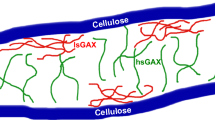
Abstract
The interaction of xylan, an abundant plant polysaccharide, with cellulose microfibrils is essential for secondary cell wall strength. A deeper understanding of these interactions is crucial both to improve our understanding of plant cell wall architecture and to design alternate strategies to overcome cellulose recalcitrance for the production of biofuels and sustainable biomaterials. Naturally occurring acetate or glucuronic acid substitutions on xylan have been shown to influence xylan-cellulose interactions. Here, we use unrestrained molecular dynamics simulations to determine the interactions with the (110) hydrophilic face of cellulose fibers of four different xylans. In the absence of cellulose, all xylans, independent of the substitution pattern, adopt a highly flexible threefold helical screw conformation. However, when xylan is spatially close to a cellulose surface 1,2 linked acetyl xylans (2AcX) adopt rigid twofold helical screw conformations. The 2AcX conformations are primarily stabilized by interactions between the acetylated oxygen and the glycosidic linkage with C-O6 of cellulose. In contrast, the glycosidic oxygens and acetyl decorations for 1,3 linked acetyl groups (3AcX) are oriented away from the cellulose surface and the 3AcX xylans maintain threefold helical screw conformations on the cellulose surface. Our results show that evenly spaced chemical functionalization (with acetyl groups) and the position of substitution (1,2) on xylan backbone play key roles in tuning the xylan-cellulose interactions to stabilize the twofold helical screw conformations of xylan on the cellulose surface. A comparison with previous experimental findings further suggests that 1,2 substitutions induce twofold helical screw conformations of xylan on the cellulose surface irrespective of the chemical nature of the substituent, while 1,3 substitutions primarily bind lignin in threefold helical screw conformations rather than cellulose in plant cell walls.











Similar content being viewed by others
References
Abraham MJ, Murtola T, Schulz R et al (2015) Gromacs: high performance molecular simulations through multi-level parallelism from laptops to supercomputers. SoftwareX 1–2:19–25. https://doi.org/10.1016/j.softx.2015.06.001
Almond A, Sheenan JK (2003) Predicting the molecular shape of polysaccharides from dynamic interactions with water. Glycobiology 13:255–264. https://doi.org/10.1093/glycob/cwg031
Bergenstråhle M, Wohlert J, Larsson PT et al (2008) Dynamics of cellulose−water interfaces: NMR spin−lattice relaxation times calculated from atomistic computer simulations. J Phys Chem B 112:2590–2595
Berglund J, Angles d’Ortoli T, Vilaplana F et al (2016) A molecular dynamics study of the effect of glycosidic linkage type in the hemicellulose backbone on the molecular chain flexibility. Plant J 88:56–70. https://doi.org/10.1111/tpj.13259
Berglund J, Kishani S, Morais de Carvalho D et al (2020) Acetylation and sugar composition influence the (in)solubility of plant β-mannans and their interaction with cellulose surfaces. ACS Sustain Chem Eng 8:10027–10040. https://doi.org/10.1021/acssuschemeng.0c01716
Biely P, Mackenzie CR (1986) Cooperativity of esterases and xylanases in the enzymatic degradation of acetyl xylan. Nat Biotechnol 4:731–733. https://doi.org/10.1038/nbt0886-731
Bischoff V, Nita S, Neumetzler L et al (2010) TRICHOME BIREFRINGENCE and its homolog AT5G01360 encode plant-specific DUF231 proteins required for cellulose biosynthesis in arabidopsis. Plant Physiol 153:590–602. https://doi.org/10.1104/pp.110.153320
Bosmans TJ, Stépán AM, Toriz G et al (2014) Assembly of debranched xylan from solution and on nanocellulosic surfaces. Biomacromol 15:924–930. https://doi.org/10.1021/bm4017868
Bromley JR, Busse-Wicher M, Tryfona T et al (2013) GUX1 and GUX2 glucuronyltransferases decorate distinct domains of glucuronoxylan with different substitution patterns. Plant J 74:423–434. https://doi.org/10.1111/tpj.12135
Brown DM, Goubet F, Wong VW et al (2007) Comparison of five xylan synthesis mutants reveals new insight into the mechanisms of xylan synthesis. Plant J 52:1154–1168. https://doi.org/10.1111/j.1365-313X.2007.03307.x
Busse-Wicher M, Gomes TCF, Tryfona T et al (2014) The pattern of xylan acetylation suggests xylan may interact with cellulose microfibrils as a twofold helical screw in the secondary plant cell wall of Arabidopsis thaliana. Plant J 79:492–506. https://doi.org/10.1111/tpj.12575
Busse-Wicher M, Li A, Silveira RL et al (2016) Evolution of xylan substitution patterns in gymnosperms and angiosperms: implications for xylan interaction with cellulose. Plant Physiol 171:2418–2431. https://doi.org/10.1104/pp.16.00539
Bussi G, Donadio D, Parrinello M (2007) Canonical sampling through velocity rescaling. J Chem Phys 126:014101. https://doi.org/10.1063/1.2408420
Carroll A, Somerville C (2009) Cellulosic biofuels. Annu Rev Plant Biol 60:165–182. https://doi.org/10.1146/annurev.arplant.043008.092125
Chong SL, Virkki L, Maaheimo H et al (2014) O-Acetylation of glucuronoxylan in arabidopsis thaliana wild type and its change in xylan biosynthesis mutants. Glycobiology 24:494–506. https://doi.org/10.1093/glycob/cwu017
Chundawat SPS, Beckham GT, Himmel ME, Dale BE (2011) Deconstruction of lignocellulosic biomass to fuels and chemicals. Annu Rev Chem Biomol Eng 2:121–145. https://doi.org/10.1146/annurev-chembioeng-061010-114205
Cosgrove D (2014) Re-constructing our models of cellulose and primary cell wall assembly. Curr Opin Plant Biol 22:122–131
Cosgrove DJ, Jarvis MC (2012) Comparative structure and biomechanics of plant primary and secondary cell walls. Plant Sci, Front. https://doi.org/10.3389/fpls.2012.00204
Darden T, York D, Pedersen L (1993) Particle mesh Ewald: an N⋅log( N ) method for Ewald sums in large systems. J Chem Phys 98:10089–10092. https://doi.org/10.1063/1.464397
de Carvalho DM, Berglund J, Marchand C et al (2019) Improving the thermal stability of different types of xylan by acetylation. Carbohydr Polym 220:132–140. https://doi.org/10.1016/j.carbpol.2019.05.063
Ebringerova A, Heinze T (2000) Xylan and xylan derivatives – biopolymers with valuable properties, 1. Naturally occurring xylans structures, isolation procedures and properties. Macromol Rapid Commun 21:869–883. https://doi.org/10.1002/1521-3927
Evtuguin DV, Tomás JL, Silva AMS, Neto CP (2003) Characterization of an acetylated heteroxylan from Eucalyptus globulus Labill. Carbohydr Res 338:597–604. https://doi.org/10.1016/S0008-6215(02)00529-3
Falcoz-Vigne L, Ogawa Y, Molina-Boisseau S et al (2017) Quantification of a tightly adsorbed monolayer of xylan on cellulose surface. Cellulose 24:3725–3739. https://doi.org/10.1007/s10570-017-1401-z
French AD, Johnson GP (2009) Cellulose and the twofold screw axis: modeling and experimental arguments. Cellulose 16:959–973. https://doi.org/10.1007/s10570-009-9347-4
Gao Y, He C, Zhang D et al (2017) Two trichome birefringence-like proteins mediate xylan acetylation, which is essential for leaf blight resistance in rice. Plant Physiol 173:470–481. https://doi.org/10.1104/pp.16.01618
Gomes TCF, Skaf MS (2012) Cellulose-builder: a toolkit for building crystalline structures of cellulose. J Comput Chem 33:1338–1346. https://doi.org/10.1002/jcc.22959
Grantham NJ, Wurman-Rodrich J, Terrett OM et al (2017) An even pattern of xylan substitution is critical for interaction with cellulose in plant cell walls. Nat Plants 3:859–865. https://doi.org/10.1038/s41477-017-0030-8
Guvench O, Hatcher E, Venable RM et al (2009) CHARMM additive all-atom force field for glycosidic linkages between hexopyranoses. J Chem Theory Comput 5:2353–2370. https://doi.org/10.1021/ct900242e
Hess B (2008) P-LINCS: a parallel linear constraint solver for molecular simulation. J Chem Theory Comput 4:116–122. https://doi.org/10.1021/ct700200b
Hess B, Bekker H, Berendsen HJC, Fraaije JGEM (1997) LINCS: a linear constraint solver for molecular simulations. J Comput Chem 18:1463–1472. https://doi.org/10.1002/(SICI)1096-987X(199709)18:12%3c1463::AID-JCC4%3e3.0.CO;2-H
Himmel ME, Ding S, Johnson DK, Adney WS (2007) Biomass recalcitrance: engineering plants and enzymes for biofuels production. Science 315:804–808s
Humphrey W, Dalke A, Schulten K (1996) VMD: visual molecular dynamics. J Mol Graph 14:33–38. https://doi.org/10.1016/0263-7855(96)00018-5
Jaafar Z, Mazeau K, Boissière A et al (2019) Meaning of xylan acetylation on xylan-cellulose interactions: a quartz crystal microbalance with dissipation (QCM-D) and molecular dynamic study. Carbohydr Polym 226:115315. https://doi.org/10.1016/j.carbpol.2019.115315
Jo S, Kim T, Iver V, Im W (2008) CHARMM-GUI: a web- based graphical user interface for CHARMM. J Comput Chem 29:1859–1865
Jo S, Song KC, Desaire H et al (2011) Glycan reader: Automated sugar identification and simulation preparation for carbohydrates and glycoproteins. J Comput Chem 32:3135–3141. https://doi.org/10.1002/jcc.21886
Johnson AM, Kim H, Ralph J, Mansfield SD (2017) Natural acetylation impacts carbohydrate recovery during deconstruction of Populus trichocarpa wood. Biotechnol Biofuels 10:1–12. https://doi.org/10.1186/s13068-017-0734-z
Jorgensen WL, Chandrasekhar J, Madura JD et al (1983) Comparison of simple potential functions for simulating liquid water comparison of simple potential functions for simulating liquid water. J Chem Phys 79:926. https://doi.org/10.1063/1.445869
Kabel MA, van den Borne H, Vincken JP et al (2007) Structural differences of xylans affect their interaction with cellulose. Carbohydr Polym 69:94–105. https://doi.org/10.1016/j.carbpol.2006.09.006
Kang X, Kirui A, Dickwella Widanage MC et al (2019) Lignin-polysaccharide interactions in plant secondary cell walls revealed by solid-state NMR. Nat Commun 10:347. https://doi.org/10.1038/s41467-018-08252-0
Koutaniemi S, Guillon F, Tranquet O et al (2012) Substituent-specific antibody against glucuronoxylan reveals close association of glucuronic acid and acetyl substituents and distinct labeling patterns in tree species. Planta 236:739–751. https://doi.org/10.1007/s00425-012-1653-7
Kubicki JD, Yang H, Sawada D et al (2018) The shape of native plant cellulose microfibrils. Sci Rep 8:13983. https://doi.org/10.1038/s41598-018-32211-w
Kumar R, Bhagia S, Smith MD et al (2018) Cellulose-hemicellulose interactions at elevated temperatures increase cellulose recalcitrance to biological conversion. Green Chem 20:921–934. https://doi.org/10.1039/c7gc03518g
Larsson PT (2003) Interaction between cellulose I and hemicelluloses studied by spectral fitting of CP/MAS 13C-NMR spectra. ACS Symp Ser Hemicelluloses Sci Technol 864:254–268
Larsson PT, Hult EL, Wickholm K et al (1999) CP/MAS 13C-NMR spectroscopy applied to structure and interaction studies on cellulose I. Solid State Nucl Magn Reson 15:31–40. https://doi.org/10.1016/S0926-2040(99)00044-2
Linder A, Bergman R, Bodin A, Gatenholm P (2003) Mechanism of assembly of xylan onto cellulose surfaces. Langmuir 19:5072–5077. https://doi.org/10.1021/la0341355
Ling Z, Edwards JV, Nam S et al (2020) Conformational analysis of xylobiose by DFT quantum mechanics. Cellulose 27:1207–1224. https://doi.org/10.1007/s10570-019-02874-3
Lunin V, Wang H-T, Bharadwaj V et al (2020) Molecular mechanism of polysaccharide acetylation by the Arabidopsis xylan O-acetyltransferase XOAT1. Plant Cell 32:2367–2382. https://doi.org/10.1105/tpc.20.00028
Luzar A (2000) Resolving the hydrogen bond dynamics conundrum. J Chem Phys 113:10663–10675. https://doi.org/10.1063/1.1320826
Martínez-Abad A, Berglund J, Toriz G et al (2017) Regular motifs in xylan modulate molecular flexibility and interactions with cellulose surfaces. Plant Physiol 175:1579–1592. https://doi.org/10.1104/pp.17.01184
Martínez-abad A, Jiménez-quero A (2020) Influence of the molecular motifs of mannan and xylan populations on their recalcitrance and organization in spruce softwoods. Green Chem 22:3956–3970. https://doi.org/10.1039/d0gc01207f
Mazeau K, Moine C, Krausz P, Gloaguen V (2005) Conformational analysis of xylan chains. Carbohydr Res 340:2752–2760. https://doi.org/10.1016/j.carres.2005.09.023
Meng X, Ragauskas AJ (2014) Recent advances in understanding the role of cellulose accessibility in enzymatic hydrolysis of lignocellulosic substrates. Curr Opin Biotechnol 27:150–158. https://doi.org/10.1016/j.copbio.2014.01.014
Mikkelsen D, Flanagan BM, Wilson SM et al (2015) Interactions of arabinoxylan and (1,3)(1,4)-β-glucan with cellulose networks. Biomacromol 16:1232–1239. https://doi.org/10.1021/acs.biomac.5b00009
Mortimer JC, Miles GP, Brown DM et al (2010) Absence of branches from xylan in Arabidopsis gux mutants reveals potential for simplification of lignocellulosic biomass. Proc Nat Acad Sci USA 107:17409–17414. https://doi.org/10.1073/pnas.1005456107
Nieduszynski A, Marchessault H (1972) Structure of beta, D (1–4’)-Xylan Hydrate. Biopolymers 11:1335–1344
Nishiyama Y, Langan P, Chanzy H (2002) Crystal structure and hydrogen-bonding system in cellulose Iβ from synchrotron X-ray and neutron fiber diffraction. J Am Chem Soc 124:9074–9082. https://doi.org/10.1021/ja0257319
Park SJ, Lee J, Patel DS et al (2017) Glycan reader is improved to recognize most sugar types and chemical modifications in the protein data bank. Bioinformatics 33:3051–3057. https://doi.org/10.1093/bioinformatics/btx358
Park SJ, Lee J, Qi Y et al (2019) CHARMM-GUI Glycan Modeler for modeling and simulation of carbohydrates and glycoconjugates. Glycobiology 29:320–331. https://doi.org/10.1093/glycob/cwz003
Parrinello M, Rahman A (1981) Polymorphic transitions in single crystals: A new molecular dynamics method. J Appl Phys 52:7182–7190. https://doi.org/10.1063/1.328693
Pauly M, Keegstra K (2010) Plant cell wall polymers as precursors for biofuels. Curr Opin Plant Biol 13:304–311. https://doi.org/10.1016/j.pbi.2009.12.009
Pawar PMA, Koutaniemi S, Tenkanen M, Mellerowicz EJ (2013) Acetylation of woody lignocellulose: significance and regulation. Front Plant Sci 4:1–8. https://doi.org/10.3389/fpls.2013.00118
Pereira CS, Silveira RL, Dupree P, Skaf MS (2017) Effects of xylan side-chain substitutions on xylan-cellulose interactions and implications for thermal pretreatment of cellulosic biomass. Biomacromol 18:1311–1321. https://doi.org/10.1021/acs.biomac.7b00067
Qaseem MF, Wu AM (2020) Balanced xylan acetylation is the key regulator of plant growth and development, and cell wall structure and for industrial utilization. Int J Mol Sci 21:7875. https://doi.org/10.3390/ijms21217875
Van RR, Nieves IU, Shanmugam KT et al (2018) Techno-economic evaluation of cellulosic ethanol production based on pilot biorefinery data: a case study of sweet sorghum bagasse processed via L + SScF. Bioenergy Res 11:414–425
Scheller HV, Ulvskov P (2010) Hemicelluloses. Annu Rev Plant Biol 61:263–289. https://doi.org/10.1146/annurev-arplant-042809-112315
Shrestha UR, Smith S, Pingali SV et al (2019) Arabinose substitution effect on xylan rigidity and self-aggregation. Cellulose 26:2267–2278. https://doi.org/10.1007/s10570-018-2202-8
Simmons TJ, Mortimer JC, Bernardinelli OD et al (2016) Folding of xylan onto cellulose fibrils in plant cell walls revealed by solid-state NMR. Nat Commun 7:13902. https://doi.org/10.1038/ncomms13902
Song B, Zhao S, Shen W et al (2020) Direct measurement of plant cellulose microfibril and bundles in native cell walls. Front Plant Sci 11:1–11. https://doi.org/10.3389/fpls.2020.00479
Tarasov D, Leitch M, Fatehi P (2018) Lignin-carbohydrate complexes: properties, applications, analyses, and methods of extraction: a review. Biotechnol Biofuels 11:269. https://doi.org/10.1186/s13068-018-1262-1
Teleman A, Tenkanen M, Jacobs A, Dahlman O (2002) Characterization of O-acetyl-(4-O-methylglucurono)xylan isolated from birch and beech. Carbohydr Res 337:373–377. https://doi.org/10.1016/S0008-6215(01)00327-5
Thomas LH, Forsyth VT, Adriana Š et al (2013) Structure of cellulose microfibrils in primary cell walls from collenchyma. Plant Physiol 161:465–476. https://doi.org/10.1104/pp.112.206359
Torshin IY, Weber IT, Harrison RW (2002) Geometric criteria of hydrogen bonds in proteins and identification of “bifurcated” hydrogen bonds. Protein Eng 15:359–363. https://doi.org/10.1093/protein/15.5.359
Westbye P, Svanberg C, Gatenholm P (2006) The effect of molecular composition of xylan extracted from birch on its assembly onto bleached softwood kraft pulp. Holzforschung 60:143–148. https://doi.org/10.1515/HF.2006.023
Xiong G, Cheng K, Pauly M (2013) Xylan O-acetylation impacts xylem development and enzymatic recalcitrance as indicated by the arabidopsis mutant tbl29. Mol Plant 6:1373–1375. https://doi.org/10.1093/mp/sst014
Xiong G, Dama M, Pauly M (2015) Glucuronic Acid Moieties on Xylan Are Functionally Equivalent to O-Acetyl-Substituents. Mol Plant 8:1119–1121. https://doi.org/10.1016/j.molp.2015.02.013
Zhang B, Zhang L, Li F et al (2017) Control of secondary cell wall patterning involves xylan deacetylation by a GDSL esterase. Nat Plants 3:1–9. https://doi.org/10.1038/nplants.2017.17
Zhao Z, Crespi VH, Kubicki JD et al (2014) Molecular dynamics simulation study of xyloglucan adsorption on cellulose surfaces: effects of surface hydrophobicity and side-chain variation. Cellulose 21:1025–1039. https://doi.org/10.1007/s10570-013-0041-1
Acknowledgments
MG acknowledges Micholas Dean Smith and Utsab Shreshtha for useful discussions. This research was supported by the Center for Lignocellulose Structure and Formation, an Energy Frontier Research Center funded by the U.S. Department of Energy (DOE), Office of Science, Basic Energy Sciences under Award DE- SC0001090. JCS acknowledges support for analysis of MD simulations from the Genomic Science Program, Office of Biological and Environmental Research, DOE under Contract FWP ERKP752. This research used resources of the National Energy Research Scientific Computing Center, supported under Contract No. DE-AC02- 05CH11231. Oak Ridge National Laboratory, which is supported by the Office of Science of the U.S. Department of Energy under Contract No. DE-AC05-00OR22725. This work used resources of the Compute and Data Environment for Science (CADES) at ORNL.This manuscript has been authored in part by UT-Battelle, LLC, under contract DE-AC05-00OR22725 with the US Department of Energy (DOE). The US government retains and the publisher, by accepting the article for publication, acknowledges that the US government retains a nonexclusive, paid-up, irrevocable, worldwide license to publish or reproduce the published form of this manuscript, or allow others to do so, for US government purposes. DOE will provide public access to these results of federally sponsored research in accordance with the DOE Public Access Plan (http://energy.gov/downloads/doe-public-access-plan).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflicts of interest
There are no conflicts to declare.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
About this article
Cite this article
Gupta, M., Rawal, T.B., Dupree, P. et al. Spontaneous rearrangement of acetylated xylan on hydrophilic cellulose surfaces. Cellulose 28, 3327–3345 (2021). https://doi.org/10.1007/s10570-021-03706-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10570-021-03706-z