Abstract
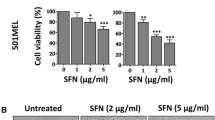
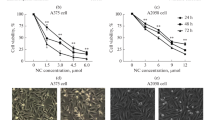
In malignant melanoma complex reprogramming of cell death and survival pathways leads to increased chemoresistance and poor longer-term survival. Sulforaphane (SF) is a promising isothiocyanate compound occurring in cruciferous plants with reported antiproliferative and proapoptotic activity in several tumor cell lines including melanoma. In this work we investigated the effects of SF in several melanoma cell lines and fresh melanoma cultivates. We found that SF is cytotoxic and induces mitochondrial, caspase-dependent apoptosis in our study model, however with lower efficiency in fresh melanoma cultivates. Moreover, our results indicate that in melanoma cell lines and fresh melanoma cultivates SF induces multiple signaling including oxidative stress-mediated activation of DNA-damage response pathway, changes in p38 kinase activity and enhanced expression of Bax and Puma proapoptotic proteins. In addition, in SF-exposed p53-mutant melanoma cells Puma expression seem to be under p38 control and acts as a compensatory proapoptotic mechanism. Conversely, decreased apoptosis in SF-exposed melanoma cultivates might be attributed to Akt-mediated suppression of p38 as well as p53 activity. Together, our results suggest that SF inhibits growth and proliferation and induces mitochondrial apoptosis both in melanoma cell lines as well as in fresh melanoma cultivates. This proapoptotic effect might be enhanced in combination with Akt inhibitors, in particular in melanoma samples. SF is thus commendable for further preclinical testing, both as a single agent as well as in combination regimens.








Similar content being viewed by others
REFERENCES
Gray-Schopfer V, Wellbrock C, Marais R (2007) Melanoma biology and new targeted therapy. Nature 445:851–857
Hocker TL, Singh MK, Tsao H (2008) Melanoma genetics and therapeutic approaches in the 21st century: moving from the benchside to the bedside. J Invest Dermatol 128:2575–2595
Lopez-Bergami P (2011) The role of mitogen- and stress-activated protein kinase pathways in melanoma. Pigment Cell Melanoma Res 24:902–921
Jazirehi AR, Wenn PB, Damavand M (2012) Therapeutic implications of targeting the PI3Kinase/AKT/mTOR signaling module in melanoma therapy. Am J Cancer Res 2:178–191
Ivanov VN, Bhoumik A, Ronai Z (2003) Death receptors and melanoma resistance to apoptosis. Oncogene 22:3152–3161
Soengas MS, Lowe SW (2003) Apoptosis and melanoma chemoresistance. Oncogene 22:3138–3151
Sigalotti L, Covre A, Fratta E, Parisi G, Colizzi F, Rizzo A, Danielli R, Nicolay HJ, Coral S, Maio M (2010) Epigenetics of human cutaneous melanoma: setting the stage for new therapeutic strategies. J Transl Med 8:56
Dhomen N, Marais R (2009) BRAF signaling and targeted therapies in melanoma. Hematol Oncol Clin North Am 23:529–545, ix
Zhang Y, Talalay P, Cho CG, Posner GH (1992) A major inducer of anticarcinogenic protective enzymes from broccoli: isolation and elucidation of structure. Proc Natl Acad Sci USA 89:2399–2403
Juge N, Mithen RF, Traka M (2007) Molecular basis for chemoprevention by sulforaphane: a comprehensive review. Cell Mol Life Sci 64:1105–1127
Singh SV, Herman-Antosiewicz A, Singh AV, Lew KL, Srivastava SK, Kamath R, Brown KD, Zhang L, Baskaran R (2004) Sulforaphane-induced G2/M phase cell cycle arrest involves checkpoint kinase 2-mediated phosphorylation of cell division cycle 25C. J Biol Chem 279:25813–25822
Jakubikova J, Sedlak J, Mithen R, Bao Y (2005) Role of PI3K/Akt and MEK/ERK signaling pathways in sulforaphane- and erucin-induced phase II enzymes and MRP2 transcription, G2/M arrest and cell death in Caco-2 cells. Biochem Pharmacol 69:1543–1552
Myzak MC, Karplus PA, Chung FL, Dashwood RH (2004) A novel mechanism of chemoprotection by sulforaphane: inhibition of histone deacetylase. Cancer Res 64:5767–5774
Gamet-Payrastre L, Li P, Lumeau S, Cassar G, Dupont MA, Chevolleau S, Gasc N, Tulliez J, Terce F (2000) Sulforaphane, a naturally occurring isothiocyanate, induces cell cycle arrest and apoptosis in HT29 human colon cancer cells. Cancer Res 60:1426–1433
Pappa G, Lichtenberg M, Iori R, Barillari J, Bartsch H, Gerhauser C (2006) Comparison of growth inhibition profiles and mechanisms of apoptosis induction in human colon cancer cell lines by isothiocyanates and indoles from Brassicaceae. Mutat Res 599:76–87
Jeong WS, Kim IW, Hu R, Kong AN (2004) Modulatory properties of various natural chemopreventive agents on the activation of NF-kappaB signaling pathway. Pharm Res 21:661–670
Shen G, Xu C, Chen C, Hebbar V, Kong AN (2006) p53-independent G1 cell cycle arrest of human colon carcinoma cells HT-29 by sulforaphane is associated with induction of p21CIP1 and inhibition of expression of cyclin D1. Cancer Chemother Pharmacol 57:317–327
Hamsa TP, Thejass P, Kuttan G (2011) Induction of apoptosis by sulforaphane in highly metastatic B16F-10 melanoma cells. Drug Chem Toxicol 34:332–340
Lombardi T, Castellucci M (1990) Bowes human melanoma cell line. An immunocytochemical study. Virchows Arch B Cell Pathol Incl Mol Pathol 58:181–183
Wu H, Goel V, Haluska FG (2003) PTEN signaling pathways in melanoma. Oncogene 22:3113–3122
Hao H, Muniz-Medina VM, Mehta H, Thomas NE, Khazak V, Der CJ, Shields JM (2007) Context-dependent roles of mutant B-Raf signaling in melanoma and colorectal carcinoma cell growth. Mol Cancer Ther 6:2220–2229
Ubezio P, Civoli F (1994) Flow cytometric detection of hydrogen peroxide production induced by doxorubicin in cancer cells. Free Radic Biol Med 16:509–516
Keuling AM, Andrew SE, Tron VA (2010) Inhibition of p38 MAPK enhances ABT-737-induced cell death in melanoma cell lines: novel regulation of PUMA. Pigment Cell Melanoma Res 23:430–440
Robertson GP (2005) Functional and therapeutic significance of Akt deregulation in malignant melanoma. Cancer Metastasis Rev 24:273–285
Gullett NP, Ruhul Amin AR, Bayraktar S, Pezzuto JM, Shin DM, Khuri FR, Aggarwal BB, Surh YJ, Kucuk O (2010) Cancer prevention with natural compounds. Semin Oncol 37:258–281
Cheung KL, Kong AN (2010) Molecular targets of dietary phenethyl isothiocyanate and sulforaphane for cancer chemoprevention. AAPS J 12:87–97
Misiewicz I, Skupinska K, Kasprzycka-Guttman T (2003) Sulforaphane and 2-oxohexyl isothiocyanate induce cell growth arrest and apoptosis in L-1210 leukemia and ME-18 melanoma cells. Oncol Rep 10:2045–2050
Sestili P, Paolillo M, Lenzi M, Colombo E, Vallorani L, Casadei L, Martinelli C, Fimognari C (2010) Sulforaphane induces DNA single strand breaks in cultured human cells. Mutat Res 689:65–73
Negrette-Guzman M, Huerta-Yepez S, Tapia E, Pedraza-Chaverri J (2013) Modulation of mitochondrial functions by the indirect antioxidant sulforaphane:a seemingly contradictory dual role and an integrative hypothesis. Free Radic Biol Med 65C:1078–1089
Torres M, Forman HJ (2003) Redox signaling and the MAP kinase pathways. Biofactors 17:287–296
Kim AH, Khursigara G, Sun X, Franke TF, Chao MV (2001) Akt phosphorylates and negatively regulates apoptosis signal-regulating kinase 1. Mol Cell Biol 21:893–901
Dhawan P, Singh AB, Ellis DL, Richmond A (2002) Constitutive activation of Akt/protein kinase B in melanoma leads to up-regulation of nuclear factor-kappaB and tumor progression. Cancer Res 62:7335–7342
Pawlik A, Wiczk A, Kaczynska A, Antosiewicz J, Herman-Antosiewicz A (2013) Sulforaphane inhibits growth of phenotypically different breast cancer cells. Eur J Nutr 52(8):1949–1958
Suppipat K, Park CS, Shen Y, Zhu X, Lacorazza HD (2012) Sulforaphane induces cell cycle arrest and apoptosis in acute lymphoblastic leukemia cells. PLoS One 7:e51251
Chaudhuri D, Orsulic S, Ashok BT (2007) Antiproliferative activity of sulforaphane in Akt-overexpressing ovarian cancer cells. Mol Cancer Ther 6:334–345
Mayo LD, Dixon JE, Durden DL, Tonks NK, Donner DB (2002) PTEN protects p53 from Mdm2 and sensitizes cancer cells to chemotherapy. J Biol Chem 277:5484–5489
Acknowledgments
This work was supported by the program PRVOUK P37/01.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Rudolf, K., Cervinka, M. & Rudolf, E. Sulforaphane-induced apoptosis involves p53 and p38 in melanoma cells. Apoptosis 19, 734–747 (2014). https://doi.org/10.1007/s10495-013-0959-7
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10495-013-0959-7