Abstract
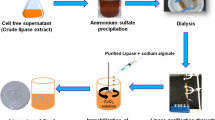
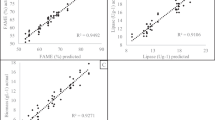
Herein, response surface methodology (RSM) was employed to optimize the bioprocess parameters for improved production of extracellular lipase by Aspergillus melleus and evaluated its biocatalytic potential for degradation of polyester vylon-200. Our previous report showed that pH, incubation time, temperature, and additional nitrogen source had significant effects on lipase biosynthesis. The variance analysis revealed that the established RSM model based on a central composite design for lipase production was significant (p < 0.0001, R2 = 0.9925). Under the optimized bioprocess conditions of pH 5.68, incubation time 96 h, temperature 30 °C, and diammonium tartrate as a nitrogen source, maximum lipase titer of 1346.87 U/gds was achieved, 1.92-fold higher than lipase yield in basal medium. The optimally synthesized cell-free lipase extract was partially purified by ammonium sulfate fractionation and dialysis and used to degrade polyester vylon 200. The degradation profile revealed that the lipolytic enzyme demonstrated excellent hydrolytic potential resulting in a 76% weight of polyester vylon-200. Differential scanning calorimetry revealed a noticeable decrease in the glass transition temperature of PV-200 (from 72.6 ºC to 63.9 ºC). Scanning electron microscopy envisaged various micron-scale cracks and holes on the surface of film after enzymatic treatment. Likewise, significant weight loss of the PV-200 films was also corroborated by FTIR analysis. This study's findings illustrate lipase's potential as a green and ecofriendly biocatalyst for robust polyester degradation and depolymerization.
Graphic Abstract


source were fixed at one level

source were fixed at one level

source and b temperature and nitrogen source on lipase production by A. melleus





Similar content being viewed by others
References
Chandra P, Singh R, Arora PK (2020) Microbial lipases and their industrial applications: a comprehensive review. Microb Cell Fact 19(1):1–42
Melani NB, Tambourgi EB, Silveira E (2020) Lipases: from production to applications. Sep Purif Rev 49(2):143–158
Contesini FJ, Davanço MG, Borin GP, Vanegas KG, Cirino JP, Melo RR, Mortensen UH, Hildén K, Campos DR, Carvalho PD (2020) Advances in recombinant lipases: production, engineering, immobilization and application in the pharmaceutical industry. Catalysts 10(9):1032
Mehta A, Guleria S, Sharma R, Gupta R (2020) The lipases and their applications with emphasis on food industry. Microbial biotechnology in food and health. Academic Press, USA, pp 143–164
Marques TA, Baldo C, Borsato D, Buzato JB, Celligoi MA (2014) Production and partial characterization of a thermostable, alkaline and organic solvent tolerant lipase from Trichoderma atroviride 676. Int J Sci Technol Res 3(5):77–83
Rehman S, Bhatti HN, Bilal M, Asgher M, Wang P (2017) Catalytic, kinetic and thermodynamic characteristics of an extracellular lipase from Penicillium notatum. Catal Lett 147(1):281–291
Sethi BK, Nanda PK, Sahoo S (2016) Characterization of biotechnologically relevant extracellular lipase produced by Aspergillus terreus NCFT 4269.10. Braz J Microbiol 47(1):143–149
Helal SE, Abdelhady HM, Abou-Taleb KA, Hassan MG, Amer MM (2017) Evaluation of factors affecting the fungal lipase production using one factor at a time approach and response surface methodology. Egypt J Microbiol 52(1):1–6
Asgher M, Khan SW, Bilal M (2016) Optimization of lignocellulolytic enzyme production by Pleurotus eryngii WC 888 utilizing agro-industrial residues and bio-ethanol production. Romanian Biotechnol Lett 21(1):11133
Amin F, Bhatti HN, Bilal M, Asgher M (2017) Multiple parameter optimizations for enhanced biosynthesis of exo-polygalacturonase enzyme and its application in fruit juice clarification. Int J Food Eng. https://doi.org/10.1515/ijfe-2016-0256
Mehta A, Sharma R, Gupta R (2019) Statistical optimization by response surface methodology to enhance lipase production by Aspergillus fumigatus. Open Microbiol J 13(1):86–93
Asgher M, Ijaz A, Bilal M (2016) Lignocellulose-degrading enzyme production by Pleurotus sapidus WC 529 and its application in lignin degradation. Turk J Biochem 41(1):26–36
Kumar DS, Ray S (2014) Fungal lipase production by solid state fermentation-an overview. J Anal Bioanal Tech 6(230):1
Rehman S, Bhatti HN, Bilal M, Asgher M (2019) Optimization of process variables for enhanced production of extracellular lipase by Pleurotus ostreatus IBL-02 in solid-state fermentation. Pak J Pharm Sci 32(2):617–624
Abdullah R, Qaiser H, Iftikhar T, Kaleem A, Iqtedar M, Saleem F, Haq I (2018) Application of response surface methodology for statistical optimization of lipase production by Penicilliumsp. employing solid state fermentation. Revista Mexicana de Ingeniería Química 17(3):863–875
Colla LM, Primaz AL, Benedetti S, Loss RA, de Lima M, Reinehr CO, Bertolin TE, Costa JA (2016) Surface response methodology for the optimization of lipase production under submerged fermentation by filamentous fungi. Braz J Microbiol 47(2):461–467
Açıkel Ü, Erşan M, Açıkel YS (2010) Optimization of critical medium components using response surface methodology for lipase production by Rhizopus delemar. Food Bioprod Process 88(1):31–39
Peng H, Tan J, Bilal M, Wang W, Hu H, Zhang X (2018) Enhanced biosynthesis of phenazine-1-carboxamide by Pseudomonas chlororaphis strains using statistical experimental designs. World J Microbiol Biotechnol 34(9):129
Jia J, Yang X, Wu Z, Zhang Q, Lin Z, Guo H, Lin CS, Wang J, Wang Y (2015) Optimization of fermentation medium for extracellular lipase production from Aspergillus niger using response surface methodology. Biomed Res Int 20:2015
Gururaj P, Ramalingam S, Devi GN, Gautam P (2016) Process optimization for production and purification of a thermostable, organic solvent tolerant lipase from Acinetobacter sp. AU07. Braz J Microbiol 47(3):647–657
Asgher M, Wahab A, Bilal M, Iqbal HM (2016) Lignocellulose degradation and production of lignin modifying enzymes by Schizophyllum commune IBL-06 in solid-state fermentation. Biocatal Agric Biotechnol 1(6):195–201
Lanka S, Latha JN (2015) Response surface methodology as a statistical tool for fermentation media optimization in lipase production by palm oil mill effluent (POME) isolate Emericella nidulans NFCCI 3643. Methodology (RSM) 4(4):2535–2545
Lo CF, Yu CY, Kuan IC, Lee SL (2012) Optimization of lipase production by Burkholderiasp. using response surface methodology. Int J Mol Sci 13(11):14889–14897
Papagora C, Roukas T, Kotzekidou P (2013) Optimization of extracellular lipase production by Debaryomyces hansenii isolates from dry-salted olives using response surface methodology. Food Bioprod Process 91(4):413–420
Danso D, Schmeisser C, Chow J, Zimmermann W, Wei R, Leggewie C, Li X, Hazen T, Streit WR (2018) New insights into the function and global distribution of polyethylene terephthalate (PET)-degrading bacteria and enzymes in marine and terrestrial metagenomes. Appl Environ Microbiol 84(8):1–13
Gan Z, Zhang H (2019) PMBD: a comprehensive plastics microbial biodegradation database. Database 2019:1–11
Yagiz F, Kazan D, Akin AN (2007) Biodiesel production from waste oils by using lipase immobilized on hydrotalcite and zeolites. Chem Eng J 134(1–3):262–267
Bradford MM (1976) A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem 72(1–2):248–254
Bhatti HN, Asgher M, Abbas A, Nawaz R, Sheikh MA (2006) Studies on kinetics and thermostability of a novel acid invertase from Fusarium solani. J Agric Food Chem 54(13):4617–4623
Umare SS, Chandure AS (2008) Synthesis, characterization and biodegradation studies of poly (ester urethane) s. Chem Eng J 142(1):65–77
Behera AR, Veluppal A, Dutta K (2019) Optimization of physical parameters for enhanced production of lipase from Staphylococcus hominis using response surface methodology. Environ Sci Pollut Res 26(33):34277–34284
Haider MA, Pakshirajan K (2007) Screening and optimization of media constituents for enhancing lipolytic activity by a soil microorganism using statistically designed experiments. Appl Biochem Biotechnol 141(2–3):377–390
Bezerra MA, Santelli RE, Oliveira EP, Villar LS, Escaleira LA (2008) Response surface methodology (RSM) as a tool for optimization in analytical chemistry. Talanta 76(5):965–977
Gangadharan D, Sivaramakrishnan S, Nampoothiri KM, Pandey A (2006) Solid culturing of Bacillus amyloliquefaciens for α-amylase production. Food Technol Biotechnol 44(2):269–274
Faisal PA, Hareesh ES, Priji P, Unni KN, Sajith S, Sreedevi S, Josh MS, Benjamin S (2014) Optimization of parameters for the production of lipase from Pseudomonassp. BUP6 by solid state fermentation. Adv Enzyme Res 2(04):125
Amin F, Bhatti HN, Bhatti IA, Asgher M (2013) Utilization of wheat bran for enhanced production of exopolygalacturonase by Penicillium notatum using response surface methodology. Pak J Agric Sci 50(3):469–477
Shi K, Jing J, Song L, Su T, Wang Z (2020) Enzymatic hydrolysis of polyester: degradation of poly (ε-caprolactone) by Candida antarctica lipase and Fusarium solani cutinase. Int J Biol Macromol 1(144):183–189
Hwang Y, Ree M, Kim H (2006) Enzymatic degradation of poly (propylene carbonate) and poly (propylene carbonate-co-ɛ-caprolactone) synthesized via CO2 fixation. Catal Today 115(1–4):288–289
Acknowledgements
The authors are thankful to the Higher Education Commission (HEC) of Pakistan for financial assistance under the Indigenous Ph.D. 5000 Scholarship Program.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that there is no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Amin, M., Bhatti, H.N., Sadaf, S. et al. Enhancing Lipase Biosynthesis by Aspergillus Melleus and its Biocatalytic Potential for Degradation of Polyester Vylon-200. Catal Lett 151, 2257–2271 (2021). https://doi.org/10.1007/s10562-020-03476-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10562-020-03476-6