Abstract
Background
Due to the high rate of febrile neutropenia (FN) with docetaxel–cyclophosphamide (DC) chemotherapy, primary FN prophylaxis is recommended. However, the optimal choice of prophylaxis [i.e., granulocyte-colony stimulating factors (G-CSF) or antibiotics] is unknown. A systematic review was performed to address this knowledge gap.
Methods
Embase, Ovid Medline, Pubmed, the Cochrane database of systematic reviews, and Cochrane register of controlled trials were searched from 1946 to April 2016 for studies evaluating primary prophylactic FN treatments in breast cancer patients receiving DC chemotherapy. Outcome measures evaluated included: incidence of FN and treatment-related hospitalizations, chemotherapy dose reduction/delays/discontinuations, and adverse events. Screening and data collection were performed by two independent reviewers.
Results
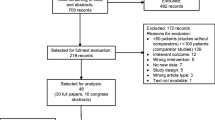
Of 2105 identified records, 7 studies (n = 2535) met the pre-specified eligibility criteria. Seven additional studies (n = 621) were identified from prior systematic reviews. There were 3 randomized controlled trials (RCTs) (n = 2256) and 11 retrospective studies (n = 900). Study sample sizes ranged from 30 to 982 patients (median 99.5), evaluating pegfilgrastim (n = 1274), filgrastim (n = 1758), and oral ciprofloxacin (n = 108). Given the heterogeneity of patients and study design, a narrative synthesis of results was performed. Median FN rates with and without primary prophylaxis were 6.6 % (IQR 3.9–10.6 %) and 31.3 % (IQR 25–33 %), respectively. No FN-related deaths were reported. No RCT directly compared G-CSF with antibiotic interventions.
Conclusions
Primary FN prophylaxis reduces the incidence of FN. Despite considerable cost and toxicity differences between G-CSF and antibiotics, there is insufficient data to make a recommendation of one strategy over another.

Similar content being viewed by others
References
Fishe B, Jeong JH, Dignam J et al (2001) Findings from recent National Surgical Adjuvant Breast and Bowel Project (NSABP) adjuvant studies in stage I breast cancer. J Natl Cancer Inst Monogr 30:62–66
National Comprehensive Cancer Network. Clinical Practice guidelines in oncology: breast cancer. V2.2013. http://www.nccn.org. Accessed 1 Sept 2016
Rastogi P, Anderson SJ, Bear HD et al (2008) Preoperative chemotherapy: updates of National surgical adjuvant breast and bowel protocols B-18 and B-27. J Clin Oncol 26(5):778–785
Jones S, Holmes FA, O’Shaughnessy J et al (2009) Docetaxel with cyclophosphamide is associated with an overall survival benefit compared with doxorubicin and cyclophosphamide: 7-year follow-up of US Oncology Research Trial 9735. J Clin Oncol 27(8):1177–1183
Jones SE, Savin MA, Holmes FA et al (2006) Phase III trial comparing doxorubicin plus cyclophosphamide with docetaxel plus cyclophosphamide as adjuvant therapy for operable breast cancer. J Clin Oncol 24(34):5381–5387
Culakova E, Thota R, Poniewierski MS et al (2014) Patterns of chemotherapy-associated toxicity and supportive care in US oncology practice: a nationwide prospective cohort study. Cancer Med 2:434–444
Lathia N, Mittmann N, De Angelis C et al (2010) Evaluation of direct medical costs of hospitalization for febrile neutropenia. Cancer 116:742–748
Dinan MA, Hirsch BR, Lyman GH (2015) Management of chemotherapy-induced neutropenia: measuring quality, cost, and value. J Natl Compr Cancer Netw 13:e1–e7
Do T, Medhekar R, Bhat R et al (2015) The risk of febrile neutropenia and need for G-CSF primary prophylaxis with the docetaxel and cyclophosphamide regimen in early-stage breast cancer patients: a meta-analysis. Breast Cancer Res Treat 153(3):591–597
Younis T, Rayson D, Thompson K (2012) Primary G-CSF prophylaxis for adjuvant TC or FEC-D chemotherapy outside of clinical trial settings: a systematic review and meta-analysis. Support Care Cancer 20(10):2523–2530
Cancer Care Ontario (2016) Cancer Canre Ontario GCSF Recommendations 2016. Version March 21, 2016. https://www.cancercare.on.ca/common/pages/UserFile.aspx?fileId=352101. Accessed 5 Aug 2016
Crawford J, Allen J, Armitage J et al (2012) NCCN Clinical Practice Guidelines in Oncology (NCCN Guidelines®) Myeloid Growth Factors. Version 1.2012. https://www.tri-kobe.org/nccn/guideline/hematologic/english/myeloid_growth.pdf. Accessed 10 Aug 2016
Aapro M, Bohlius J, Cameron D et al (2011) 2010 update of EORTC guidelines for the use of granulocyte-colony stimulating factor to reduce the incidence of chemotherapy-induced febrile neutropenia in adult patients with lymphoproliferative disorders and solid tumors. Eur J Cancer 47:8–32
Smith TJ, Bohlke K, Lyman GH et al (2015) Recommendations for the use of WBC growth factors: American Society of Clinical Oncology Clinical Practice Guideline Update. J Clin Oncol 33(28):3199–3212
Skedgel C, Rayson D, Younis T (2016) Is febrile neutropenia prophylaxis with granulocyte-colony stimulating factors economically justified for adjuvant TC chemotherapy in breast cancer? Support Care Cancer 24(1):387–394
Higgins JPT, Green S (ed) (2011) Cochrane handbook for systematic reviews of interventions Version 5.1.0. The Cochrane Collaboration
Moher D, Liberati A, Tetzlaff J et al (2009) Preferred reporting items for systematic reviews and meta-analyses: the PRISMA statement. PLoS Med 6(7):e1000097
Gluz O, Hofmann D, Von SR et al (2014) Febrile neutropenia (FN) and infections under adjuvant chemotherapy of breast cancer with 6 × TC vs. 4 × EC-4 × Doc: toxicity data of the WSG planB trial. Oncol Res Treat 37:14–15
Kosaka Y, Rai Y, Masuda N et al (2015) Phase III placebo-controlled, double-blind, randomized trial of pegfilgrastim to reduce the risk of febrile neutropenia in breast cancer patients receiving docetaxel/cyclophosphamide chemotherapy. Support Care Cancer 23(4):1137–1143
Yu JL, Chan K, Kurin M et al (2015) Clinical outcomes and cost-effectiveness of primary prophylaxis of febrile neutropenia during adjuvant docetaxel and cyclophosphamide chemotherapy for breast cancer. Breast J 21(6):658–664
Yerushalmi R, Goldvaser H, Sulkes A et al (2014) Adjuvant docetaxel and cyclophosphamide (DC) with prophylactic granulocyte colony-stimulating factor (G-CSF) on days 8 & 12 in breast cancer patients: a retrospective analysis. PLoS ONE 9(10):e107273
Caley A, Bertelli G, Rolles M et al (2010) Adjuvant taxane chemotherapy is associated with a significant risk of febrile neutropenia. Eur J Cancer Suppl 8(3):70
Yau T-K, Ng T-Y, Soong IS et al (2009) Toxicity of docetaxel plus cyclophosphamide as adjuvant therapy for breast cancer in Chinese patients - The Hong Kong experience. Asia Pac J Clin Oncol 5(2):123–128
Vogel C, Rader M, Tyulandin S et al (2005) Pegfilgrastim nearly abrogates occurrence of neutropenic events early in the course of chemotherapy: results of a phase III, randomized, double-blind, placebo-controlled study of patients with breast cancer receiving docetaxel. J Support Oncol 3(2 SUPPL. 1):58–59
Marinho FDS, Lopes MDS, Monteiro MMF et al (2011) Incidence of febrile neutropenia with adjuvant docetaxel and cyclophosphamide in patients with early breast cancer. J Clin Oncol 29(suppl):e11501
Soni A, Brufsky A, Jankowitz RC et al (2011) Incidence of febrile neutropenia with docetaxel plus cyclophosphamide in a university-based breast oncology clinic. J Clin Oncol 29(suppl):9061
Kotasek D (2011) Febrile neutropenia rates during docetaxel and cyclophosphamide (TC) adjuvant therapy in early breast cancer (EBC). J Clin Oncol 29(suppl):1101
Santos FN, Cruz MR, Cezana L et al (2010) Hematologic toxicity with adjuvant docetaxel and cyclophosphamide in early breast cancer. J Clin Oncol 28(suppl)(15):e11081
Vandenberg T, Younus J, Al-Khayyat S (2010) Febrile neutropenia rates with adjuvant docetaxel and cyclophosphamide chemotherapy in early breast cancer: discrepancy between published reports and community practice—a retrospective analysis. Curr Oncol 17(2):2–3
Ngamphaiboon N, Advani PP, O’Connor TL et al (2011) Febrile neutropenia in adjuvant docetaxel and cyclophosphamide (TC) with prophylactic pegfilgrastim in patients with breast cancer: a retrospective analysis. J Clin Oncol 29(suppl):1134
Chan KK, Trudeau ME, Eisen A et al (2011) The cost-effectiveness of primary prophylaxis with granulocyte colony-stimulating factor in docetaxel-containing adjuvant chemotherapy in early breast cancer: the impact of risk of febrile neutropenia and its mortality. J Clin Oncol 29(suppl)(15):6086
Bayer Inc. (2016) Product Monograph Cipro XL. Version March 9, 2015. http://www.bayer.ca/omr/online/cipro-xl-pm-eng-9mar2015.pdf. Accessed 6 Sept 2016
Amgen Canada Inc. (2016) Product Monograph Neupogen (filgrastim). Version May 26, 2015. https://www.amgen.ca/Neupogen_PM.pdf. Accessed 6 Sept 6 2016
Hautmann MG, Hipp M, Kölbl O (2011) Clostridium difficile-associated diarrhea in radiooncology: an underestimated problem for the feasibility of the radiooncological treatment? Radiat Oncol 6:89
Bishop KD, Castillo JJ (2012) Risk factors associated with Clostridium difficile infection in adult oncology patients with a history of recent hospitalization for febrile neutropenia. Leuk Lymphoma 53:1617–1619
Masaoka T, Urabe A, Ohno R et al (1998) Evidence-based recommendations on antimicrobial use in febrile neutropenia in Japan. Int J Hematol 68(Suppl 1):S5–S6
Clinicaltrials.gov (2016) REaCT integrated consent model to compare two standard of care regimens NCT02173262. https://clinicaltrials.gov/ct2/show/NCT02173262. Accessed 9 Sept 2016
Hilton J, Mazzarello S, Fergusson D et al (2016) Novel methodology for comparing standard-of-care interventions in patients with cancer. J Oncol Pract. JOPR013474
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
This article does not contain any studies with human participants or animals performed by any of the authors.
Appendices
Appendix 1: Systematic review supplement: electronic literature search strategy
Database: Embase Classic + Embase <1947 to 2016 April 13>, Ovid MEDLINE(R) In-Process & Other Non-Indexed Citations and Ovid MEDLINE(R) <1946 to Present>.
Search Strategy:
1 | exp Breast Neoplasms/ |
2 | (breast adj2 (cancer$ or neoplasm$ or carcinoma$)).tw. |
3 | 1 or 2 |
4 | Granulocyte Colony-Stimulating Factor/ |
5 | Granulocyte-colony stimulating factor$.tw. |
6 | gcsf.tw. |
7 | g csf.tw. |
8 | Neupogen.tw. |
9 | Neulasta.tw. |
10 | PegFilgrastim.tw. |
11 | Filgrastim/ or Filgrastim.tw. |
12 | colony-stimulating factor 3.tw. |
13 | or/4–12 |
14 | 3 and 13 |
15 | exp Anti-Bacterial Agents/ and pc.fs. |
16 | exp Anti-Bacterial Agents/ and (prevention or prophyla$).tw. |
17 | Antibiotic Prophylaxis/ |
18 | (antibiotic$ or antibacterial$) adj5 (prevent$ or prophyla$)).tw. |
19 | (septra or Trimethoprim-Sulfamethoxazole or cipro or Ciprofloxacin or Moxifloxacin) adj5 (prophyla$ or prevent$)).tw. |
20 | or/15–19 |
21 | 3 and 20 |
22 | 14 or 21 |
23 | exp antineoplastic agents/ |
24 | Antineoplastic Combined Chemotherapy Protocols/ |
25 | Neoadjuvant Therapy/ |
26 | Cyclophosphamide.tw. |
27 | Fluorouracil.tw. |
28 | Epirubicin.tw. |
29 | Docetaxel.tw. |
30 | (Paclitaxel or Taxol).tw. |
31 | (Adriamycin or doxorubicin).tw. |
32 | Cytoxan.tw. |
33 | Methotrexate.tw. |
34 | (chemotherap$ or antineoplastic agent$).tw. |
35 | Chemotherapy, Adjuvant/ |
36 | or/23–35 |
37 | 22 and 36 |
PRESS EBC search submission
AMED | □ |
C2-SPCTRE | □ |
CINAHL | □ |
Cochrane Database of Systematic Reviews (CDSR; Cochrane Reviews) | □ |
Cochrane Central Register of Controlled Trials (CENTRAL; Clinical Trials) | □ |
Cochrane Methodology Register (CMR; Methods Studies) | □ |
Cochrane Library (all databases) | □ |
Database of Abstracts of Reviews of Effects (DARE; Other Reviews) | □ |
Embase | □ |
ERIC | □ |
ICTRP (International Clinical Trials Registry Platform) | □ |
LILACS (Latin American and Caribbean Health Sciences Literature) | □ |
MEDLINE | □ |
PreMEDLINE | □ |
PsycINFO | □ |
Other | □ |
Other | □ |
1. Translation of the research question | x |
2. Boolean and proximity operators | x |
3. Subject headings | x |
4. Natural language/free-text | x |
5. Spelling, syntax and line numbers | x |
6. Limits and filters | x |
7. Search strategy adaptations | x |
Appendix 2: Risk of bias assessment of Included Randomized Trials

Trials at low risk of bias (green), high risk of bias (red) or unclear risk of bias (yellow)
Green (−) = low risk bias
Red (+) = high risk bias
Yellow (?) = unclear risk of bias
Appendix 3: PRISMA Checklist
Section/topic | # | Checklist item | Reported on page # |
|---|---|---|---|
Title | |||
Title | 1 | Identify the report as a systematic review, meta-analysis, or both | 1 |
Abstract | |||
Structured summary | 2 | Provide a structured summary including, as applicable: background; objectives; data sources; study eligibility criteria, participants, and interventions; study appraisal and synthesis methods; results; limitations; conclusions and implications of key findings; systematic review registration number | 2,3 |
Introduction | |||
Rationale | 3 | Describe the rationale for the review in the context of what is already known | 4,5 |
Objectives | 4 | Provide an explicit statement of questions being addressed with reference to participants, interventions, comparisons, outcomes, and study design (PICOS) | 5 |
Methods | |||
Protocol and registration | 5 | Indicate if a review protocol exists, if and where it can be accessed (e.g., Web address), and, if available, provide registration information including registration number | N/A |
Eligibility criteria | 6 | Specify study characteristics (e.g., PICOS, length of follow-up) and report characteristics (e.g., years considered, language, publication status) used as criteria for eligibility, giving rationale | 6,7 |
Information sources | 7 | Describe all information sources (e.g., databases with dates of coverage, contact with study authors to identify additional studies) in the search and date last searched | 6,7 |
Search | 8 | Present full electronic search strategy for at least one database, including any limits used, such that it could be repeated | 18-22 |
Study selection | 9 | State the process for selecting studies (i.e., screening, eligibility, included in systematic review, and, if applicable, included in the meta-analysis) | 6,7 |
Data collection process | 10 | Describe method of data extraction from reports (e.g., piloted forms, independently, in duplicate) and any processes for obtaining and confirming data from investigators | 6,7 |
Data items | 11 | List and define all variables for which data were sought (e.g., PICOS, funding sources) and any assumptions and simplifications made | 6,7 |
Risk of bias in individual studies | 12 | Describe methods used for assessing risk of bias of individual studies (including specification of whether this was done at the study or outcome level), and how this information is to be used in any data synthesis | 7,28 |
Summary measures | 13 | State the principal summary measures (e.g., risk ratio, difference in means) | N/A |
Synthesis of results | 14 | Describe the methods of handling data and combining results of studies, if done, including measures of consistency (e.g., I2) for each meta-analysis | N/A |
Risk of bias across studies | 15 | Specify any assessment of risk of bias that may affect the cumulative evidence (e.g., publication bias, selective reporting within studies) | 7, 28 |
Additional analyses | 16 | Describe methods of additional analyses (e.g., sensitivity or subgroup analyses, meta-regression), if done, indicating which were pre-specified | N/A |
Results | |||
Study selection | 17 | Give numbers of studies screened, assessed for eligibility, and included in the review, with reasons for exclusions at each stage, ideally with a flow diagram | 6-7,18 |
Study characteristics | 18 | For each study, present characteristics for which data were extracted (e.g., study size, PICOS, follow-up period) and provide the citations | 6-9 |
Risk of bias within studies | 19 | Present data on risk of bias of each study and, if available, any outcome level assessment (see item 12) | 7,28 |
Results of individual studies | 20 | For all outcomes considered (benefits or harms), present, for each study: (a) simple summary data for each intervention group (b) effect estimates and confidence intervals, ideally with a forest plot | 6-110 |
Synthesis of results | 21 | Present results of each meta-analysis done, including confidence intervals and measures of consistency | N/A |
Risk of bias across studies | 22 | Present results of any assessment of risk of bias across studies (see Item 15) | 7,28 |
Additional analysis | 23 | Give results of additional analyses, if done (e.g., sensitivity or subgroup analyses, meta-regression [see Item 16]) | N/A |
Discussion | |||
Summary of evidence | 24 | Summarize the main findings including the strength of evidence for each main outcome; consider their relevance to key groups (e.g., healthcare providers, users, and policy makers) | 6-10 |
Limitations | 25 | Discuss limitations at study and outcome level (e.g., risk of bias), and at review-level (e.g., incomplete retrieval of identified research, reporting bias) | 10-13 |
Conclusions | 26 | Provide a general interpretation of the results in the context of other evidence, and implications for future research | 10-13 |
Funding | |||
Funding | 27 | Describe sources of funding for the systematic review and other support (e.g., supply of data); role of funders for the systematic review | N/A |
Rights and permissions
About this article
Cite this article
Fernandes, R., Mazzarello, S., Stober, C. et al. Optimal primary febrile neutropenia prophylaxis for patients receiving docetaxel–cyclophosphamide chemotherapy for breast cancer: a systematic review. Breast Cancer Res Treat 161, 1–10 (2017). https://doi.org/10.1007/s10549-016-4028-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10549-016-4028-0