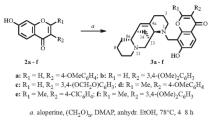
The corresponding 9-azolyl-8-imino-4H,8H-pyrano[2,3-f]chromen-4-ones have been synthesized by the condensation of 7-hydroxy-3-(5-methoxycarbonyl-2-methylfuran-3-yl)-8-formylchromones with 2-aza-hetarylacetonitriles. Acid hydrolysis of the products led to the furyl analogs of 9-azolyl-α-pyrono-[2,3-f]isoflavones.
Similar content being viewed by others
References
B. S. Joshi and V. N. Kamat, Tetrahedron Lett., 7, 5767 (1966).
B. S. Joshi, V. N. Kamat, and A. K. Saksena, Tetrahedron, 23, 4785 (1967).
F. Asai, M. Iinuma, T. Tanaka, and M. Mizuno, Heterocycles, 33, 229 (1992).
M. Iinuma, T. Tanaka, and F. Asai, Phytochemistry, 36, 941 (1994).
E. Schlittler and U. Spitaler, Tetrahedron Lett., 19, 2911 (1978).
P. J. Houghton, Planta Medica, 53, 264 (1987).
T. R. Kelly and M. H. Kim, J. Org. Chem., 57, 1593 (1992).
M. Vijava Lakshmi and N. V. Subba Rao, Curr. Sci., 42, 19 (1973).
K. A. Thakar and N. R. Manjaramkar, Indian J. Chem., 9, 892 (1971).
T. V. Shokol, N. V. Gorbulenko, T. M. Tkachuk, and V. P. Khilya, Khim. Geterotsikl. Soedin., 455 (2009). [Chem. Heterocycl. Comp., 45, 370 (2009)].
T. V. Shokol, V. A. Turov, V. V. Semenyuchenko, and V. P. Khilya, Khim. Prir. Soedin., 544 (2006).
T. V. Shokol, O. A. Lozinskii, A. V. Turov, and V. P. Khilya, Khim. Geterotsikl. Soedin., 1361 (2009). [Chem. Heterocycl. Comp., 45, 1089 (2009)].
S. Rangaswami and T. R. Seshadry, Proc. Indian Acad. Sci., 9A, 7 (1939).
M. Krishnamurty and T. R. Seshadry, J. Sci. Ind. Res., 14B, 258 (1955).
Y. Kawase, T. Sekiba, and K. Fukui, Bull. Chem. Soc. Jpn., 31, 997 (1958).
Author information
Authors and Affiliations
Corresponding author
Additional information
Translated from Khimiya Geterotsiklicheskikh Soedinenii, No. 6, pp. 843-848, June, 2010.
Rights and permissions
About this article
Cite this article
Shokol, T.V., Lozinskii, O.A., Tkachuk, T.M. et al. Furyl analogs of α-pyrono[2,3-f]isoflavones with an azole substituent in the α-pyrone nucleus. Chem Heterocycl Comp 46, 675–680 (2010). https://doi.org/10.1007/s10593-010-0568-0
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10593-010-0568-0