Abstract
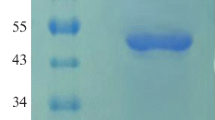
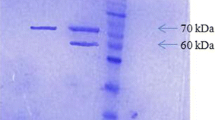
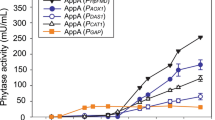
A phytase gene from Aspergillus niger was isolated and two Escherichia coli expression systems, based on T7 RNA polymerase promoter and tac promoter, were used for its recombinant expression. Co-expression of molecular chaperone, GroES/EL, aided functional cytosolic expression of the phytase in E. coli BL21 (DE3). Untagged and maltose-binding protein-tagged recombinant phytase showed an activity band of ~49 and 92 kDa, respectively, on a zymogram. Heterologously-expressed phytase was fractionated from endogenous E. coli phytase by (NH4)2SO4 precipitation. The enzyme had optimum activity at 50 °C and pH 6.5.





Similar content being viewed by others
References
Angov E (2011) Codon usage: nature’s road map to expression and folding of proteins. Biotechnology 6:650–659
Baneyx F, Mujacic M (2004) Recombinant proteinfolding and misfolding in Escherichia coli. Nat Biotechnol 22:1399–1408
Khan RH, Rasheedi S, Haq SK (2003) Effect of pH, temperature and alcohols on the stability of glycosylated and deglycosylated stem bromelain. J Biosci 28:709–714
Kumar V, Sinha AK, Harinder PSM, Becker K (2010) Dietary roles of phytate and phytase in human nutrition: a review. Food Chem 120:945–959
Li Y, Yang G, Huang X, Ye B, Liu M, Lin Z, Li C, Cao ZA (2009) Recombinant glutamine synthetase (GS) from C. glutamicum existed as both hexamers and dedocamers and C-terminal His-tag enhanced inclusion bodies formation in E. coli. Appl Biochem Biotechnol 159:614–622
Nishihara K, Kanemori M, Kitagawa M, Yanagi H, Yura T (1998) Chaperone co-expression plasmids: differential and synergistic roles of DnaK-DnaJ-GrpE and GroEL-GroES in assisting folding of an allergen of Japanese cedar pollen, Cryj2, in Escherichia coli. Appl Environ Microbiol 64:1694–1699
Phillippy BQ, Mullaney EJ (1997) Expression of an Aspergillus niger phytase (phyA) in Escherichia coli. J Agric Food Chem 45:3337–3342
Senerovic L, Stankovic N, Ljubijankic G, Vasiljevic B (2009) Glycosylation and pH stability of penicillin G acylase from Providencia rettgeri produced in Pichia pastoris. Arch Biol Sci 61:581–586
Shi XW, M-Lv Sun, Zhou B, Wang XY (2009) Identification, characterization, over-expression of phytase with potential industrial interest. Can J Microbiol 55:599–604
Ullah AHJ, Gibson DM (1987) Extracellular phytase (E.C.3.1.3.8) from Aspergillus ficuum NRRL 3135: purification and characterization. Prep Biochem 17:63–91
Ushasree MV, Sumayya HBS, Pandey A (2011) Comparative analysis of recombinant expression and solubility screening of two phytases in E. coli. Food Technol Biotechnol 49:304–309
Ushasree MV, Gunasekaran P, Pandey A (2012) Single-step purification and immobilization of MBP-phytase fusion on starch agar beads: application in dephytination of soymilk. Appl Biochem Biotechnol 167:981–990
van Hastingsveldt W, Van ZCMJ, Harteveld MG, Gouka RJ, Suykerbuyk MEG, Luiten RGM, van Paridon PA, Selten GCM, Veenstra AE, van Gorcom RF, van den Hondel CAMJJ (1993) Cloning, characterization and overexpression of the phytase-encoding gene (phyA) of Aspergillus niger. Gene 127:87–94
Wang S, Tan A, Lv J, Wang P, Yin X, Chen Y (2012) Soluble expression of recombinant human CD137 ligand in Escherichia coli by co-expression of chaperones. J Ind Microbiol Biotechnol 39:471–476
Acknowledgments
The authors would like to thank Department of Biotechnology, New Delhi and Indian Council of Medical Research, New Delhi, India for providing financial support.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Ushasree, M.V., Vidya, J. & Pandey, A. Gene cloning and soluble expression of Aspergillus niger phytase in E. coli cytosol via chaperone co-expression. Biotechnol Lett 36, 85–91 (2014). https://doi.org/10.1007/s10529-013-1322-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10529-013-1322-3