Abstract
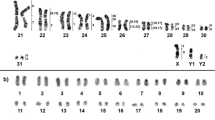
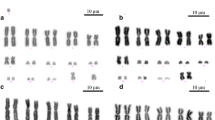
The African pygmy mice (genus Mus, subgenus Nannomys) are recognized for their highly conserved morphology but extensive chromosomal diversity, particularly involving sex-autosome translocations, one of the rarest chromosomal rearrangements among mammals. It has been shown that in the absence of unambiguous diagnostic morphological traits, sex-autosome translocations offer accurate taxonomic markers. For example, in Mus minutoides, irrespective of the diploid number (which ranges from 2n = 18 to 34), all specimens possess the sex-autosome translocations (X.1) and (Y.1) that are unique to this species. In this study, we describe a new cytotype that challenges this view. Males are characterized by the translocation (Y.1) only, while females carry no sex-autosome translocation, the X chromosome being acrocentric. Hence, although sex-autosome translocations (X.1) and (Y.1) are still diagnostic when one or both are present, their absence does not rule out M. minutoides. This cytotype has a large distribution, with specimens found in Tanzania and in the eastern part of South Africa. The nonpervasive distribution of Rb(X.1) provides an opportunity to investigate different evolutionary scenarios of sex-autosome translocations using a phylogenetic framework and the distribution of telomeric repeats. The results tend to support a scenario involving a reversal event, i.e., fusion then fission of Rb(X.1), and highlighted the existence of a new X1X1X2X2/X1X2Y sex chromosome system, confirming the remarkable diversity of neo-sex chromosomes and sex determination systems in the African pygmy mice.




Similar content being viewed by others
Abbreviations
- AIC:
-
Akaike information criterion
- DAPI:
-
4′,6-Diamino-2-phenylindole
- DNA:
-
Deoxyribonucleic acid
- EMBL:
-
European molecular biology laboratory
- FISH:
-
Fluorescence in situ hybridization
- GTR + G + I model:
-
General-time reversible + Gamma + proportion invariant model
- ML:
-
Maximum likelihood
- PCR:
-
Polymerase chain reaction
- Rb fusion:
-
Robertsonian fusion
References
Ashley T (2002) X-autosome translocations, meiotic synapsis, chromosome evolution and speciation. Cytogenet Genome Res 96:33–39
Banaszek A, Taylor JRE, Chetnicki W (2009) Robertsonian polymorphism in the common shrew (Sorex araneus) and selective advantage of heterozygotes indicated by their higher maximum metabolic rates. Heredity 102:155–162
Barasc H, Mary N, Letron R, Calgaro A, Dudez AM, Bonnet N, Lahbib-Mansais Y, Yerle M, Ducos A, Pinton A (2012) Y-autosome translocation interferes with meiotic sex inactivation and expression of autosomal genes: a case study in the pig. Sex Dev 6:143–150
Bayani J, Squire JA (2004) Traditional, banding of chromosomes for cytogenetic analysis. Current Protocols in Cell Biol 22.3
Brannan CI, Disteche CM, Park LS, Copeland NG, Jenkins NA (2001) Autosomal telomere exchange results in the rapid amplification and dispersion and Csf2ra genes in wild-derived mice. Mamm Genome 12:882–886
Britton-Davidian J, Robinson TJ, Veyrunes F (2012) Systematics and evolution of the African pygmy mice, subgenus Nannomys: a review. Acta Oecol 42:41–49
Castiglia R, Gormung E, Corti M (2002) Cytogenetic analyses of chromosomal rearrangements in Mus minutoides/musculoides from North-West Zambia through mapping of the telomeric sequence (TTAGGG)n and banding techniques. Chromosome Res 10:399–406
Castiglia R, Garagna S, Merico V, Oguge N, Corti M (2006) Cytogenetics of a new cytotype of Mus (subgenus Nannomys) minutoides (Rodentia, Muridae) from Kenya: C- and G-banding and distribution of (TTAGGG)n telomeric sequences. Chromosome Res 14:587–594
Cazaux B, Catalan J, Veyrunes F, Douzery EJP, Britton-Davidian J (2011) Are ribosomal DNA clusters rearrangement hotspots? A case study in the genus Mus (Rodentia, Muridae). BMC Evol Biol 11:124
Charlesworth D, Charlesworth B (1980) Sex differences in fitness and selection for centric fusions between sex-chromosomes and autosomes. Genetical Res 35:205–214
Charlesworth D, Charlesworth B, Marais G (2005) Steps in the evolution of heteromorphic sex chromosomes. Heredity 95:118–128
Chatti N, Britton-Davidian J, Catalan J, Auffray J, Saïd K (2005) Reproductive trait divergence and hybrid fertility patterns between chromosomal races of the house mouse in Tunisia: analysis of wild and laboratory-bred males and females. Biol J Linn Soc 84:407–416
Chevret P, Robinson TJ, Perez J, Veyrunes F, Britton-Davidian J (2014) A phylogeographic survey of the pygmy mouse Mus minutoides in South Africa: taxonomic and karyotypic inference from cytochrome b sequences of museum specimens. PLoS One 9:e98499
Darriba D, Taboada GL, Doallo R, Posada D (2012) jModelTest 2: more models, new heuristics and parallel computing. Nat Methods 9:772
Denys C, Lalis A, Lecompte E, Cornette R, Moulin S, Makundi RH, Machang’u RS, Volobouev V, Aniskine VM (2011) A faunal survey in Kingu Pira (south Tanzania), with new karyotypes of several small mammals and the description of a new Murid species (Mammalia, Rodentia). Zoosystema 33:5–47
Deuve JL, Bennett NC, Ruiz-Herrera A, Waters PD, Britton-Davidian J, Robinson TJ (2008) Dissection of a Y-autosome translocation in Cryptomys hottentotus (Rodentia, Bathyergidae) and implications for the evolution of a meiotic sex chromosome chain. Chromosoma 117:211–217
Dobigny G, Ozouf-Costaz C, Bonillo C, Volobouev V (2004) Viability of X-autosome translocations in mammals: an epigenomic hypothesis from a rodent case-study. Chromosoma 113:34–41
Fagundes V, Yonenaga-Yassuda Y (1998) Evolutionary conservation of whole homeologous chromosome arms in the Akodont rodents Bolomys and Akodon (Muridae, Sigmodontinae): maintenance of interstitial telomeric segmenst (ITBs) in recent event of centric fusion. Chromosome res 6:643–648
Fischer C, Ozouf-Costaz C, Roest Crollius H, Dasilva C, Jaillon O, Bouneau L, Bonillo C, Weissenbach J, Bernot A (2000) Karyotype and chromosomal location of characteristic tandem repeats in the pufferfish Tetraodon nigroviridis. Cytogenet Cell Genet 88:50–55
Fredga K (1970) Unusual sex chromosome inheritance in mammals. Phil Trans R Soc Lond B 259:15–36
Garagna S, Broccoli D, Redi CA, Searle JB, Cooke HJ, Capanna E (1995) Robertsonian metacentrics of the house mouse lose telomeric sequences but retain some minor satellite DNA in the pericentromeric area. Chromosoma 103:685–692
Garagna S, Ronchetti E, Mascheretti S, Crovella S, Formenti D, Rumpler Y, Manfredi Romanini MG (1997) Non-telomeric chromosome localization of (TTAGGG)n repeats in the genus Eulemur. Chromosome Res 5:487–491
Gilbert C, Maree S, Robinson TJ (2008) Chromosomal evolution and distribution of telomeric repeats in golden moles (Chrysochloridae, Mammalia). Cytogenet Genome Res 121:110–119
Gropp A, Winking H, Redi C (1982) Consequences of Robertsonian heterozygosity: segregational impairment of fertility versus male-limited sterility. In: Crosignani PG, Rubin BL (eds) Serono Clinical Colloquia on Reproduction. Academic, New York
Guindon S, Gascuel O (2003) A simple, fast, and accurate algorithm to estimate large phylogenies by maximum likelihood. Syst Biol 52:696–704
Happold DCD (2013) Mammals of Africa. Volume 3: Rodentia. University of California Press, Berkeley
Henegariu O, Heerema N, Lowe Wright L, Bray-Ward P, Ward D, Vance G (2001) Improvements in cytogenetic slide preparation: controlled chromosome spreading, chemical aging and gradual denaturing. Cytometry 43:101–109
Hima K, Thiam M, Catalan J, Gauthier P, Duplantier JM, Piry S, Sembène M, Britton-Davidian J, Granjon L, Dobigny G (2011) Extensive Robertsonian polymorphism in the African rodent Gerbillus nigeriae: geographic aspects and meiotic data. J Zool 284:276–285
Jotterand-Bellomo M (1986) Le genre Mus africain, un exemple d’homogénéité caryotypique: étude cytogénétique de Mus minutoides/musculoides (Côte d’Ivoire), de M. setulosus (République Centrafricaine), et de M. mattheyi (Burkina Faso). Cytogenet Cell Genet 42:99–104
Jotterand-Bellomo M (1988) Chromosome analysis of five specimens of Mus bufo-triton (Muridae) from Burundi (Africa): three cytogenetic entities, a special type of chromosomal sex determination, taxonomy, and phylogeny. Cytogenet Cell Genet 48:88–91
Kan Kouassi S, Nicolas V, Aniskine A, Lalis A, Cruaud C, Couloux A, Colyn M, Dosso M, Koivogui L, Verheyen E, Akoua-Koffi C, Denys C (2008) Taxonomy and biogeography of the African pygmy mice, subgenus Nannomys (Rodentia, Murinae, Mus) in Ivory Coast and Guinea (West Africa). Mammalia 72:237–252
King M (1993) Species evolution. The role of chromosome change. Cambridge University Press, Cambridge
Kitano J, Ross J, Mori S, Kume M, Jones F, Chan Y, Absher D, Grimwood J, Schmutz J, Myers R, Kingsley D, Peichel C (2009) A role for a neo-sex chromosome in stickleback speciation. Nature 461:1079–1083
Linardopoulou EV, Williams EM, Fan Y, Friedman C, Young JM, Trask BJ (2005) Human subtelomeres are hot spots of interchromosomal recombination and segmental duplication. Nature 437:94–100
Matthey R (1965) Un type nouveau de chromosomes sexuels multiples chez une souris africaine du groupe Mus (Leggada) minutoides (Mammalia-Rodentia). Chromosoma 16:351–364
Matthey R (1966) Le polymorphisme chromosomique des Mus africains du sous-genre Leggada. Révision générale portant sur l’analyse de 213 individus. Rev Suisse Zool 73:585–607
Matthey R (1970) L’“éventail robertsonien” chez les Mus (Leggada) africains du groupe minutoides-musculoides. Rev Suisse Zool 77:625–629
McDonough MM, Sotero-Caio CG, Ferguson AW, Lewis PJ, Tswiio M, Thies ML (2013) Mitochondrial DNA and karyotypic data confirm the presence of Mus indutus and Mus minutoides (Mammalia, Rodentia, Muridae, Nannomys) in Botswana. Zoo Keys 359:35–51
Medarde N, López-Fuster MJ, Muñoz-Muñoz F, Ventura J (2012) Spatio-temporal variation in the structure of a chromosomal polymorphism zone in the house mouse. Heredity 109:78–89
Nanda I, Schneider-Rasp S, Winking H, Schmid M (1995) Loss of telomeric sites in the chromosomes of Mus musculus domesticus (Rodentia: Muridae) during Robertsonian rearrangements. Chromosome Res 3:399–409
Pagnozzi JM, Silva MJJ, Yonenaga-Yassuda Y (2000) Intraspecific variation in the distribution of the interstitial telomeric (TTAGGG)n sequences in Macoureus demerarae (Marsupiallia: Didelphidae). Chromosome Res 8:585–591
Pialek J, Hauffe HC, Searle JB (2005) Chromosomal variation in the house mouse. Biol J Linnean Soc 84:535–563
Rambaut A, Drummond AJ (2009) Tracer. Available: http://tree.bio.ed.ac.uk/software/tracer/
Ronquist F, Teslenko M, van der Mark P, Ayres DL, Darling A et al (2012) MrBayes 3.2: efficient Bayesian phylogenetic inference and model choice across a large model space. Syst Biol 61:539–542
Rovatsos MT, Marchal JA, Romero-Fernandez I, Fernandez FJ, Giagia-Athanosopoulou EB, Sanchez A (2011) Rapid, independent, and extensive amplification of telomeric repeats in pericentromeric regions in karyotypes of arvicoline rodents. Chromosome Res 19:869–882
Rubes J, Kubickova S, Pagacova E, Cernohorska H, Di Berardino D, Antoninova M, Vahala J, Robinson TJ (2008) Phylogenomic study of spiral-horned antelope by cross-species chromosome painting. Chromosome Res 16:935–947
Ruiz-Herrera A, Nergadze SG, Santagostino M, Giulotto E (2008) Telomeric repeats far from the end: mechanisms of origin and role in evolution. Cytogenet Genome Res 122:219–228
Seabright J (1971) A rapid technique for human chromosomes. Lancet II:971–972
Searle JB (1993) Chromosomal hybrid zones in Eutherian mammals. In: Harrison RG (ed) Hybrid zones and the evolutionary process. Oxford University Press, Oxford, pp 309–353
Searle JB, Wojcik JM (1998) Chromosomal evolution: the case of Sorex araneus. In: Wojcik JM, Wolsan M (eds) Evolution of shrews. Mammal Research Institute PAS, Bialowieza, pp 219–268
Veltsos P, Keller I, Nichols RA (2008) The inexorable spread of a newly arisen neo-Y chromosome. PLoS Genet 4:e1000082
Ventura K, Silva MJJ, Fagundes V, Christoff AU, Yonenaga-Yassuda Y (2006) Non-telomeric sites as evidence of chromosomal rearrangement and repetitive (TTAGGG)n arrays in heterochromatic and euchromatic regions in four species of Akodon (Rodentia, Muridae). Cytogenet Genome Res 115:169–175
Veyrunes F, Catalan J, Sicard B, Robinson TJ, Duplantier JM, Granjon L, Dobigny G, Britton-Davidian J (2004) Autosome and sex chromosome diversity among the African pygmy mice, subgenus Nannomys (Muridae; Mus). Chromosome Res 12:369–382
Veyrunes F, Britton-Davidian J, Robinson TJ, Calvet E, Denys C, Chevret P (2005) Molecular phylogeny of the African pygmy mice, subgenus Nannomys (Rodentia, Murinae, Mus): implications for chromosomal evolution. Mol Phylogenet Evol 36:358–369
Veyrunes F, Dobigny G, Yang F, O’Brien PCM, Catalan J, Robinson TJ, Britton-Davidian J (2006) Phylogenomics of the genus Mus (Rodentia; Muridae): extensive genome repatterning is not restricted to the house mouse. Proc R Soc B 273:2925–2934
Veyrunes F, Watson J, Robinson TJ, Britton-Davidian J (2007) Accumulation of rare sex chromosome rearrangements in the African pygmy mouse, Mus (Nannomys) minutoides: a whole-arm reciprocal translocation (WART) involving an X-autosome fusion. Chromosome Res 15:223–230
Veyrunes F, Catalan J, Tatard C, Cellier-Holzem E, Watson J, Chevret P, Robinson TJ, Britton-Davidian J (2010a) Mitochondrial and chromosomal insights into karyotypic evolution of the pygmy mouse, Mus minutoides, in South Africa. Chromosome Res 18:563–574
Veyrunes F, Chevret P, Catalan J, Castiglia R, Watson J, Dobigny G, Robinson TJ, Britton-Davidian J (2010b) A novel sex determination system in a close relative of the house mouse. Proc R Soc B 277:1049–1056
White MJD (1973) Animal cytology and evolution, 3rd edn. Cambridge University Press, New York
White WM, Willard HF, Van Dyke DL, Wolff DJ (1998) The spreading of X inactivation into autosomal material of an X-autosome translocation: evidence for a difference between autosomal and X-chromosomal DNA. Am J Hum Genet 63:20–28
Yoshida K, Kitano J (2012) The contribution of female meiotic drive to the evolution of neo-sex chromosomes. Evolution 66:3198–3208
Zhdanova NS, Karamisheva TV, Minina J, Astakhova NM, Lansdorp P, Kammori M, Rubtsov NB, Searle JB (2005) Unusual distribution pattern of telomeric repeats in the shrews Sorex araneus and Sorex granarius. Chromosome Res 13:617–625
Zhdanova N, Minina J, Karamisheva T, Draskovic I, Rubtsov N, Londono-Vallejo J (2007) The very long telomeres in Sorex granarius (Soricidae, Eulipothyphla) contain ribosomal DNA. Chromosome Res 15:881–890
Zhou Q, Bachtrog D (2012) Sex-specific adaptation drives early sex chromosome evolution in Drosophila. Science 337:341–345
Acknowledgments
This study was supported by the ANR grant “SEXYMUS” (N° 10-JCJC-1605) to FV, and the Plateforme Cytogénomique évolutive of the LabEx CeMEB. The specimens from Tanzania were collected as part of a University of Antwerp Special Fund for Research project, and those from South Africa were collected as part of a EKZN Wildlife registered project under collecting permit OP4189/2013. BB and SG are research fellows of Research Foundation Flanders (FWO). Our gratitude goes out to the staff of the Pest Management Center at the Sokoine University of Agriculture in Morogoro, Tanzania, for help with the field work.
Author information
Authors and Affiliations
Corresponding author
Additional information
Responsible Editor: Fengtang Yang
Rights and permissions
About this article
Cite this article
Veyrunes, F., Perez, J., Borremans, B. et al. A new cytotype of the African pygmy mouse Mus minutoides in Eastern Africa. Implications for the evolution of sex-autosome translocations. Chromosome Res 22, 533–543 (2014). https://doi.org/10.1007/s10577-014-9440-x
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10577-014-9440-x