvt. Ltd., Alkem Laboratories, Natco Pharma Ltd., BDR Pharmaceuticals Intl. Pvt. Ltd, and Roche Holding AG (all research grants paid to the institution). All the other authors declared no conflicts of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Noronha, V., Patil, V.M., Menon, N.S. et al. Oral metronomic chemotherapy after definitive chemoradiation in esophageal squamous cell carcinoma: a randomized clinical trial. Esophagus 19, 670–682 (2022). https://doi.org/10.1007/s10388-022-00923-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10388-022-00923-8