Abstract
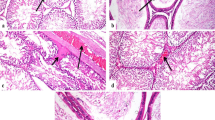
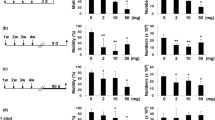
With the continuing development of nanomaterials, the assessment of their potential impact on human health, and especially human reproductive toxicity, is a major issue. The testicular biodistribution of nanoparticles remains poorly studied. This study investigated whether gold-silica nanoparticles could be detected in mouse testes after intramuscular injection, with a particular focus on their ability to cross the blood–testis barrier. To that purpose, well-characterized 70-nm gold core–silica shell nanoparticles were used to ensure sensitive detection using high-resolution techniques. Testes were collected at different time points corresponding to spermatogenesis stages in mice. Transmission electron microscopy and confocal microscopy were used for nanoparticle detection, and nanoparticle quantification was performed by atomic emission spectroscopy. All these techniques showed that no particles were able to reach the testes. Results accorded with the normal histological appearance of testes even at 45 days post sacrifice. High-resolution techniques did not detect 70-nm silica-gold nanoparticles in mouse testes after intramuscular injection. These results are reassuring about the safety of nanoparticles with regard to male human reproduction, especially in the context of nanomedicine.







Similar content being viewed by others
References
N. Asare, C. Instanes, W.J. Sandberg, M. Refsnes, P. Schwarze, M. Kruszewski, G. Brunborg, Toxicology 291, 65 (2012)
Y. Bai, Y. Zhang, J. Zhang, Q. Mu, W. Zhang, E.R. Butch, S.E. Snyder, B. Yan, Nat. Nanotechnol. 5, 683 (2010)
S.K. Balasubramanian, J. Jittiwat, J. Manikandan, C.-N. Ong, L.E. Yu, W.-Y. Ong, Biomaterials 31, 2034 (2010)
L. Braydich-Stolle, S. Hussain, J.J. Schlager, M.-C. Hofmann, Toxicol. Sci. Off. J. Soc. Toxicol. 88, 412 (2005)
J. Chen, H. Wang, W. Long, X. Shen, D. Wu, S.-S. Song, Y.-M. Sun, P.-X. Liu, S. Fan, F. Fan, X.-D. Zhang, Int. J. Nanomedicine 8, 2409 (2013)
W.H. De Jong, W.I. Hagens, P. Krystek, M.C. Burger, A.J.A.M. Sips, R.E. Geertsma, Biomaterials 29, 1912 (2008)
M. Ema, N. Kobayashi, M. Naya, S. Hanai, J. Nakanishi, Reprod. Toxicol. Elmsford N 30, 343 (2010)
T. Fifis, A. Gamvrellis, B. Crimeen-Irwin, G.A. Pietersz, J. Li, P.L. Mottram, I.F.C. McKenzie, M. Plebanski, J. Immunol. 173, 3148 (2004)
L. Geraets, A.G. Oomen, J.D. Schroeter, V.A. Coleman, F.R. Cassee, Toxicol. Sci. Off. J. Soc. Toxicol. 127, 463 (2012)
A.E. Gregory, R. Titball, D. Williamson, Front. Cell. Infect. Microbiol. 3, 13 (2013)
J. Gromadzka-Ostrowska, K. Dziendzikowska, A. Lankoff, M. Dobrzyńska, C. Instanes, G. Brunborg, A. Gajowik, J. Radzikowska, M. Wojewódzka, M. Kruszewski, Toxicol. Lett. 214, 251 (2012)
J.-P. Klein, D. Boudard, J. Cadusseau, S. Palle, V. Forest, J. Pourchez, M. Cottier, Biomed. Microdevices 15, 427 (2013)
T. Komatsu, M. Tabata, M. Kubo-Irie, T. Shimizu, K.-I. Suzuki, Y. Nihei, K. Takeda, Toxicol. Vitro Int. J. Publ. Assoc. BIBRA 22, 1825 (2008)
J. Lackner, G. Schatzl, T. Waldhör, K. Resch, C. Kratzik, M. Marberger, Fertil. Steril. 84, 1657 (2005)
Z. Lan, W.-X. Yang, NanoMed. 7, 579 (2012)
D.P.K. Lankveld, A.G. Oomen, P. Krystek, A. Neigh, A. Troost-de Jong, C.W. Noorlander, J.C.H. Van Eijkeren, R.E. Geertsma, W.H. De Jong, Biomaterials 31, 8350 (2010)
J.H. Lee, Y.S. Kim, K.S. Song, H.R. Ryu, J.H. Sung, J.D. Park, H.M. Park, N.W. Song, B.S. Shin, D. Marshak, K. Ahn, J.E. Lee, I.J. Yu, Part. Fibre Toxicol. 10, 36 (2013)
C. Li, S. Taneda, K. Taya, G. Watanabe, X. Li, Y. Fujitani, Y. Ito, T. Nakajima, A.K. Suzuki, Inhal. Toxicol. 21, 803 (2009)
M. Martini, P. Perriat, M. Montagna, R. Pansu, C. Julien, O. Tillement, S. Roux, J. Phys. Chem. C 113, 17669 (2009)
Y. Morishita, Y. Yoshioka, H. Satoh, N. Nojiri, K. Nagano, Y. Abe, H. Kamada, S. Tsunoda, H. Nabeshi, T. Yoshikawa, Y. Tsutsumi, Biochem. Biophys. Res. Commun. 420, 297 (2012)
G. Oberdörster, J. Ferin, B.E. Lehnert, Environ. Health Perspect. 102(5), 173 (1994)
M.O. Oyewumi, A. Kumar, Z. Cui, Expert Rev. Vaccines 9, 1095 (2010)
E.-J. Park, E. Bae, J. Yi, Y. Kim, K. Choi, S.H. Lee, J. Yoon, B.C. Lee, K. Park, Environ. Toxicol. Pharmacol. 30, 162 (2010)
L.J. Peek, C.R. Middaugh, C. Berkland, Adv. Drug Deliv. Rev. 60, 915 (2008)
J.R. Peralta-Videa, L. Zhao, M.L. Lopez-Moreno, G. de la Rosa, J. Hong, J.L. Gardea-Torresdey, J. Hazard. Mater. 186, 1 (2011)
D.H. Ramdhan, Y. Ito, Y. Yanagiba, N. Yamagishi, Y. Hayashi, C. Li, S. Taneda, A.K. Suzuki, G. Watanabe, K. Taya, M. Kamijima, T. Nakajima, Toxicol. Lett. 191, 103 (2009)
L. Reimer, Transmission Electron Microscopy, Second Edition (Springer-Verlag, 1989)
M. Rolland, J. Le Moal, V. Wagner, D. Royère, J. De Mouzon, Hum. Reprod. Oxf. Engl. 28, 462 (2013)
R. Tassinari, F. Cubadda, G. Moracci, F. Aureli, M. D’Amato, M. Valeri, B. De Berardis, A. Raggi, A. Mantovani, D. Passeri, M. Rossi, F. Maranghi, Nanotoxicology 8, 654 (2014)
M. van der Zande, R.J. Vandebriel, E. Van Doren, E. Kramer, Z. Herrera Rivera, C.S. Serrano-Rojero, E.R. Gremmer, J. Mast, R.J.B. Peters, P.C.H. Hollman, P.J.M. Hendriksen, H.J.P. Marvin, A.A.C.M. Peijnenburg, H. Bouwmeester, ACS Nano 6, 7427 (2012)
D. B. Williams, Practical Analytical Electron Microscopy in Materials Science (Philips Electronic Instruments, 1984)
Y. Xu, N. Wang, Y. Yu, Y. Li, Y.-B. Li, Y.-B. Yu, X.-Q. Zhou, Z.-W. Sun, PLoS ONE 9, e101572 (2014)
S. Yoshida, K. Hiyoshi, T. Ichinose, H. Takano, S. Oshio, I. Sugawara, K. Takeda, T. Shibamoto, Int. J. Androl. 32, 337 (2009)
Acknowledgments
The authors would like to acknowledge the financial support of the French Rhône Alpes Region through the ARC3 Environnement Research Program. They are grateful to Dr N. Blanchard for his help in operating the TOPCON microscope at ILM Institute.
Authors’ contributions
LL, JPK, MGB and MC contributed to the conception and the design of research.
LL, JPK, VF, DB, MM and MGB materially participated in the research experiments analysis and interpretation of data.
LL and JPK were involved with drafting article.
JP and all authors were involved with reviewing the article.
All authors have approved the final version of this article.
Compliance with ethical standards
All experiments were performed in compliance with the French laws and institutional guidelines. An authorization approving the experiments was obtained from the French Ministère de l’Enseignement Supérieur et de la Recherche (MESR, field number 00462.02).
Conflict of interest
The authors declare that they have no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Additional information
Lara Leclerc and Jean-Philippe Klein contributed equally to this work.
Rights and permissions
About this article
Cite this article
Leclerc, L., Klein, JP., Forest, V. et al. Testicular biodistribution of silica-gold nanoparticles after intramuscular injection in mice. Biomed Microdevices 17, 66 (2015). https://doi.org/10.1007/s10544-015-9968-3
Published:
DOI: https://doi.org/10.1007/s10544-015-9968-3