Abstract
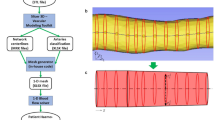
Arterial hemodynamics is markedly characterized by the presence of helical flow patterns. Previous observations suggest that arterial helical blood flow is of physiological significance, and that its quantitative analysis holds promise for clinical applications. In particular, it has been reported that distinguishable helical flow patterns are potentially atheroprotective in the carotid bifurcation as they suppress flow disturbances. In this context, there is a knowledge gap about the physiological significance of helical flow in coronary arteries, a prominent site of atherosclerotic plaque formation. This study aimed at the quantitative assessment of helical blood flow in coronary arteries, and to investigate its possible associations with vascular geometry and with atherogenic wall shear stress (WSS) phenotypes in a representative sample of 30 swine coronary arteries. This study demonstrates that in coronary arteries: (1) the hemodynamics is characterized by counter-rotating bi-helical flow structures; (2) unfavorable conditions of WSS are strongly and inversely associated with helicity intensity (r = − 0.91; p < 0.001), suggesting an atheroprotective role for helical flow in the coronary tree; (3) vascular torsion dictates helical flow features (r = 0.64; p < 0.001). The findings of this work support future studies on the role of helical flow in atherogenesis in coronary arteries.







Similar content being viewed by others
References
Aristokleous, N., I. Seimenis, G. C. Georgiou, A. Nicolaides, and A. S. Anayiotos. The effect of head rotation on the geometry and hemodynamics of healthy vertebral arteries. Ann. Biomed. Eng. 43(6):1287–1297, 2015.
Arnold, R., M. Neu, D. Hirtler, C. Gimpel, M. Markl, and J. Geiger. Magnetic resonance imaging 4-D flow-based analysis of aortic hemodynamics in Turner syndrome. Pediatr. Radiol. 47:382–390, 2017.
Bachler, P., N. Pinochet, J. Sotelo, G. Crelier, P. Irarrazaval, C. Tejos, and S. Uribe. Assessment of normal flow patterns in the pulmonary circulation by using 4D magnetic resonance velocity mapping. Magn. Reson. Imaging 31:178–188, 2013.
Bammer, R., T. A. Hope, M. Aksoy, and M. T. Alley. Time-resolved 3D quantitative flow MRI of the major intracranial vessels: initial experience and comparative evaluation at 1.5 T and 3.0 T in combination with parallel imaging. Magn. Reson. Med. 57(1):127–140, 2007.
Bogren, H. G., and M. H. Buonocore. Blood flow measurements in the aorta and major arteries with MR velocity mapping. J. Magn. Reson. Imaging 4(2):119–130, 1994.
Bogren, H. G., M. H. Buonocore, and R. J. Valente. Four-dimensional magnetic resonance velocity mapping of blood flow patterns in the aorta in patients with atherosclerotic coronary artery disease compared to age-matched normal subjects. J. Magn. Reson. Imaging 19:417–427, 2004.
Bogren, H. G., R. H. Mohiaddin, P. J. Kilner, L. I. Jimenez-Borreguero, G. Z. Yang, and D. N. Firmin. Blood flow patterns in the thoracic aorta studied with three-directional MR velocity mapping: the effects of age and coronary artery disease. J. Magn. Reson. Imaging 7:784–793, 1997.
Bürk, J., P. Blanke, Z. Stankovic, A. Barker, M. Russe, J. Geiger, A. Frydrychowicz, M. Langer, and M. Markl. Evaluation of 3D blood flow patterns and wall shear stress in the normal and dilated thoracic aorta using flow-sensitive 4D CMR. J. Cardiovasc. Magn. Reson. 14(1):84, 2012.
Chatzizisis, Y. S., M. Jonas, A. U. Coskun, R. Beigel, B. V. Stone, C. Maynard, R. G. Gerrity, W. Daley, C. Rogers, E. R. Edelman, C. L. Feldman, and P. H. Stone. Prediction of the localization of high-risk coronary atherosclerotic plaques on the basis of low endothelial shear stress: an intravascular ultrasound and histopathology natural history study. Circulation 117:993–1002, 2008.
Chiastra, C., D. Gallo, P. Tasso, F. Iannaccone, F. Migliavacca, J. J. Wentzel, and U. Morbiducci. Healthy and diseased coronary bifurcation geometries influence near-wall and intravascular flow: a computational exploration of the hemodynamic risk. J. Biomech. 58:79–88, 2017.
Condemi, F., S. Campisi, M. Viallon, T. Troalen, G. Xuexin, A. J. Barker, M. Markl, P. Croisille, O. Trabelsi, C. Cavinato, A. Duprey, and S. Avril. Fluid- and biomechanical analysis of ascending thoracic aorta aneurysm with concomitant aortic insufficiency. Ann. Biomed. Eng. 45(12):2921–2932, 2017.
Fox, B., K. James, B. Morgan, and W. A. Seed. Distribution of fatty and fibrous plaques in young human coronary arteries. Atherosclerosis 41:337–347, 1982.
Fox, B., and W. A. Seed. Location of early atheroma in the human coronary arteries. J. Biomech. Eng. 103:208–212, 1981.
François, C. J., S. Srinivasan, M. L. Schiebler, S. B. Reeder, E. Niespodzany, B. R. Landgraf, O. Wieben, and A. Frydrychowicz. 4D cardiovascular magnetic resonance velocity mapping of alterations of right heart flow patterns and main pulmonary artery hemodynamics in tetralogy of Fallot. J. Cardiovasc. Magn. Reson. 14:16, 2012.
Frazin, L. J., G. Lanza, M. Vonesh, F. Khasho, C. Spitzzeri, S. McGee, D. Mehlman, K. B. Chandran, J. Talano, and D. McPherson. Functional chiral asymmetry in descending thoracic aorta. Circulation 82(6):1985–1994, 1990.
Frydrychowicz, A., A. Berger, A. Munoz Del Rio, M. F. Russe, J. Bock, A. Harloff, and M. Markl. Interdependencies of aortic arch secondary flow patterns, geometry, and age analysed by 4-dimensional phase contrast magnetic resonance imaging at 3 Tesla. Eur. Radiol. 22(5):1122–1130, 2012.
Frydrychowicz, A., J. T. Winterer, M. Zaitsev, B. Jung, J. Hennig, M. Langer, and M. Markl. Visualization of iliac and proximal femoral artery hemodynamics using time-resolved 3D phase contrast MRI at 3T. J. Magn. Reson. Imaging 25:1085–1092, 2007.
Gallo, D., P. B. Bijari, U. Morbiducci, Y. Qiao, Y. Xie, M. Etesami, D. Haabets, E. G. Lakatta, B. A. Wasserman, and D. A. Steinman. Segment-specific associations between local haemodynamic and imaging markers of early atherosclerosis at the carotid artery: an in vivo human study. J. R. Soc. Interface 15:20180352, 2018.
Gallo, D., D. A. Steinman, P. B. Bijari, and U. Morbiducci. Helical flow in carotid bifurcation as surrogate marker of exposure to disturbed shear. J. Biomech. 45:2398–2404, 2012.
Gallo, D., D. A. Steinman, and U. Morbiducci. An insight into the mechanistic role of the common carotid artery on the hemodynamics at the carotid bifurcation. Ann. Biomed. Eng. 43:68, 2015.
Gallo, D., D. A. Steinman, and U. Morbiducci. Insights into the co-localization of magnitude-based versus direction-based indicators of disturbed shear at the carotid bifurcation. J. Biomech. 49(12):2413–2419, 2016.
Garcia, J., A. J. Barker, J. D. Collins, J. C. Carr, and M. Markl. Volumetric quantification of absolute local normalized helicity in patients with bicuspid aortic valve and aortic dilatation. Magn. Reson. Med. 78(2):689–701, 2017.
Geiger, J., M. Markl, L. Herzer, D. Hirtler, F. Loeffelbein, B. Stiller, M. Langer, and R. Arnold. Aortic flow patterns in patients with Marfan syndrome assessed by flow-sensitive four-dimensional MRI. J. Magn. Reson. Imaging 35:594–600, 2012.
Hansen, K. L., H. Moller-Sorensen, J. Kjaergaard, M. B. Jensen, J. T. Lund, M. M. Pedersen, T. Lange, J. A. Jensen, and M. B. Nielsen. Analysis of systolic backflow and secondary helical blood flow in the ascending aorta using vector flow imaging. Ultrasound Med. Biol. 42:899–908, 2016.
Himburg, H. A., D. M. Grzybowski, A. Hazel, J. A. LaMack, X. M. Li, and M. H. Friedman. Spatial comparison between wall shear stress measures and porcine arterial endothelial permeability. Am. J. Physiol. Heart Circ. Physiol. 286:H1916–H1922, 2004.
Hope, T. A., and R. J. Herfkens. Imaging of the thoracic aorta with time-resolved three-dimensional phase-contrast MRI: a review. Semin. Thorac. Cardiovasc. Surg. 20(4):358–364, 2008.
Hope, M. D., T. A. Hope, S. E. Crook, K. G. Ordovas, T. H. Urbania, M. T. Alley, and C. B. Higgins. 4D flow CMR in assessment of valve-related ascending aortic disease. JACC Cardiovasc. Imaging 4(7):781–787, 2011.
Houston, J. G., S. J. Gandy, J. Milne, C. Dick, J. F. Belch, and P. A. Stonebridge. Spiral laminar flow in the abdominal aorta: a predictor of renal impairment deterioration in patients with renal artery stenosis? Nephrol. Dial. Transplant. 19:1786–1791, 2004.
Houston, J. G., S. J. Gandy, D. G. Sheppard, J. B. Dick, J. J. Belch, and P. A. Stonebridge. Two-dimensional flow quantitative MRI of aortic arch blood flow patterns: effect of age, sex, and presence of carotid atheromatous disease on prevalence of spiral blood flow. J. Magn. Reson. Imaging 18:169–174, 2003.
Huo, Y., and G. S. Kassab. Intraspecific scaling laws of vascular trees. J. R. Soc. Interface 9(66):190–200, 2012.
Karino, T. Microscopic structure of disturbed flows in the arterial and venous systems, and its implication in the localization of vascular diseases. Int. Angiol. 5:297–313, 1986.
Kilner, P. J., G. Z. Yang, R. H. Mohiaddin, D. N. Firmin, and D. B. Longmore. Helical and retrograde secondary flow patterns in the aortic arch studied by three-directional magnetic resonance velocity mapping. Circulation 88:2235–2247, 1993.
Knobloch, V., P. Boesige, and S. Kozerke. Sparsity transform k-t principal component analysis for accelerating cine three-dimensional flow measurements. Magn. Reson. Med. 70:53–63, 2013.
Ku, D. N., and D. P. Giddens. Laser Doppler anemometer measurements of pulsatile flow in a model carotid bifurcation. J. Biomech. 20(4):407–421, 1987.
Ku, D. N., D. P. Giddens, D. J. Phillips, and D. E. Strandness, Jr. Hemodynamics of the normal human carotid bifurcation: in vitro and in vivo studies. Ultrasound Med. Biol. 11(1):13–26, 1985.
Liu, X., Y. Fan, and X. Deng. Effect of spiral flow on the transport of oxygen in the aorta: a numerical study. Ann. Biomed. Eng. 38(3):917–926, 2010.
Liu, X., F. Pu, Y. Fan, X. Deng, D. Li, and S. Li. A numerical study on the flow of blood and the transport of LDL in the human aorta: the physiological significance of the helical flow in the aortic arch. Am. J. Physiol. Heart Circ. Physiol. 297(1):H163–H170, 2009.
Liu, X., A. Sun, Y. Fan, and X. Deng. Physiological significance of helical flow in the arterial system and its potential clinical applications. Ann. Biomed. Eng. 43(1):3–15, 2015.
Malek, A. M., S. L. Alper, and S. Izumo. Hemodynamic shear stress and its role in atherosclerosis. JAMA 282(21):2035–2042, 1999.
Malvè, M., A. García, J. Ohayon, and M. A. Martínez. Unsteady blood flow and mass transfer of a human left coronary artery bifurcation: FSI vs CFD. Int. Commun. Heat Mass Transf. 39:745–751, 2012.
Markl, M., M. T. Draney, M. D. Hope, J. M. Levin, F. P. Chan, M. T. Alley, N. J. Pelc, and R. J. Herfkens. Time-resolved 3-dimensional velocity mapping in the thoracic aorta: visualization of 3-directional blood flow patterns in healthy volunteers and patients. J. Comput. Assist. Tomogr. 28:459–468, 2005.
Markl, M., F. Wegent, T. Zech, S. Bauer, C. Strecker, M. Schumacher, C. Weiller, J. Hennig, and A. Harloff. In vivo wall shear stress distribution in the carotid artery: effect of bifurcation geometry, internal carotid artery stenosis, and recanalization therapy. Circ. Cardiovasc. Imaging 3:647–655, 2010.
Meckel, S., L. Leitner, L. H. Bonati, F. Santini, T. Schubert, A. F. Stalder, P. Lyrer, M. Markl, and S. G. Wetzel. Intracranial artery velocity measurement using 4D PC MRI at 3 T: comparison with transcranial ultrasound techniques and 2D PC MRI. Neuroradiology 55(4):389–398, 2013.
Moffatt, H. K., and A. Tsinober. Helicity in laminar and turbulent flow. Ann. Rev. Fluid Mech. 24:281–312, 1992.
Mohamied, Y., S. J. Sherwin, and P. D. Weinberg. Understanding the fluid mechanics behind transverse wall shear stress. J. Biomech. 50:102–109, 2017.
Morbiducci, U., D. Gallo, S. Cristofanelli, R. Ponzini, M. A. Deriu, G. Rizzo, and D. A. Steinman. A rational approach to defining principal axes of multidirectional wall shear stress in realistic vascular geometries, with application to the study of the influence of helical flow on wall shear stress directionality in aorta. J. Biomech. 48(6):899–906, 2015.
Morbiducci, U., A. M. Kok, B. R. Kwak, P. H. Stone, D. A. Steinman, and J. J. Wentzel. Atherosclerosis at arterial bifurcations: evidence for the role of haemodynamics and geometry. Thromb. Haemost. 115(3):484–492, 2016.
Morbiducci, U., R. Ponzini, D. Gallo, C. Bignardi, and G. Rizzo. Inflow boundary conditions for image-based computational hemodynamics: impact of idealized versus measured velocity profiles in the human aorta. J. Biomech. 46:102–109, 2013.
Morbiducci, U., R. Ponzini, M. Grigioni, and A. Redaelli. Helical flow as fluid dynamic signature for atherogenesis in aortocoronary bypass. A numeric study. J. Biomech. 40:519–534, 2007.
Morbiducci, U., R. Ponzini, G. Rizzo, M. Cadioli, A. Esposito, F. De Cobelli, A. Del Maschio, F. M. Montevecchi, and A. Redaelli. In vivo quantification of helical blood flow in human aorta by time-resolved three-dimensional cine phase contrast MRI. Ann. Biomed. Eng. 37:516–531, 2009.
Morbiducci, U., R. Ponzini, G. Rizzo, M. Cadioli, A. Esposito, F. M. Montevecchi, and A. Redaelli. Mechanistic insight into the physiological relevance of helical blood flow in the human aorta. An in vivo study. Biomech. Model. Mechanobiol. 10:339–355, 2011.
Nakashima, T., Y. Iwanaga, and Y. Nakaura. Pathologic study of hypertensive heart. Acta Pathol. Jpn. 14(1):129–141, 1964.
Nakashima, T., and T. Tashiro. Early morphologic stage of human coronary atherosclerosis. Kurume Med. J. 15(4):235–242, 1968.
National Research Council (US). Committee for the Update of the Guide for Care and Use of Laboratory Animals, Guide for the Care and Use of Laboratory Animals (8th ed.). Washington, DC: National Academies Press (US), 2011.
Peiffer, V., S. J. Sherwin, and P. D. Weinberg. Computation in the rabbit aorta of a new metric—the transverse wall shear stress—to quantify the multidirectional character of disturbed blood flow. J. Biomech. 46(15):2651–2658, 2013.
Pirola, S., O. A. Jarral, D. P. O’Regan, G. Asimakopoulos, J. R. Anderson, J. R. Pepper, T. Athanasiou, and X. Y. Xu. Computational study of aortic hemodynamics for patients with an abnormal aortic valve: the importance of secondary flow at the ascending aorta inlet. APL Bioeng. 2:026101, 2018.
Ponzini, R., C. Vergara, A. Redaelli, and A. Veneziani. Reliable CFD-based estimation of flow rate in haemodynamics measures. Ultrasound Med. Biol. 32(10):1545–1555, 2006.
Sabbah, H. N., F. J. Walburn, and P. D. Stein. Patterns of flow in the left coronary artery. J. Biomech. Eng. 106(3):272–279, 1984.
Schaap, M., L. Neefjes, C. Metz, A. van der Giessen, A. Weustink, N. Mollet, J. J. Wentzel, T. W. van Walsum, and W. Niessen. Coronary lumen segmentation using graph cuts and robust kernel regression. Inf. Process. Med. Imaging 21:528–539, 2009.
Schäfer, M., A. J. Barker, V. Kheyfets, K. R. Stenmark, J. Crapo, M. E. Yeager, U. Truong, J. K. Buckner, B. E. Fenster, and K. S. Hunter. Helicity and vorticity of pulmonary arterial flow in patients with pulmonary hypertension: quantitative analysis of flow formations. J. Am. Heart Assoc. 6(12):e007010, 2017.
Siasos, G., J. D. Sara, M. Zaromytidou, K. H. Park, A. U. Coskun, L. O. Lerman, E. Oikonomou, C. C. Maynard, D. Fotiadis, K. Stefanou, M. Papafaklis, L. Michalis, C. Feldman, A. Lerman, and P. H. Stone. Local low shear stress and endothelial dysfunction in patients with nonobstructive coronary atherosclerosis. J. Am. Coll. Cardiol. 71:2092–2102, 2018.
Siebes, M., B. J. Verhoeff, M. Meuwissen, R. J. de Winter, A. E. Spaan, and J. J. Piek. Single-wire pressure and flow velocity measurement to quantify coronary stenosis hemodynamics and effects of percutaneous interventions. Circulation 109:756–762, 2004.
Sigfridsson, A., S. Petersson, C. J. Carlhäll, and T. Ebbers. Four-dimensional flow MRI using spiral acquisition. Magn. Reson. Med. 68:1065–1073, 2012.
Stone, P. H., S. Saito, S. Takahashi, Y. Makita, S. Nakamura, T. Kawasaki, A. Takahashi, T. Katsuki, S. Nakamura, A. Namiki, A. Hirohata, T. Matsumura, S. Yamazaki, H. Yokoi, S. Tanaka, S. Otsuji, F. Yoshimachi, J. Honye, D. Harwood, M. Reitman, A. U. Coskun, M. I. Papafaklis, and C. L. Feldman. Prediction of progression of coronary artery disease and clinical outcomes using vascular profiling of endothelial shear stress and arterial plaque characteristics: The prediction study. Circulation 126:172–181, 2012.
Stonebridge, P. A., P. R. Hoskins, P. L. Allan, and J. F. Belch. Spiral laminar flow in vivo. Clin. Sci. (Lond) 91(1):17–21, 1996.
Stonebridge, P. A., S. A. Suttie, R. Ross, and J. Dick. Spiral laminar flow: a survey of a three-dimensional arterial flow pattern in a group of volunteers. Eur. J. Vasc. Endovasc. Surg. 52(5):674–680, 2016.
Theodorakakos, A., M. Gavaises, A. Andriotis, A. Zifan, P. Liatsis, I. Pantos, E. P. Efstathopoulos, and D. Katritsis. Simulation of cardiac motion on non-Newtonian, pulsating flow development in the human left anterior descending coronary artery. Phys. Med. Biol. 53:4875–4892, 2008.
Torii, R., J. Keegan, N. B. Wood, A. W. Dowsey, A. D. Hughes, G. Z. Yang, D. N. Firmin, S. A. Thom, and X. Y. Xu. MR image-based geometric and hemodynamic investigation of the right coronary artery with dynamic vessel motion. Ann. Biomed. Eng. 38:2606–2620, 2010.
Vorobtsova, N., C. Chiastra, M. A. Stremler, D. C. Sane, F. Migliavacca, and P. Vlachos. Effects of vessel tortuosity on coronary hemodynamics: an idealized and patient-specific computational study. Ann. Biomed. Eng. 44(7):2228–2239, 2016.
Wentzel, J. J., Y. S. Chatzizisis, F. J. Gijsen, G. D. Giannoglou, C. L. Feldman, and P. H. Stone. Endothelial shear stress in the evolution of coronary atherosclerotic plaque and vascular remodelling: current understanding and remaining questions. Cardiovasc. Res. 96(2):234–243, 2012.
Wetzel, S., S. Meckel, A. Frydrychowicz, L. Bonati, E. W. Radue, K. Scheffler, J. Hennig, and M. Markl. In vivo assessment and visualization of intracranial arterial hemodynamics with flow-sensitized 4D MR imaging at 3T. AJNR Am. J. Neuroradiol. 28(3):433–438, 2007.
Winkel, L. C., A. Hoogendoorn, R. Xing, J. J. Wentzel, and K. Van der Heiden. Animal models of surgically manipulated flow velocities to study shear stress-induced atherosclerosis. Atherosclerosis 241:100–110, 2015.
Zeng, D., Z. Ding, M. H. Friedman, and C. R. Ethier. Effects of cardiac motion on right coronary artery hemodynamics. Ann. Biomed. Eng. 31:420–429, 2003.
Conflict of interest
All authors declare that they have no financial and personal relationships with other people or organizations that could have inappropriately influenced the submitted work.
Author information
Authors and Affiliations
Corresponding author
Additional information
Associate Editor Andreas Anayiotos oversaw the review of this article.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
De Nisco, G., Kok, A.M., Chiastra, C. et al. The Atheroprotective Nature of Helical Flow in Coronary Arteries. Ann Biomed Eng 47, 425–438 (2019). https://doi.org/10.1007/s10439-018-02169-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10439-018-02169-x