Abstract
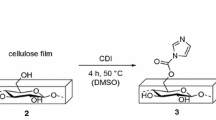
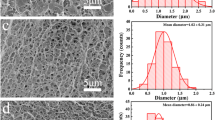
Scavenging carbonylated proteins that are toxic substances inducing various diseases attract the most attention in the field of blood purification. In our study, functionalized cellulose beads modified by carbazate groups as affinity ligands of carbonyls were prepared for specific scavenging of carbonylated proteins based on the carbazate-aldehyde/ ketone reaction. Results showed that the carbazate-functionalized cellulose beads with different degrees of substitution (DS) maintained the original multi-porous structure with the diameter of about 50 μm and thermal stability. Spectroscopic and gel electrophoresis analysis indicated that the modified cellulose beads could effectively scavenge carbonylated proteins and the scavenging effects increased with the increase of DS. Thus, the carbazate functionalized cellulose beads may be developed as fillers in the column for the scavenging of carbonylated proteins.








Similar content being viewed by others
References
Aldini G, Dalle-Donne I, Facino RM, Milzani A, Carini M (2007) Intervention strategies to inhibit protein carbonylation by lipoxidation-derived reactive carbonyls. Med Res Rev 27:817–868. https://doi.org/10.1002/med.20073
Bethell GS, Ayers JS, Hearn MTW, Hancock WS (1987) Investigation of the activation of various insoluble polysaccharides with 1,1′-carbonyldiimidazole and of the properties of the activated matrices. J Chromatogr A 219:361–371. https://doi.org/10.1016/S0021-9673(00)80379-9
Baker G, Matveychuk D, MacKenzie EM, Holt A, Wang Y, Kar S (2019) Attenuation of the effects of oxidative stress by the MAO-inhibiting antidepressant and carbonyl scavenger phenelzine. Chem Biol Interact 304:139–147. https://doi.org/10.1016/j.cbi.2019.03.003
Chen J et al (2017) Non-ionic macroporous polystyrene adsorbents for removal of serum toxins in liver failure by hemoperfusion Artif Cells Nanomed. Biotechnol 45:174–183. https://doi.org/10.3109/21691401.2016.1138494
Dibbert N, Krause A, Rios-Camacho JC, Gruh I, Kirschning A, Drager G (2016) A synthetic toolbox for the in situ formation of functionalized homo- and heteropolysaccharide-based hydrogel libraries. Chemistry 22:18777–18786. https://doi.org/10.1002/chem.201603748
Fedorova M, Bollineni RC, Hoffmann R (2014) Protein carbonylation as a major hallmark of oxidative damage: update of analytical strategies. Mass Spectrom Rev 33:79–97. https://doi.org/10.1002/mas.21381
Fernando N et al (2016) Protein carbonyl as a biomarker of oxidative stress in severe leptospirosis, and its usefulness in differentiating leptospirosis from dengue infections. PLoS ONE 11:e0156085. https://doi.org/10.1371/journal.pone.0156085
Girardot T, Schneider A, Rimmelé T (2019) Blood purification techniques for sepsis and septic aki. Semin Nephrol 39:505–514. https://doi.org/10.1016/j.semnephrol.2019.06.010
Hauck AK, Huang Y, Hertzel AV, Bernlohr DA (2019) Adipose oxidative stress and protein carbonylation. J Biol Chem 294:1083–1088
Hecker M, Wagner AH (2018) Role of protein carbonylation in diabetes. J Inherit Metab Dis 41:29–38. https://doi.org/10.1007/s10545-017-0104-9
Kolawole TA, Ilochi ON, Oluwatayo BO, Chuemere AN, Dapper DV (2019) Tissue protein carbonylation in aging: A strategic analysis of age-related protein modification. J Adv Med Pharm Sci 20:1–5. https://doi.org/10.9734/jamps/2019/v20i330111
Kolmel DK, Kool ET (2017) Oximes and hydrazones in bioconjugation: mechanism and catalysis. Chem Rev 117:10358–10376. https://doi.org/10.1021/acs.chemrev.7b00090
Kecili R, Nivhede D, Billing J, Leeman M, Sellergren B, Yilmaz E (2012) Removal of acrolein from active pharmaceutical ingredients using aldehyde scavengers. Org Process Res Dev 16:1225–1229. https://doi.org/10.1021/op3000459
Li Y, Han M, Wang Y, Liu Q, Zhao W, Su B, Zhao C (2018) A mussel-inspired approach towards heparin-immobilized cellulose gel beads for selective removal of low density lipoprotein from whole blood. Carbohydr Polym 202:116–124. https://doi.org/10.1016/j.carbpol.2018.08.118
Lanzillotto M, Konnert L, Lamaty F, Martinez J, Colacino E (2015) Mechanochemical 1,1′-carbonyldiimidazole-mediated synthesis of carbamates. ACS Sustain Chem Eng 3:2882–2889. https://doi.org/10.1021/acssuschemeng.5b0081
Malik DJ et al (2009) Synthesis and characterization of size-selective nanoporous polymeric adsorbents for blood purification. Sep Purif Technol 66:578–585. https://doi.org/10.1016/j.seppur.2009.01.016
Ngamcherdtrakul W, Morry J, Sangvanich T, Reda M, Bejan DS, Fryxell GE, Yantasee W (2019) Removal of a gadolinium based contrast agent by a novel sorbent hemoperfusion in a chronic kidney disease (ckd) rodent model. Sci Rep 9:709. https://doi.org/10.1038/s41598-018-37348-2
Oommen OP, Wang SJ, Kisiel M, Sloff M, Hilborn J, Varghese OP (2013) Smart design of stable extracellular matrix mimetic hydrogel: Synthesis, characterization, and in vitro and in vivo evaluation for tissue engineering. Adv Funct Mater 23:1273–1280. https://doi.org/10.1002/adfm.201201698
Puleo DA, Kissling RA, Sheu MS (2002) A technique to immobilize bioactive proteins, includingbone morphogenetic protein-4 (BMP-4), on titanium alloy. Biomaterials 23(9):2079–2087. https://doi.org/10.1016/s0142-9612(01)00339-8
Peng B et al (2019) Tuned cationic dendronized polymer: Molecular scavenger for rheumatoid arthritis treatment. Angew Chem Int Ed Engl 58:4254–4258. https://doi.org/10.1002/anie.201813362
Rani V, Deep G, Singh RK, Palle K, Yadav UCS (2016) Oxidative stress and metabolic disorders: pathogenesis and therapeutic strategies. Life Sci 148:183–193. https://doi.org/10.1016/j.lfs.2016.02.002
Rocha I, Ferraz N, Mihranyan A, Strømme M, Lindh J (2018) Sulfonated nanocellulose beads as potential immunosorbents. Cellulose 25:1899–1910. https://doi.org/10.1007/s10570-018-1661-2
Ruan CQ, Stromme M, Lindh J (2018) Preparation of porous 2,3-dialdehyde cellulose beads crosslinked with chitosan and their application in adsorption of Congo red dye. Carbohydr Polym 181:200–207. https://doi.org/10.1016/j.carbpol.2017.10.072
Su X, Aprahamian I (2014) Hydrazone-based switches, metallo-assemblies and sensors. Chem Soc Rev 43:1963–1981. https://doi.org/10.1039/c3cs60385g
Sharma PK, Singh Y (2019) Glyoxylic hydrazone linkage-based peg hydrogels for covalent entrapment and controlled delivery of doxorubicin. Biomacromol 20:2174–2184. https://doi.org/10.1021/acs.biomac.9b00020
Yu Y (2013) Adsorbents in blood purification: from lab search to clinical therapy. Chin Sci Bul 558:4357–4361
Yang YF, Gao M, Zhou B, Cai PA, Larsson TE, Zhao JM, Bowden TM (2020) Weak acidic stable carbazate modified cellulose membranes target for scavenging carbonylated proteins in hemodialysis. Carbohydr polym 231:115727. https://doi.org/10.1016/j.carbpol.2019.115727
Yang XF et al (2017) Highly efficient self-healable and dual responsive cellulose-based hydrogels for controlled release and 3d cell culture. Adv Funct Mater. https://doi.org/10.1002/adfm.201703174
Yamamoto S et al (2018) Adsorption of protein-bound uremic toxins through direct hemoperfusion with hexadecyl-immobilized cellulose beads in patients undergoing hemodialysis. Artif Organs 42:88–93. https://doi.org/10.1111/aor.12961
Zhou B et al. (2020) Carbazate modified dextrans as scavengers for carbonylated proteins. Carbohydrate Polymers 232: 115802. http://www.sciencedirect.com/science/article/pii/S0144861719314705
Acknowledgments
This study was financially supported by National Key R&D Program of China (2018YFC1105900), the Guangxi Science and Technology Major Project (Guike.AA19254002), the Natural Science Foundation of Guangxi (2020GXNSFAA159134), the Seventh batch of special experts in Guangxi (Professor Wei Yao), Youth Science Foundation of college of pharmacy of Guangxi Medical University (No. YXY[2019]10- 15) and the Distinguished Young Scholars Program of Guangxi Medical University, and the Youth Science Foundation of Guangxi Medical University (GXMUYSF201905).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical statement and informed consent
This article does not contain any studies with human participants or animals performed by any of the authors. In this experiment, we did not collect any samples of human and animals. All authors declared that there were no ethical problems in this article.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Huang, L., Zhou, X., Feng, X. et al. Carbazate functionalized cellulose beads as potential scavengers specific for carbonylated proteins. Cellulose 28, 3029–3039 (2021). https://doi.org/10.1007/s10570-020-03659-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10570-020-03659-9