Abstract
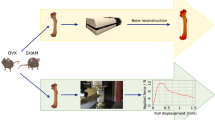
A moderate radiation dose, in vivo µCT scanning protocol was developed and validated for long-term monitoring of multiple skeletal sites (femur, tibia, vertebra) in mice. A customized, 3D printed mouse holder was designed and utilized to minimize error associated with animal repositioning, resulting in good to excellent reproducibility in most cortical and trabecular bone microarchitecture and density parameters except for connectivity density. Repeated in vivo µCT scans of mice were performed at the right distal femur and the 4th lumbar vertebra every 3 weeks until euthanized at 9 weeks after the baseline scan. Comparing to the non-radiated counterparts, no radiation effect was found on trabecular bone volume fraction, osteoblast and osteoblast number/surface, or bone formation rate at any skeletal site. However, trabecular number, thickness, and separation, and structure model index were sensitive to ionizing radiation associated with the µCT scans, resulting in subtle but significant changes over multiple scans. Although the extent of radiation damage on most trabecular bone microarchitecture measures are comparable or far less than the age-related changes during the monitoring period, additional considerations need to be taken to minimize the confounding radiation factors when designing experiments using in vivo µCT imaging for long-term monitoring of mouse bone.




Similar content being viewed by others
References
Altman, A. R., W. J. Tseng, C. M. de Bakker, A. Chandra, S. Lan, B. K. Huh, S. Luo, M. Leonard, L. Qin, and X. S. Liu. Quantification of skeletal growth, modeling, and remodeling by in vivo micro computed tomography. Bone 81:370–379, 2015.
Birkhold, A. I., H. Razi, G. N. Duda, R. Weinkamer, S. Checa, and B. M. Willie. The influence of age on adaptive bone formation and bone resorption. Biomaterials 35:9290–9301, 2014.
Birkhold, A. I., H. Razi, G. N. Duda, R. Weinkamer, S. Checa, and B. M. Willie. Mineralizing surface is the main target of mechanical stimulation independent of age: 3D dynamic in vivo morphometry. Bone 66:15–25, 2014.
Brouwers, J. E., B. van Rietbergen, and R. Huiskes. No effects of in vivo micro-CT radiation on structural parameters and bone marrow cells in proximal tibia of wistar rats detected after eight weekly scans. J Orthop. Res. 25:1325–1332, 2007.
Campbell, G. M., H. R. Buie, and S. K. Boyd. Signs of irreversible architectural changes occur early in the development of experimental osteoporosis as assessed by in vivo micro-CT. Osteoporos. Int. 19(10):1409–1419, 2008.
Campbell, G. M., and A. Sophocleous. Quantitative analysis of bone and soft tissue by micro-computed tomography: applications to ex vivo and in vivo studies. Bonekey Rep. 3:564, 2014.
Cao, X., X. Wu, D. Frassica, B. Yu, L. Pang, L. Xian, M. Wan, W. Lei, M. Armour, E. Tryggestad, J. Wong, C. Y. Wen, W. W. Lu, and F. J. Frassica. Irradiation induces bone injury by damaging bone marrow microenvironment for stem cells. Proc. Natl. Acad. Sci. U.S.A. 108:1609–1614, 2011.
Chandra, A., T. Lin, M. B. Tribble, J. Zhu, A. R. Altman, W. J. Tseng, Y. Zhang, S. O. Akintoye, K. Cengel, X. S. Liu, and L. Qin. PTH1-34 alleviates radiotherapy-induced local bone loss by improving osteoblast and osteocyte survival. Bone 67:33–40, 2014.
Collignon, A., F. Maes, D. Delaere, D. Vandermeulen, P. Suetens, and G. Marchal. Automated multi-modality image registration based on information theory. In: Information processing in medical imaging, edited by Y. Bizais, C. Barillot, and R. Di Paola. Dordrecht: Kluwer, 1995, pp. 263–274.
de Bakker, C. M., A. R. Altman, C. Li, M. B. Tribble, C. Lott, W. J. Tseng, and X. S. Liu. Minimizing interpolation bias and precision error in in vivo microCT-based measurements of bone structure and dynamics. Ann. Biomed. Eng. 44:2518–2528, 2016.
Dudziak, M. E., P. B. Saadeh, B. J. Mehrara, D. S. Steinbrech, J. A. Greenwald, G. K. Gittes, and M. T. Longaker. The effects of ionizing radiation on osteoblast-like cells in vitro. Plast. Reconstr. Surg. 106:1049–1061, 2000.
Gal, T. J., T. Munoz-Antonia, C. A. Muro-Cacho, and D. W. Klotch. Radiation effects on osteoblasts in vitro: a potential role in osteoradionecrosis. Arch. Otolaryngol. Head Neck Surg. 126:1124–1128, 2000.
Glatt, V., E. Canalis, L. Stadmeyer, and M. L. Bouxsein. Age-related changes in trabecular architecture differ in female and male C57BL/6J mice. J. Bone Miner. Res. 22:1197–1207, 2007.
Klinck, R. J., G. M. Campbell, and S. K. Boyd. Radiation effects on bone architecture in mice and rats resulting from in vivo micro-computed tomography scanning. Med. Eng. Phys. 30:888–895, 2008.
Kohler, T., M. Beyeler, D. Webster, and R. Muller. Compartmental bone morphometry in the mouse femur: reproducibility and resolution dependence of microtomographic measurements. Calcif. Tissue Int. 77:281–290, 2005.
Kondo, H., N. D. Searby, R. Mojarrab, J. Phillips, J. Alwood, K. Yumoto, E. A. Almeida, C. L. Limoli, and R. K. Globus. Total-body irradiation of postpubertal mice with (137)Cs acutely compromises the microarchitecture of cancellous bone and increases osteoclasts. Radiat. Res. 171:283–289, 2009.
Lambers, F. M., F. A. Schulte, G. Kuhn, D. J. Webster, and R. Muller. Mouse tail vertebrae adapt to cyclic mechanical loading by increasing bone formation rate and decreasing bone resorption rate as shown by time-lapsed in vivo imaging of dynamic bone morphometry. Bone 49:1340–1350, 2011.
Lan, S., S. Luo, B. K. Huh, A. Chandra, A. R. Altman, L. Qin, and X. S. Liu. 3D image registration is critical to ensure accurate detection of longitudinal changes in trabecular bone density, microstructure, and stiffness measurements in rat tibiae by in vivo micro computed tomography (μCT). Bone 56:83–90, 2013.
Laperre, K., M. Depypere, N. van Gastel, S. Torrekens, K. Moermans, R. Bogaerts, F. Maes, and G. Carmeliet. Development of micro-CT protocols for in vivo follow-up of mouse bone architecture without major radiation side effects. Bone 49:613–622, 2011.
Lu, Y., M. Boudiffa, E. Dall’Ara, I. Bellantuono, and M. Viceconti. Evaluation of in vivo measurement errors associated with micro-computed tomography scans by means of the bone surface distance approach. Med. Eng. Phys. 37:1091–1097, 2015.
Lu, Y., M. Boudiffa, E. Dall’Ara, I. Bellantuono, and M. Viceconti. Development of a protocol to quantify local bone adaptation over space and time: quantification of reproducibility. J. Biomech. 49:2095–2099, 2016.
Matsumura, S., A. Jikko, H. Hiranuma, A. Deguchi, and H. Fuchihata. Effect of X-ray irradiation on proliferation and differentiation of osteoblast. Calcif. Tissue Int. 59:307–308, 1996.
Nishiyama, K. K., G. M. Campbell, R. J. Klinck, and S. K. Boyd. Reproducibility of bone micro-architecture measurements in rodents by in vivo micro-computed tomography is maximized with three-dimensional image registration. Bone 46:155–161, 2010.
Sawajiri, M., J. Mizoe, and K. Tanimoto. Changes in osteoclasts after irradiation with carbon ion particles. Radiat. Environ. Biophys. 42:219–223, 2003.
Schroeder, L. I. W., L. Ng, and J. Cates. The ITK Software Guide. The Insight Consortium. Cambridge: Cambridge University Press, 2003.
Schroeder, W., L. Ng, and J. Cates. The ITK software guide second edition updated for ITK version 2.4. FEBS Lett. 525:53–58, 2015.
Schulte, F. A., F. M. Lambers, G. Kuhn, and R. Muller. In vivo micro-computed tomography allows direct three-dimensional quantification of both bone formation and bone resorption parameters using time-lapsed imaging. Bone 48:433–442, 2011.
Schulte, F. A., F. M. Lambers, D. J. Webster, G. Kuhn, and R. Muller. In vivo validation of a computational bone adaptation model using open-loop control and time-lapsed micro-computed tomography. Bone 49:1166–1172, 2011.
Shrout, P. E., and J. L. Fleiss. Intraclass correlations: uses in assessing rater reliability. Psychol. Bull. 86:420–428, 1979.
Szymczyk, K. H., I. M. Shapiro, and C. S. Adams. Ionizing radiation sensitizes bone cells to apoptosis. Bone 34:148–156, 2004.
Waarsing, J. H., J. S. Day, J. C. van der Linden, A. G. Ederveen, C. Spanjers, N. De Clerck, A. Sasov, J. A. Verhaar, and H. Weinans. Detecting and tracking local changes in the tibiae of individual rats: a novel method to analyse longitudinal in vivo micro-CT data. Bone 34:163–169, 2004.
Willey, J. S., S. A. Lloyd, M. E. Robbins, J. D. Bourland, H. Smith-Sielicki, L. C. Bowman, R. W. Norrdin, and T. A. Bateman. Early increase in osteoclast number in mice after whole-body irradiation with 2 Gy X rays. Radiat Res. 170:388–392, 2008.
Willie, B. M., A. I. Birkhold, H. Razi, T. Thiele, M. Aido, B. Kruck, A. Schill, S. Checa, R. P. Main, and G. N. Duda. Diminished response to in vivo mechanical loading in trabecular and not cortical bone in adulthood of female C57Bl/6 mice coincides with a reduction in deformation to load. Bone 55:335–346, 2013.
Acknowledgements
Research reported in this publication was supported by the Penn Center for Musculoskeletal Diseases (PCMD) NIH/NIAMS P30-AR069619, NIH/NIAMS K01-AR066743 (to XSL), NIH/NIAMS R01-AR071718 (to XSL), and NSF Graduate Research Fellowship (to CMJdB). We would like to thank Carlos Osuna for constructive criticism of the manuscript.
Conflict of interest
All authors have no conflict of interest.
Author information
Authors and Affiliations
Corresponding authors
Additional information
Associate Editor Michael S. Detamore oversaw the review of this article.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
A customized holder fixture for in vivo mouse scanning is available for download as a .stl file for 3D printing: For use, the anesthetized mouse is elongated and laid in a supine position along the bed of the holder fixture. The leg of interest is carefully extruded through the hole in the center of the smaller disk. Then, a 6 inch strip of Coban selfadherent wrap (3M, St. Paul, Minnesota, United States) is cut in half longitudinally, stretched, and then wrapped along the leg until it is comfortably secured in the leg holder, while ensuring that the ankle of the mouse is extended beyond the leg holder by approximately 5mm. The vertebra is fixated by wrapping a 6 inch strip of Coban selfadherent wrap around the distal portion of the mouse body (including the tail) and the bed of the holder fixture three times while ensuring that the binding does not hinder breathing. Once the mouse is restrained appropriately, the scanner door may be shut to load the 2D scout view, followed by selecting the regions of interest for in vivo scanning.
Rights and permissions
About this article
Cite this article
Zhao, H., Chang, CC., Liu, Y. et al. Reproducibility and Radiation Effect of High-Resolution In Vivo Micro Computed Tomography Imaging of the Mouse Lumbar Vertebra and Long Bone. Ann Biomed Eng 48, 157–168 (2020). https://doi.org/10.1007/s10439-019-02323-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10439-019-02323-z