Abstract
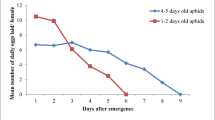
This study investigated the effects of a strain of the fungus Lecanicillium lecanii (Zimm.) Zare and Gams (Hypocreales: Ascomycota) on the aphid Schizaphis graminum (Rondani) (Hemiptera: Aphididae). The fungus was administered to fourth instar nymphs and to alate and apterous adult morphs as a ground rice-kernel formulation. This study showed that L. lecanii formulation affected the survival of the aphids and interacted differently with the studied morphs, the Lethal Time values being lower for alate compared to apterous morphs and nymphs. The treatment also caused a significant reduction in the fecundity of the three treated aphid groups. Histological analysis revealed that the hyphae invaded the host hemocoel of a limited number of alate and apterous morphs: the fungus only entered through the spiracles. Scanning Electron Microscope observations revealed that L. lecanii adhered to the body surface of both adults and nymphs without differentiation. In conclusion, the present study suggests that this strain might be a good candidate for a programme of biological control of S. graminum and other aphid species.





Similar content being viewed by others
References
Abbott WS (1925) A method of computing the effectiveness of an insecticide. J Econ Entomol 18:265–267
Alavo TBC, Sermann H, Bochow H (2001) Virulence of strains of the entomopathogenic fungus Verticillium lecanii to aphids: strain improvement. Arch Phytopath Pflanz 34:379–398
Ashouri S, Arzanian N, Askary H, Rasoulian GR (2004) Pathogenicity of the fungus, Verticillium lecanii, to the green peach aphid, Myzus persicae (Hom.: Aphididae). Commun Agric Appl Biol Sci 69:205–209
Askary H, Carriere R, Belanger RR, Brodeur J (1998) Pathogenicity of the fungus Verticillium lecanii to aphids and powdery mildew. Biocontrol Sci Technol 8:23–32
Askary H, Benhamou N, Brodeur J (1999) Ultrastructural and cytochemical characterisation of aphids invasion by the Hyphomycete Verticillium lecanii. J Invertebr Pathol 74:1–13
Åsman K (2007) Aphid infestation in field grown lettuce and biological control with entomopathogenic fungi (Deuteromycotina: Hyphomycetes). Biol Agric Hortic 25:153–173
Baverstock J, Roy HE, Clark SJ, Alderson PG, Pell JK (2006) Effect of fungal infection on the reproductive potential of aphids and their progeny. J Invertebr Pathol 91:136–139
Brey PT, Latgé JP, Prevost MC (1986) Integumental penetration of the pea aphid, Acyrthosiphon pisum, by Conidiobolus obscurus (Entomophthoraceae). J Invertebr Pathol 48:34–41
Butt TM, Goettel MS (2000) Bioassays of entomogenous fungi. In: Navon A, Ascher KRS (eds) Bioassays of Entomopathogenic Microbes and Nematodes. CAB International Publishing, Wallingford, Oxford, pp 141–195
Butt TM, Ibrahim L, Clark SJ, Beckett A (1995) The germination behaviour of Metarhizium anisopliae on the surface of aphid and flea beetle cuticles. Mycol Res 99:945–950
Butt TM, Jackson C, Magan N (2001) Introduction–fungal biological control agents: progress, problems and potential. In: Butt TM, Jackson C, Magan N (eds) Fungi as biocontrol agents: progress. problems and potential. CAB International Publishing, Wallingford, Oxford, pp 1–8
Claydon N, Grove JF (1982) Insecticidal secondary metabolic products from the entomogenous fungus Verticillium lecanii. J Invertebr Pathol 40:413–418
Cortez-Madrigal H, Alatorre-Rosas R, Mora-Aguilera G, Bravo-Mojica H, Ortiz-Garcia CF, Aceves-Navarro LA (2003) Characterization of multisporic and monosporic isolates of Lecanicillium (= Verticillium) lecanii for the management of Toxoptera aurantii in cocoa. BioControl 48:321–334
Diaz BM, Oggerin M, Lopez Lastra CC, Rubio V, Fereres A (2009) Characterization and virulence of Lecanicillium lecanii against different aphid species. BioControl. doi:10.1007/s10526-009-9218-9
Dromph KM, Pell JK, Eilenberg J (2002) Influence of flight and colour morph on susceptibility of Sitobion avenae to infection by Erynia neoaphidis. Biocontrol Sci Technol 12:753–756
Fargues J, Vidal C, Smits N, Rougier M, Boulard T, Mermier M, Nicot P, Reich P, Jeannequin B, Ridray G, Lagier J (2003) Climatic factors on entomopathogenic hyphomycetes infection of Trialeurodes vaporariorum (Homoptera: Aleyrodidae) in Mediterranean glasshouse tomato. Biol Control 28:320–331
Feng MG, Johnson JB, Kish LP (1990) Virulence of Verticillium lecanii and an aphid-derived isolate of Beauveria bassiana (Fungi: Hyphomycetes) for six species of cereal-infesting aphids (Homoptera: Aphididae). Environ Entomol 19:815–820
Finney DJ (1964) Probit analysis: a statistical treatment of the sigmoid response curve. Cambridge University Press, London
Fournier V, Brodeur J (2000) Dose-response susceptibility of pest aphids (Homoptera: Aphididae) and their control of hydroponically grown lettuce with the entomopathogenic fungus Verticillium lecanii, azaridactin and insecticidal soap. Environ Entomol 29:568–578
Ganassi S, Moretti A, Stornelli C, Fratello B, Bonvicini Pagliai AM, Logrieco A, Sabatini MA (2000) Effects of Fusarium, Paecilomyces and Trichoderma formulations against Schizaphis graminum. Mycopathologia 151:131–138
Ganassi S, Moretti A, Logrieco A, Bonvicini Pagliai AM, Sabatini MA (2004) A laboratory study on colonisation of aphids by some filamentous fungi. In: Simon J-C, Dedryver CA, Rispe C, Hullé M (eds) Aphids in a New Millennium. Proceedings of the VIth International Symposium on Aphids, Versailles, INRA Editions, pp 299–306
Gindin G, Barash I, Harari N, Raccah B (1994) Effect of endotoxic compounds isolated from Verticillium lecanii on the Sweet-potato whitefly, Bemisia tabaci. Phytoparasitica 22:189–196
Hall RA (1984) Epizootic potential for aphids of different isolates of the fungus Verticillium lecanii. Entomophaga 29:311–321
Hall RA, Papierok B (1982) Fungi as biological control agents of arthropods of agricultural and medical importance. Parasitology 84:205–240
Jackson CW, Heale JB, Hall RA (1985) Traits associated with virulence to the aphid Macrosiphoniella sanborni in eighteen isolates of Verticillium lecanii. Ann Appl Biol 106:39–48
James RR, Buckner JS, Freeman TP (2003) Cuticular lipids and silverleaf whitefly stage affect conidial germination of Beauveria bassiana and Paecilomyces fumosoroseus. J Invertebr Pathol 84:67–74
Kaufman PE, Reasor C, Rutz DA, Ketzis JK, Arends JJ (2005) Evaluation of Beauveria bassiana applications against adult house fly, Musca domestica, in commercial caged-layer poultry facilities in New York state. Biol Control 33:360–367
Kim JJ (2007) Influence of Lecanicillium attenuatum on the development and reproduction of the cotton aphid, Aphis gossypii. BioControl 52:789–799
Kim JJ, Goettel MS, Gillespie DR (2007) Potential of Lecanicillium species for dual microbial control of aphids and the cucumber powdery mildew fungus, Sphaerotheca fuliginea. Biol Control 40:327–332
Kope HH, Alfaro RI, Lavallée R (2008) Effects of temperature and water activity on Lecanicillium spp. conidia germination and growth, and mycosis of Pissodes strobe. BioControl 53:489–500
Latgé JP, Sampedro L, Brey P, Diaquin M (1987) Aggressiveness of Conidiobolus obscurus against the pea aphid: influence of cuticular extracts on ballistospore germination of aggressive and non-aggressive strains. J Gen Microbiol 133:1987–1997
Lizen E, Latteur G, Oger R (1985) Sensibilité à l’infection par l’entomophthorale Erynia neoaphidis Remaud. et Henn. du puceron Acyrthosiphon pisum (Harris) selon sa forme, son stade et son âge. Parasitica 41:163–170
Loureiro ES, Moino A Jr (2006) Pathogenicity of Hyphomycete fungi to aphids Aphis gossypii Glover and Myzus persicae (Sultzer) (Hemiptera: Aphididae) Neotropical Entomol 35:660–665
Mulè G, Susca A, Stea G, Moretti A (2004) Specific detection of the toxigenic species Fusarium proliferatum and F. oxysporum from asparagus plants using primers based on calmodulin gene sequences. FEMS Microbiology Letters 230:235–240
Rabasse JM, van Steenis MJ (1999) Biological control of aphids. In: Albajes R, Gullino ML, van Lenteren JC, Elad Y (eds) Integrated Pest and disease Management in Greenhouse Crops. Kluwer Academic Publisher, Dordrecht, pp 235–243
Roberts DW, Humber RA (1981) Entomogenous fungi. In: Cole GT, Kendirick B (eds) Biology of conidial fungi vol 2. Academic Press London, pp 201–236
Roditakis E, Couzin ID, Franks NR, Charnley AK (2008) Effects of Lecanicillium longisporum infection on the behaviour of the green peach aphid Myzus persicae. J Insect Physiol 54:128–136
Safavi SA, Rassulian GR, Askary H, Pakdel AK (2002) Pathogenicity and virulence of entomogenous fungus Verticillium lecanii (Zimm.) Viegas on the pea aphid, Acyrthosiphon pisum (Harris). J Sci Technol Agric Nat Resour 6:245–255
Saito T, Aoki J (1983) Toxicity of free fatty acids on the larval surfaces of two lepidopterous insects towards Beauveria bassiana (Bals.) Vuill. and Paecilomyces fumosoroseus (Wize) Brown and Smith (Deuteromycetes: Moniliales). Appl Entomol Zool 18:225–233
Schreiter G, Butt TM, Beckett A, Vestergaard S, Moritz G (1994) Invasion and development of Verticillium lecanii in the western flower thrips, Frankliniella occidentalis. Mycol Res 98:1025–1034
Seifert KA, Samson RA, Deward JR, Houbraken J, Levesque CA, Monvalvo JM, Louise-Seize G, Hebert PDN (2007) Prospects for fungus identification using CO1 DNA barcodes, with Penicillium as a test case. Proc Natl Acad Sci USA 104:3901–3906
Shah PA, Pell JK (2003) Entomopathogenic fungi as biological control agents. Appl Microbiol Biotechnol 61:413–423
Smith RJ, Grula EA (1982) Toxic components on the larval surface of the corn earworm (Heliothis zea) and their effects on germination and growth of Beauveria bassiana. J Invertebr Pathol 39:15–22
Sosa-Gomez DR, Boucias DG, Nation JL (1997) Attachment of Metarhizium anisopliae to the southern green stink bug Nezara viridula cuticle and fungistatic effect of cuticular lipids and aldehydes. J Invertebr Pathol 69:31–39
SPSS (2007) Statistical product and service solution, system user’s guide. Version 15.01, SPSS Inc., Chicago
St Leger RJ, Goettel M, Roberts DW, Staples RC (1991) Prepenetration events during infection of host cuticle by Metarhizium anisopliae. J Invertebr Pathol 58:168–179
Steenberg T, Humber RA (1999) Entomopathogenic potential of Verticillium and Acremonium species (Deuteromycotina: Hyphomycetes). J Invertebr Pathol 73:309–314
Steinkraus DC (2006) Factors affecting transmission of fungal pathogens of aphids. J Invertebr Pathol 92:125–131
Sugimoto M, Koike M, Hiyama N, Nagao H (2003) Genetic, morphological, and virulence characterisation of the entomopathogenic fungus Verticillium lecanii. J Invertebr Pathol 82:176–187
Vacca LL (1985) Laboratory manual of histochemistry. Raven Press, New York
van Lenteren JC (2000) A greenhouse without pesticide: fact or fantasy? Crop Prot 19:375–384
Vu VH, Hong SI, Kim K (2007) Selection of entomopathogenic fungi for aphid control. J Biosci Bioeng 104:498–505
Zare R, Gams W (2001) A revision of Verticillium section Prostrata. IV. The genera Lecanicillium and Simplicillium gen. nov. Nova Hedwigia 73:1–50
Acknowledgments
We thank Monica Santamaria, Istituto di Tecnologie Biomediche, CNR, Bari, Italy and Bachir Balech, International Center for Advanced Mediterranean Agronomic Studies (IAM.B), Valenzano - (Bari), Italy, for designing and providing the COX1 primers developed in the behalf of the thesis of Bachir Balech “An integrated molecular and morphological study to design a barcode discrimination protocol for Fusarium species involved in dry root disease of Citrus” for the Master of Science in “Integrated Pest Management of Mediterranean Fruit Tree Crops” supported by the Italian Minister of Foreign Affairs. We thank Vincenzo Ricci for its valuable technical assistance. We thank the “Centro Interdipartimentale Grandi Strumenti” of the University of Modena and Reggio Emilia for the availability of the Philips XL-30 Scanning Electron Microscope.
Author information
Authors and Affiliations
Corresponding author
Additional information
Handling Editor: Helen Roy.
Rights and permissions
About this article
Cite this article
Ganassi, S., Grazioso, P., Moretti, A. et al. Effects of the fungus Lecanicillium lecanii on survival and reproduction of the aphid Schizaphis graminum . BioControl 55, 299–312 (2010). https://doi.org/10.1007/s10526-009-9250-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10526-009-9250-9