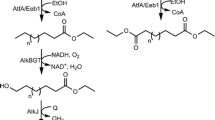
Abstract
Medium to long chain aliphatic amino alcohols have numerous applications in polymer-based industries. We herein report a biocatalytic reduction of amino fatty acids into corresponding amino alcohols using carboxylic acid reductase and E coli endogenous aldehyde reductases. Interestingly, employing this reaction in tandem with minimizing the hurdle of substrate insolubility, the biocatalysts successfully produced 5.76 mM 12-amino-1-dodecanol from 10 mM of corresponding 12-amino-dodecanoic acid. Finally, up to 9.17 mM of 8-amino-1-octanol was obtained in 24 h on a preparative scale reaction of 50 mM substrate.
Graphic Abstract





Similar content being viewed by others
References
Bozell JJ, Petersen GR (2010) Green Chem 12:539
Reetz MT (2013) J Am Chem Soc 135:12480
Lee JW, Kim HU, Choi S, Yi J, Lee SY (2011) Curr Opin Biotechnol 22:758
Biermann U, Bornscheuer U, Meier MA, Metzger JO, Schafer HJ (2011) Angew Chem Int Ed 50:3854
Patil MD, Grogan G, Bommarius A, Yun H (2018) ACS Catal 8:10985
Patil MD, Grogan G, Bommarius A, Yun H (2018) Catalysts 8:254
Sattler JH, Fuchs M, Mutti FG, Grischek B, Engel P, Pfeffer J, Woodley JM, Kroutil W (2014) Angew Chem Int Ed 53:14153
Seo JH, Lee SM, Lee J, Park JB (2015) J Biotechnol 216:158
Ahsan MM, Sung S, Jeon H, Patil MD, Chung T, Yun H (2018) Catalysts 8:4
Ahsan MM, Jeon H, Nadarajan S, Chung T, Yoo HW, Kim BG, Patil MD, Yun H (2018) Biotechnol J 13:1700562
Jang YS, Kim B, Shin JH, Choi YJ, Choi S, Song CW, Lee J, Park HG, Lee SY (2012) Biotechnol Bioeng 109:2437
Sung S, Jeon H, Sarak S, Ahsan MM, Patil MD, Kroutil W, Kim BG, Yun H (2018) Green Chem 20:4591
Akhtar MK, Turner NJ, Jones PR (2013) Proc Natl Acad Sci USA 110:87
Fedorchuk TP, Khusnutdinova AN, Evdokimova E, Flick R, Di Leo R, Stogios P, Savchenko A, Yakunin AF (2020) J Am Chem Soc 142:1038
Huang L, Sayoga GV, Hollmann F, Kara S (2018) ACS Catal 8:8680
Versteegen RM, Sijbesma RP, Meijer EW (1999) Angew Chem Int Ed 38:2917
Vera M, Almontassir A, Rodriguez-Galan A, Puiggali J (2003) Macromolecules 36:9784
Fernandez CE, Bermudez M, Versteegen RM, Meijer EW, Vancso GJ, Munoz-Guerra S (2010) Eur Polym J 46:2089
Khusnutdinova AN, Flick R, Popovic A, Brown G, Tchigvintsev A, Nocek B, Correia K, Joo JC, Mahadevan R, Yakunin AF (2017) Biotechnol J 12:1600751
Chung H, Yang JE, Ha JY, Chae TU, Shin JH, Gustavsson M, Lee SY (2015) Curr Opin Biotechnol 36:73
Napora-Wijata K, Strohmeier GA, Winkler M (2014) Biotechnol J 9:822
Sasano Y, Nagasawa S, Yamazaki M, Shibuya M, Park J, Iwabuchi Y (2014) Angew Chem Int Ed 53:3236
Bornscheuer UT, Huisman GW, Kazlauskas RJ, Lutz S, Moore JC, Robins K (2012) Nature 485:185
Alonso S, Rendueles M, Diaz M (2015) Crit Rev Biotechnol 35:497
Hollmann F, Arends IW, Holtmann D (2011) Green Chem 13:2285
Mutti FG, Knaus T, Scrutton NS, Breuer M, Turner NJ (2015) Science 349:1525
Resch V, Fabian WM, Kroutil W (2010) Adv Synth Catal 352:993
Song JW, Lee JH, Bornscheuer UT, Park JB (2014) Adv Synth Catal 356:1782
Voss CV, Gruber CC, Faber K, Knaus T, Macheroux P, Kroutil W (2008) J Am Chem Soc 130:13969
Schrewe M, Ladkau N, Buhler B, Schmid A (2013) Adv Synth Catal 355:1693
Qu G, Guo J, Yang D, Sun Z (2018) Green Chem 20:777
Winkler M (2018) Curr Opin Chem Biol 43:23
Kim GH, Jeon H, Khobragade TP, Patil MD, Sung S, Yoon S, Won Y, Sarak S, Yun H (2019) ChemCatChem 11:1437
Yoon S, Patil MD, Sarak S, Jeon H, Kim GH, Khobragade TP, Sung S, Yun H (2019) ChemCatChem 11:1898
Patil MD, Dev MJ, Shinde AS, Bhilare KD, Patel G, Chisti Y, Banerjee UC (2017) Process Biochem 63:113
Ahsan MM, Patil MD, Jeon H, Sung S, Chung T, Yun H (2018) Catalysts 8:400
Kim GH, Jeon H, Khobragade TP, Patil MD, Sung S, Yoon S, Won Y, Choi IS, Yun H (2019) Enzyme Microb Technol 120:52
Acknowledgements
This work was supported by the Ministry of Trade, Industry and Energy of South Korea (MOTIE, South Korea) under the industrial Technology Innovation Program, No. 10062550 and 10076343.The authors also acknowledge the funding from ‘Konkuk University Researcher Fund’ in 2019.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no financial or commercial conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Sarak, S., Jeon, H., Patil, M.D. et al. Enzymatic Synthesis of Aliphatic Primary ω-Amino Alcohols from ω-Amino Fatty Acids by Carboxylic Acid Reductase. Catal Lett 150, 3079–3085 (2020). https://doi.org/10.1007/s10562-020-03233-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10562-020-03233-9