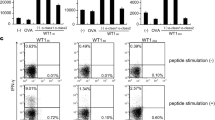
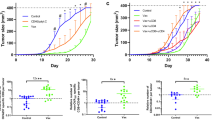
Abstract
Studies conducted in animal model of infectious diseases or H-Y antigen model suggest a crucial role for CD4+ T cells in providing help for CD8+ T-cell memory responses. This concept suggests that inclusion of T helper epitopes in vaccine formulation will result in improved CD8+ T-cell responses. Although this concept has been applied to cancer vaccine design, the role of CD4+ T cells in the memory differentiation of CD8+ T cells and retention of their anti-tumor function have never been tested in breast cancer model. Using the FVB mouse model of neu-positive breast carcinoma we report for the first time that helpless T cells showed cytostatic or tumor inhibitory effects during primary tumor challenge whereas, helped T cells showed cytotoxic effects and resulted in complete tumor rejection. Such differential effects, in vivo, were associated with higher frequency of CD8+PD-L1+ and CD8+PD-1+ T cells in animals harboring helpless T cells as well as higher titer of IL-2 in the sera of animals harboring helped T cells. However, depletion of CD4+ T cells did not alter the ability of neu-specific CD8+ T cells to differentiate into memory cells and to retain their effector function against the tumor during recall challenge. These results suggest the inhibitory role of CD4+ T cells on CD8+ T-cell exhaustion without substantial effects on the differentiation of memory T cells during priming phase of the immune responses against breast cancer.




Similar content being viewed by others
References
Shedlock DJ, Shen H (2003) Requirement for CD4 T cell help in generating functional CD8 T cell memory. Science 300(5617):337–339
Khanolkar AM, Fuller J, Zajac AJ (2004) CD4 T cell-dependent CD8 T cell maturation. J Immunol 172(5):2834–2844
Sun JC, Williams MA, Bevan MJ (2004) CD4+ T cells are required for the maintenance, not programming, of memory CD8+ T cells after acute infection. Nat Immunol 5(9):927–933
Bourgeois C, Rocha B, Tanchot C (2002) A role for CD40 expression on CD8+ T cells in the generation of CD8+ T cell memory. Science 297(5589):2060–2063
Janssen EM, Droin NM, Lemmens EE et al (2005) CD4+ T-cell help controls CD8+ T-cell memory via TRAIL-mediated activation-induced cell death. Nature 434(7029):88–93
Elsaesser H, Sauer K, Brooks DG (2009) IL-21 is required to control chronic viral infection. Science 324:1569–1572
Phan GQ, Touloukian CE, Yang JC et al (2003) Immunization of patients with metastatic melanoma using both class I- and class II-restricted peptides from melanoma-associated antigens. J Immunother 26(4):349–356
Kmieciak M, Knutson KL, Dumur CI, Manjili MH (2007) HER-2/neu antigen loss and relapse of mammary carcinoma are actively induced by T cell-mediated anti-tumor immune responses. Eur J Immunol 37(3):675–685
Worschech A, Kmieciak M, Knutson KL et al (2008) Signatures associated with rejection or recurrence in HER-2/neu-positive mammary tumors. Cancer Res 68(7):2436–2446
Wang EL, Miller D, Ohnmacht GA, Liu ET, Marincola FM (2000) High fidelity mRNA amplification for gene profiling. Nat Biotechnol 18(4):457–459
Wang E (2005) RNA amplification for successful gene profiling analysis. J Transl Med 3:28
Jin P, Zhao Y, Ngalame Y et al (2005) Selection and validation of endogenous reference genes using a high throughput approach. BMC Genom 5(1):55
Rubinfeld B, Robbins P, el Gamil M, Albert I, Porfiri E, Polakis P (1997) Stabilization of beta-catenin by genetic defects in melanoma cell lines. Science 275(5307):1790–1792
Eisen MB, Spellman PT, Brown PO, Botstein D (1998) Cluster analysis and display of genome-wide expression patterns. Proc Natl Acad Sci USA 95(25):14863–14868
Ross DT, Scherf U, Eisen MB et al (2000) Systematic variation in gene expression patterns in human cancer cell lines. Nat Genet 24(3):227–235
Wang E, Miller LD, Ohnmacht GA et al (2002) Prospective molecular profiling of melanoma metastases suggests classifiers of immune responsiveness. Cancer Res 62:3581–3586
Basil CF, Zhao Y, Zavaglia K et al (2006) Common cancer biomarkers. Cancer Res 66(6):2953–2961
Elmore LW, Turner KC, Gollahon LS, Landon MR, Jackson-Cook CK, Holt SE (2002) Telomerase protects cancer-prone human cells from chromosomal instability and spontaneous immortalization. Cancer Biol Ther 1(4):391–397
Bucks CM, Norton JA, Boesteanu AC, Mueller YM, Katsikis PD (2009) Chronic antigen stimulation alone is sufficient to drive CD8+ T cell exhaustion. J Immunol 182(11):6697–6708
Streeck H, Brumme ZL, Anastario M, et al (2008) Antigen load and viral sequence diversification determine the functional profile of HIV-1-specific CD8+ T cells. PLoS Med 6;5(5):e100
Frank GM, Lepisto AJ, Freeman ML, Sheridan BS, Cherpes TL, Hendricks RL (2010) Early CD4(+) T cell help prevents partial CD8(+) T cell exhaustion and promotes maintenance of Herpes Simplex Virus 1 latency. J Immunol 184(1):277–286
Hemann MT, Strong MA, Hao LY, Greider CW (2001) The shortest telomere, not average telomere length, is critical for cell viability and chromosome stability. Cell 107(1):67–77
Hinrichs CS, Borman ZA, Cassard L et al (2009) Adoptively transferred effector cells derived from naive rather than central memory CD8+ T cells mediate superior antitumor immunity. Proc Natl Acad Sci USA 106(41):17469–17474
Xie Y, Akpinarli A, Maris C et al (2010) Naive tumor-specific CD4+ T cells differentiated in vivo eradicate established melanoma. J Exp Med Feb 15. (Epub ahead of print)
Zhou J, Shen X, Huang J, Hodes RJ, Rosenberg SA, Robbins PF (2005) Telomere length of transferred lymphocytes correlates with in vivo persistence and tumor regression in melanoma patients receiving cell transfer therapy. J Immunol 175(10):7046–7052
Shen X, Zhou J, Hathcock KS et al (2007) Persistence of tumor infiltrating lymphocytes in adoptive immunotherapy correlates with telomere length. J Immunother 30(1):123–129
Wrzesinski C, Paulos CM, Kaiser A et al (2010) Increased intensity lymphodepletion enhances tumor treatment efficacy of adoptively transferred tumor-specific T cells. J Immunother 33(1):1–7
Rosenberg SA, Dudley ME (2009) Adoptive cell therapy for the treatment of patients with metastatic melanoma. Curr Opin Immunol 21(2):233–240
Acknowledgments
This work was supported by NIH R01 CA104757 Grant (M. H. Manjili) and flow cytometry shared resources facility supported in part by the NIH Grant P30CA16059. We thank Dr. William Lee of the U Penn for providing us with pEF2-dnIFN-γ Rα vector. We also thank Julie Farnsworth for her expertise in cell sorting and immense dedication to furthering the research at our institution. We gratefully acknowledge the support of VCU Massey Cancer Centre and the Commonwealth Foundation for Cancer Research.
Author information
Authors and Affiliations
Corresponding author
Additional information
M. Kmieciak and A. Worschech had equal contribution to this work.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Fig. S1
FVB mice (n = 3) were depleted of CD4+ and CD8+ T cells by i.p. injection of GK1.5 and 2.43 Abs, respectively. Animals were then inoculated with MMC (5 × 106 cells/mouse) and tumor growth was determined. (TIFF 2457 kb)
Rights and permissions
About this article
Cite this article
Kmieciak, M., Worschech, A., Nikizad, H. et al. CD4+ T cells inhibit the neu-specific CD8+ T-cell exhaustion during the priming phase of immune responses against breast cancer. Breast Cancer Res Treat 126, 385–394 (2011). https://doi.org/10.1007/s10549-010-0942-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10549-010-0942-8