Abstract
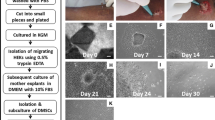
Recent studies have reported that stem cells can be isolated from a wide range of tissues including bone marrow, fatty tissue, adipose tissue and placenta. Moreover, several studies also suggest that skin dermis could serve as a source of stem cells, but are of unclear phenotype. Therefore, we isolated and investigated to determine the potential of stem cell within human skin dermis. We isolated cells from human dermis, termed here as human dermis-derived mesenchymal stem cells (hDMSCs) which is able to be isolated by using explants culture method. Our method has an advantage over the enzymatic method as it is easier, less expensive and less cell damage. hDMSCs were maintained in basal culture media and proliferation potential was measured. hDMSCs were highly proliferative and successfully expanded with no additional growth factor. In addition, hDMSCs revealed normal karyotype and expressed high levels of CD90, CD73 and CD105 while did not express the surface markers for CD34, CD45 and HLA-DR. Also, we confirmed that hDMSCs possess the capacity to differentiate into multiple lineage including adipocyte, osteocyte, chondrocyte and precursor of hepatocyte lineage. Considering these results, we suggest that hDMSCs might be a valuable source of stem cells and could potentially be a useful source of clinical application.






Similar content being viewed by others
References
Crigler L, Kazhanie A, Yoon TJ, Zakhari J, Anders J, Taylor B, Virador VM (2007) Isolation of a mesenchymal cell population from murine dermis that contains progenitors of multiple cell lineages. FASEB J 21:2050–2063
Dhanasekaran M, Indumathi S, Poojitha R, Kanmani A, Rajkumar JS, Sudarsanam D (2013) Plasticity and banking potential of cultured adipose tissue derived mesenchymal stem cells. Cell Tissue Bank 14:303–315
Gao Y, Bai C, Xiong H, Li Q, Shan Z, Huang L, Ma Y, Guan W (2013) Isolation and characterization of chicken dermis-derived mesenchymal stem/progenitor cells. Biomed Res Int 2013:626258
Giordano A, Galderisi U, Marino IR (2007) From the laboratory bench to the patient’s bedside: an update on clinical trials with mesenchymal stem cells. J Cell Physiol 211:27–35
Herzog EL, Chai L, Krause DS (2003) Plasticity of marrow-derived stem cells. Blood 102:3483–3493
Huard J, Cao B, Qu-Petersen Z (2003) Muscle-derived stem cells: potential for muscle regeneration. Birth Defects Res C Embryo Today 69:230–237
Kanazawa H, Fujimoto Y, Teratani T, Iwasaki J, Kasahara N, Negishi K, Tsuruyama T, Uemoto S, Kobayashi E (2011) Bone marrow-derived mesenchymal stem cells ameliorate hepatic ischemia reperfusion injury in a rat model. PLoS ONE 6:e19195
Kheolamai P, Dickson AJ (2009) Liver-enriched transcription factors are critical for the expression of hepatocyte marker genes in mES-derived hepatocyte-lineage cells. BMC Mol Biol 10:35
Kim SW, Han H, Chae GT, Lee SH, Bo S, Yoon JH, Lee YS, Lee KS, Park HK, Kang KS (2006) Successful stem cell therapy using umbilical cord blood-derived multipotent stem cells for Buerger’s disease and ischemic limb disease animal model. Stem Cells 24:1620–1626
Li Y, Chen J, Chen XG, Wang L, Gautam SC, Xu YX, Katakowski M, Zhang LJ, Lu M, Janakiraman N, Chopp M (2002) Human marrow stromal cell therapy for stroke in rat: neurotrophins and functional recovery. Neurology 59:514–523
Ogawa R, Mizuno H, Watanabe A, Migita M, Shimada T, Hyakusoku H (2004) Osteogenic and chondrogenic differentiation by adipose-derived stem cells harvested from GFP transgenic mice. Biochem Biophys Res Commun 313:871–877
Park JR, Jung JW, Lee YS, Kang KS (2008) The roles of Wnt antagonists Dkk1 and sFRP4 during adipogenesis of human adipose tissue-derived mesenchymal stem cells. Cell Prolif 41:859–874
Park SB, Seo KW, So AY, Seo MS, Yu KR, Kang SK, Kang KS (2012) SOX2 has a crucial role in the lineage determination and proliferation of mesenchymal stem cells through Dickkopf-1 and c-MYC. Cell Death Differ 19:534–545
Pountos I, Corscadden D, Emery P, Giannoudis PV (2007) Mesenchymal stem cell tissue engineering: techniques for isolation, expansion and application. Injury 38(Suppl 4):S23–S33
Riekstina U, Cakstina I, Parfejevs V, Hoogduijn M, Jankovskis G, Muiznieks I, Muceniece R, Ancans J (2009) Embryonic stem cell marker expression pattern in human mesenchymal stem cells derived from bone marrow, adipose tissue, heart and dermis. Stem Cell Rev 5:378–386
Shiojiri N, Lemire JM, Fausto N (1991) Cell lineages and oval cell progenitors in rat liver development. Cancer Res 51:2611–2620
Snykers S, De Kock J, Rogiers V, Vanhaecke T (2009) In vitro differentiation of embryonic and adult stem cells into hepatocytes: state of the art. Stem Cells 27:577–605
Studeny M, Marini FC, Dembinski JL, Zompetta C, Cabreira-Hansen M, Bekele BN, Champlin RE, Andreeff M (2004) Mesenchymal stem cells: potential precursors for tumor stroma and targeted-delivery vehicles for anticancer agents. J Natl Cancer Inst 96:1593–1603
Tilghman SM, Belayew A (1982) Transcriptional control of the murine albumin/alpha-fetoprotein locus during development. Proc Natl Acad Sci USA 79:5254–5257
Toma JG, Akhavan M, Fernandes KJ, Barnabe-Heider F, Sadikot A, Kaplan DR, Miller FD (2001) Isolation of multipotent adult stem cells from the dermis of mammalian skin. Nat Cell Biol 3:778–784
Tuli R, Seghatoleslami MR, Tuli S, Wang ML, Hozack WJ, Manner PA, Danielson KG, Tuan RS (2003) A simple, high-yield method for obtaining multipotential mesenchymal progenitor cells from trabecular bone. Mol Biotechnol 23:37–49
Vaculik C, Schuster C, Bauer W, Iram N, Pfisterer K, Kramer G, Reinisch A, Strunk D, Elbe-Burger A (2012) Human dermis harbors distinct mesenchymal stromal cell subsets. J Invest Dermatol 132:563–574
Yen BL, Huang HI, Chien CC, Jui HY, Ko BS, Yao M, Shun CT, Yen ML, Lee MC, Chen YC (2005) Isolation of multipotent cells from human term placenta. Stem Cells 23:3–9
Yu BD, Mukhopadhyay A, Wong C (2008) Skin and hair: models for exploring organ regeneration. Hum Mol Genet 17:R54–R59
Yu KR, Lee JY, Kim HS, Hong IS, Choi SW, Seo Y, Kang I, Kim JJ, Lee BC, Lee S, Kurtz A, Seo KW, Kang KS (2014) A p38 MAPK-mediated alteration of COX-2/PGE2 regulates immunomodulatory properties in human mesenchymal stem cell aging. PLoS ONE 9:e102426
Yucel G, Altindag B, Gomez-Ospina N, Rana A, Panagiotakos G, Lara MF, Dolmetsch R, Oro AE (2013) State-dependent signaling by Cav1.2 regulates hair follicle stem cell function. Genes Dev 27:1217–1222
Zuk PA, Zhu M, Mizuno H, Huang J, Futrell JW, Katz AJ, Benhaim P, Lorenz HP, Hedrick MH (2001) Multilineage cells from human adipose tissue: implications for cell-based therapies. Tissue Eng 7:211–228
Acknowledgments
This research was supported by the National Research Foundation of Korea (NRF) funded by the Ministry of Education (NRF-2013R1A1A2057787), (NRF-2014R1A2A2A01003737) and iPET (Korea Institute of Planning and Evaluation for Technology in Food, Agriculture, Forestry and Fisheries), Ministry of Food, Agriculture, Forestry and Fisheries (112008-5).
Conflict of interest
The authors have declared that there is no conflict of interests.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Park, JR., Kim, E., Yang, J. et al. Isolation of human dermis derived mesenchymal stem cells using explants culture method: expansion and phenotypical characterization. Cell Tissue Bank 16, 209–218 (2015). https://doi.org/10.1007/s10561-014-9471-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10561-014-9471-8