Abstract
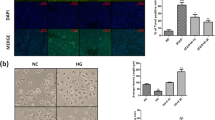
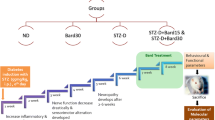
Mitochondrial dysfunction has been implicated as a one of the major factors linked to the development of painful diabetic neuropathy (DN). Several studies have demonstrated that sirtuin (SIRT1) activation recuperates nerve function by activating mitochondrial biogenesis. Polydatin, a resveratrol glycoside, has been explored to improve mitochondrial function via SIRT1 activation. However, the neuroprotective effects of polydatin in DN remain elusive. In this study, polydatin (25 and 50 mg/kg, oral) was administered for last 2 weeks of 8-week study to diabetic Sprague–Dawley rats weighing 250–300 g (post 6-weeks of streptozotocin 55 mg/kg, intraperitoneal). Treatment with polydatin significantly attenuated mechanical and thermal hyperalgesia in diabetic rats. Treated diabetic rats also showed improvement in motor/sensory nerve conduction velocities and nerve blood flow. For in vitro studies, Neuro2a cells were exposed to high-glucose (30 mM) condition to simulate short-term hyperglycemia. Polydatin was evaluated for its role in SIRT1 and Nrf2 activation at a dose of 5, 10, and 20 µM concentrations. Polydatin exposure normalized the mitochondrial superoxides, membrane potentials and improved neurite outgrowth in high-glucose-exposed Neuro2a cells. Increased SIRT1 activation by polydatin resulted in peroxisome proliferator activated receptor-gamma coactivator-1α (PGC-1α) directed mitochondrial biogenesis. SIRT1 activation also facilitated Nrf2-directed antioxidant signaling. Study results inferred that decline in mitochondrial biogenesis and oxidative function in diabetic rats and high-glucose-exposed Neuro2a cells, could be counteracted by polydatin administration, postulated via enhancing SIRT1 and Nrf2 axis.








Similar content being viewed by others
References
Aghdam SY, Gurel Z, Ghaffarieh A, Sorenson CM, Sheibani N (2013) High glucose and diabetes modulate cellular proteasome function: Implications in the pathogenesis of diabetes complications. Biochem Biophys Res Commun 432(2):339–344. https://doi.org/10.1016/j.bbrc.2013.01.101
Bass D, Parce JW, Dechatelet LR, Szejda P, Seeds M, Thomas M (1983) Flow cytometric studies of oxidative product formation by neutrophils: a graded response to membrane stimulation. J Immunol 130(4):1910–1917
Cadenas E, Boveris A, Ragan CI, Stoppani AO (1977) Production of superoxide radicals and hydrogen peroxide by NADH-ubiquinone reductase and ubiquinol-cytochrome c reductase from beef-heart mitochondria. Arch Biochem Biophys 180(2):248–257
Callaghan BC, Cheng HT, Stables CL, Smith AL, Feldman EL (2012) Diabetic neuropathy: clinical manifestations and current treatments. Lancet Neuro 11(6):521–534
Chang DT, Reynolds IJ (2006) Mitochondrial trafficking and morphology in healthy and injured neurons. Prog Neurobiol 80(5):241–268
Chen M, Hou Y, Lin D (2016) Polydatin protects bone marrow stem cells against oxidative injury: involvement of Nrf 2/ARE pathways. Stem Cells Int 1:1–10
Du X-L, Edelstein D, Rossetti L, Fantus IG, Goldberg H, Ziyadeh F, Wu J, Brownlee M (2000) Hyperglycemia-induced mitochondrial superoxide overproduction activates the hexosamine pathway and induces plasminogen activator inhibitor-1 expression by increasing Sp1 glycosylation. Proc Natl Acad Sci 97(22):12222–12226
Fernandez-Marcos PJ, Auwerx J (2011) Regulation of PGC-1α, a nodal regulator of mitochondrial biogenesis. Am J Clin Nutr 93(4):884S–890S
Friedman J (2011) Why is the nervous system vulnerable to oxidative stress? In: Gadoth N, Gobel HH (eds) Oxidative stress and free radical damage in neurology. Springer, New York, pp 19–27
Gao Y, Chen T, Lei X, Li Y, Dai X, Cao Y, Ding Q, Lei X, Li T, Lin X (2016) Neuroprotective effects of polydatin against mitochondrial-dependent apoptosis in the rat cerebral cortex following ischemia/reperfusion injury. Mol Med Rep 14(6):5481–5488
Gong W, Li J, Chen Z, Huang J, Chen Q, Cai W, Liu P, Huang H (2017) Polydatin promotes Nrf2-ARE anti-oxidative pathway through activating CKIP-1 to resist HG-induced up-regulation of FN and ICAM-1 in GMCs and diabetic mice kidneys. Free Radical Biol Med 106:393–405
Huang K, Chen C, Hao J, Huang J, Wang S, Liu P, Huang H (2015) Polydatin promotes Nrf2-ARE anti-oxidative pathway through activating Sirt1 to resist AGEs-induced upregulation of fibronetin and transforming growth factor-β1 in rat glomerular messangial cells. Mol Cell Endocrinol 399:178–189
Huang K, Gao X, Wei W (2017) The crosstalk between sirt1 and keap1/Nrf2/are anti-oxidative pathway forms a positive feedback loop to inhibit FN and TGF-β1 expressions in rat glomerular mesangial cells. Exp Cell Res 361(1):63–72
Imai S-i, Guarente L (2014) NAD+ and sirtuins in aging and disease. Trends Cell Biol 24(8):464–471
Ji H, Zhang X, Du Y, Liu H, Li S, Li L (2012) Polydatin modulates inflammation by decreasing NF-κB activation and oxidative stress by increasing Gli1, Ptch1, SOD1 expression and ameliorates blood–brain barrier permeability for its neuroprotective effect in pMCAO rat brain. Brain Res Bull 87(1):50–59
Kalvala AK, Khan I, Gundu C, Kumar A (2019) An overview on ATP dependent and independent proteases including an anterograde to retrograde control on mitochondrial function; Focus on diabetes and diabetic complications. Curr Pharm Des 25(23):2584–2594
Kalvala AK, Kumar R, Sherkhane B, Gundu C, Arruri VK, Kumar A (2020a) Bardoxolone methyl ameliorates hyperglycemia induced mitochondrial dysfunction by activating the keap1-Nrf2-ARE pathway in experimental diabetic neuropathy. Mol Neurobiol. https://doi.org/10.1007/s12035-020-01989-0
Kalvala AK, Yerra VG, Kumar A (2020b) LONP1 induction by SRT1720 attenuates mitochondrial dysfunction against high glucose induced neurotoxicity in PC12 cells. Toxicol In Vitro 62:104695
Kim H-J, Vaziri ND (2009) Contribution of impaired Nrf2-Keap1 pathway to oxidative stress and inflammation in chronic renal failure. Am J Physiol Physiol 298(3):662–671
Kumar A, Negi G, Sharma SS (2012) Suppression of NF-κB and NF-κB regulated oxidative stress and neuroinflammation by BAY 11–7082 (IκB phosphorylation inhibitor) in experimental diabetic neuropathy. Biochimie 94(5):1158–1165
Lagouge M, Argmann C, Gerhart-Hines Z, Meziane H, Lerin C, Daussin F, Messadeq N, Milne J, Lambert P, Elliott P (2006) Resveratrol improves mitochondrial function and protects against metabolic disease by activating SIRT1 and PGC-1α. Cell 127(6):1109–1122
Lanzilli G, Cottarelli A, Nicotera G, Guida S, Ravagnan G, Fuggetta MP (2012) Anti-inflammatory effect of resveratrol and polydatin by in vitro IL-17 modulation. Inflammation 35(1):240–248
Li L, Tan J, Miao Y, Lei P, Zhang Q (2015) ROS and autophagy: interactions and molecular regulatory mechanisms. Cell Mol Neurobiol 35(5):615–621
Li PA, Hou X, Hao S (2017) Mitochondrial biogenesis in neurodegeneration. J Neurosci Res 95(10):2025–2029
Li Y-g, Zhu W, Tao J-p, Xin P, Liu M-y, Li J-b, Wei M (2013) Resveratrol protects cardiomyocytes from oxidative stress through SIRT1 and mitochondrial biogenesis signaling pathways. Biochem Biophys Res Commun 438(2):270–276
Ma Y, Gong X, Mo Y, Wu S (2016) Polydatin inhibits the oxidative stress-induced proliferation of vascular smooth muscle cells by activating the eNOS/SIRT1 pathway. Int J Mol Med 37(6):1652–1660
Malone JI, Hanna S, Saporta S, Mervis RF, Park CR, Chong L, Diamond DM (2008) Hyperglycemia not hypoglycemia alters neuronal dendrites and impairs spatial memory. Pediatr Diabetes 9(6):531–539
Massicot F, Hache G, David L, Chen D, Leuxe C, Garnier-Legrand L, Rat P, Laprevote O, Coudore F (2013) P2X7 cell death receptor activation and mitochondrial impairment in oxaliplatin-induced apoptosis and neuronal injury: cellular mechanisms and in vivo approach. PLoS ONE 8(6):e66830
Narenjkar J, Roghani M, Alambeygi H, Sedaghati F (2011) The effect of the flavonoid quercetin on pain sensation in diabetic rats. Basic Clin Neurosci 2(3):51–57
Negi G, Kumar A, Sharma S (2011) Nrf2 and NF-κB modulation by sulforaphane counteracts multiple manifestations of diabetic neuropathy in rats and high glucose-induced changes. Curr Neurovasc Res 8(4):294–304
Nemoto S, Fergusson MM, Finkel T (2005) SIRT1 functionally interacts with the metabolic regulator and transcriptional coactivator PGC-1α. J Biol Chem 280(16):16456–16460
Nishikawa T, Edelstein D, Du XL, Yamagishi S-i, Matsumura T, Kaneda Y, Yorek MA, Beebe D, Oates PJ, Hammes H-P (2000) Normalizing mitochondrial superoxide production blocks three pathways of hyperglycaemic damage. Nature 404(6779):787
North BJ, Marshall BL, Borra MT, Denu JM, Verdin E (2003) The human Sir2 ortholog, SIRT2, is an NAD+-dependent tubulin deacetylase. Mol Cell 11(2):437–444
North BJ, Verdin E (2004) Sirtuins: Sir2-related NAD-dependent protein deacetylases. Genome Biol 5(5):224
Phan C-W, David P, Naidu M, Wong K-H, Sabaratnam V (2013) Neurite outgrowth stimulatory effects of culinary-medicinal mushrooms and their toxicity assessment using differentiating Neuro-2a and embryonic fibroblast BALB/3T3. BMC Complement Altern Med 13(1):261
Rolo AP, Palmeira CM (2006) Diabetes and mitochondrial function: role of hyperglycemia and oxidative stress. Toxicol Appl Pharmacol 212(2):167–178
Russell JW, Golovoy DV, Andrea M, Mahendru P, Olzmann JA, Mentzer A, Feldman EL (2002) High glucose-induced oxidative stress and mitochondrial dysfunction in neurons. FASEB J 16(13):1738–1748
Sandireddy R, Yerra VG, Areti A, Komirishetty P, Kumar A (2014) Neuroinflammation and oxidative stress in diabetic neuropathy: futuristic strategies based on these targets. Int J Endocrinol. https://doi.org/10.1155/2014/674987
Sandireddy R, Yerra VG, Komirishetti P, Areti A, Kumar A (2016) Fisetin imparts neuroprotection in experimental diabetic neuropathy by modulating Nrf2 and NF-κB pathways. Cell Mol Neurobiol 36(6):883–892
Tamaki N, Orihuela-Campos RC, Inagaki Y, Fukui M, Nagata T, Ito H-O (2014) Resveratrol improves oxidative stress and prevents the progression of periodontitis via the activation of the Sirt1/AMPK and the Nrf2/antioxidant defense pathways in a rat periodontitis model. Free Radical Biol Med 75:222–229
Tesfaye S, Boulton AJ, Dyck PJ, Freeman R, Horowitz M, Kempler P, Lauria G, Malik RA, Spallone V, Vinik A (2010) Diabetic neuropathies: update on definitions, diagnostic criteria, estimation of severity, and treatments. Diabetes Care 33(10):2285–2293
Tesfaye S, Selvarajah D (2012) Advances in the epidemiology, pathogenesis and management of diabetic peripheral neuropathy. Diabetes Metab Res Rev 28:8–14
Testa R, Bonfigli AR, Prattichizzo F, La Sala L, De Nigris V, Ceriello A (2017) The “metabolic memory” theory and the early treatment of hyperglycemia in prevention of diabetic complications. Nutrients 9(5):437
Vannucci SJ, Maher F, Simpson IA (1997) Glucose transporter proteins in brain: delivery of glucose to neurons and glia. Glia 21(1):2–21
Vincent AM, Feldman EL (2004) New insights into the mechanisms of diabetic neuropathy. Rev Endocr Metab Disord 5(3):227–236
Vincent AM, Mclean LL, Backus C, Feldman EL (2005) Short-term hyperglycemia produces oxidative damage and apoptosis in neurons. FASEB J 19(6):638–640
Xu Y, Nie L, Yin Y-G, Tang J-L, Zhou J-Y, Li D-D, Zhou S-W (2012) Resveratrol protects against hyperglycemia-induced oxidative damage to mitochondria by activating SIRT1 in rat mesangial cells. Toxicol Appl Pharmacol 259(3):395–401
Yerra VG, Kalvala AK, Kumar A (2017) Isoliquiritigenin reduces oxidative damage and alleviates mitochondrial impairment by SIRT1 activation in experimental diabetic neuropathy. J Nutr Biochem 47:41–52
Yerra VG, Kumar A (2017) Adenosine monophosphate-activated protein kinase abates hyperglycaemia-induced neuronal injury in experimental models of diabetic neuropathy: Effects on mitochondrial biogenesis, autophagy and neuroinflammation. Mol Neurobiol 54(3):2301–2312
Yu T, Robotham JL, Yoon Y (2006) Increased production of reactive oxygen species in hyperglycemic conditions requires dynamic change of mitochondrial morphology. Proc Natl Acad Sci 103(8):2653–2658
Zeng Z, Chen Z, Xu S, Song R, Yang H, Zhao K-S (2015) Polydatin alleviates small intestine injury during hemorrhagic shock as a SIRT1 activator. Oxid Med Cell Longev. https://doi.org/10.1155/2015/965961
Acknowledgements
Authors sincerely acknowledge the Department of Pharmaceuticals, Ministry of Chemicals and Fertilizers, Government of India for providing financial support in carrying out this work.
Author information
Authors and Affiliations
Contributions
MS Preethi Bheereddy (Department of Pharmacology and Toxicology, NIPER-HYDERABAD, Telangana, India-500037), performed studies, evaluated data, prepared figures and wrote manuscript; Dr Veera Ganesh Yerra (Postdoctoral Fellow at Keenan Research Centre for Biomedical Sciences, St. Michael's Hospital, Toronto, Canada), performed studies, evaluated data, prepared figures and wrote manuscript; Mr Anil Kumar Kalvala (Department of Pharmacology and Toxicology, NIPER-HYDERABAD, Telangana, India-500037), performed studies, evaluated data, prepared figures and wrote manuscript; Ms Bhoomika S (Department of Pharmacology and Toxicology, NIPER-HYDERABAD, Telangana, India-500037), performed studies, evaluated data, prepared figures and reviewed manuscript; Dr Ashutosh Kumar (Department of Pharmacology and Toxicology, NIPER-HYDERABAD, Balanagar, Telangana, India-500037), designed this work, evaluated data and wrote and reviewed manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no conflict of interest.
Research Involving Human and Animals Participants
This article does not contain any studies with human participants performed by any of the authors. All applicable international, national, and/or institutional guidelines for the care and use of animals were followed. All procedures performed in studies involving animals were in accordance with the ethical standards of the institution at which the studies were conducted. (Institutional Animal Ethics Committee (IAEC)—NIPER-Hyderabad, India + Protocol No: NIP/8/2016/PC/213).
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Bheereddy, P., Yerra, V.G., Kalvala, A.K. et al. SIRT1 Activation by Polydatin Alleviates Oxidative Damage and Elevates Mitochondrial Biogenesis in Experimental Diabetic Neuropathy. Cell Mol Neurobiol 41, 1563–1577 (2021). https://doi.org/10.1007/s10571-020-00923-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10571-020-00923-1