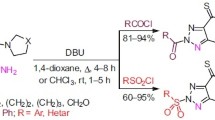
Efficient synthesis of 1-arylsulfonyl-1,2,3-triazoles from easily available (Z)-arylvinyl bromides is described. The sequential reaction involves elimination of HBr from (Z)-arylvinyl bromides promoted by KOH and copper-catalyzed (3+2) cycloaddition reaction between the alkyne intermediates and sulfonyl azides.
Similar content being viewed by others
References
Kim, C.-E.; Park, Y.; Park, S.; Lee, P. H. Adv. Synth. Catal. 2015, 357, 210.
Davies, H. M. L.; Alford, J. S. Chem. Soc. Rev. 2014, 43, 5151 and references therein.
Horneff, T.; Chuprakov, S.; Chernyak, N.; Gevorgyan, V.; Fokin, V. V. J. Am. Chem. Soc. 2008, 130, 14972.
Miura, T.; Yamauchi, M.; Murakami, M. Chem. Commun. 2009, 1470.
Chattopadhyay, B.; Gevorgyan, V. Org. Lett. 2011, 13, 3746.
Miura, T.; Hiraga, K.; Biyajima, T.; Nakamuro, T.; Murakami, M. Org. Lett. 2013, 15, 3298.
Parr, B. T.; Green, S. A.; Davies, H. M. L. J. Am. Chem. Soc. 2013, 135, 4716.
Spangler, J. E.; Davies, H. M. L. J. Am. Chem. Soc. 2013, 135, 6802.
Kwok, S. W.; Zhang L.; Grimster, N. P.; Fokin, V. V. Angew. Chem., Int. Ed. 2014, 53, 3452.
Rostovtsev, V. V.; Green, L. G.; Fokin, V. V.; Sharpless, K. B. Angew. Chem., Int. Ed. 2002, 41, 2596.
Wang, Q.; Chan, T. R.; Hilgraf, R.; Fokin, V. V.; Sharpless, K. B.; Finn, M. G. J. Am. Chem. Soc. 2003, 125, 3192.
Zhang, W.-S.; Kuang, C.-X.; Yang, Q. Chin. J. Org. Chem. 2011, 31, 54.
Jiang, Y. B.; Kuang, C. X. Mini-Rev. Med. Chem. 2013, 13, 713.
Rachwal, S.; Katritzky, A. R. In Comprehensive Heterocyclic Chemistry III; Katrizky, A. R.; Ramsden, C. A.; Scriven, E. F. V.; Taylor, R. J. K., Eds.; Elsevier: Oxford, 2008, Vol. 5, p. 3.
Liang, L.; Astruc, D. Coord. Chem. Rev. 2011, 255, 2933.
Bakulev, V. A.; Berezkina, T. V. Chem. Heterocycl. Compd. 2016, 52, 4. [Khim. Geterotsikl. Soedin. 2016, 52, 4.]
Shafran, E. A.; Bakulev, V. A.; Rozin, Yu. A.; Shafran, Yu. M. Chem. Heterocycl. Compd. 2008, 44, 1040. [Khim. Geterotsikl. Soedin. 2008, 1295.]
Bae, I.; Han, H.; Chang, S. J. Am. Chem. Soc. 2005, 127, 2038.
Yavari, I.; Sheikhi, A.; Nematpour, M.; Taheri, Z. Synth. Commun. 2015, 45, 1089.
Zhang, W; Xu, W; Kuang, C. X. Chin. J. Org. Chem. 2015, 35, 2059.
Liu, Y.; Wang, X.; Xu, J.; Zhang, Q.; Zhao, Y.; Hu, Y. Tetrahedron 2011, 67, 6294 and references therein.
Yoo, E. J.; Ahlquist, M.; Kim, S. H.; Bae, I.; Fokin, V. V.; Sharpless, K. B.; Chang, S. Angew. Chem., Int. Ed. 2007, 46, 1730.
Wang, F.; Fu, H.; Jiang, Y.; Zhao, Y. Adv. Synth. Catal. 2008, 350, 1830.
Beryozkina, T. V.; Efimov, I. V.; Fabian, W. M. F.; Beliaev, N. A.; Slepukhin, P. A.; Isenov, M. L.; Dehaen, W.; Lubec, G.; Eltsov, O. S.; Fan, Z.; Thomas, J.;. Bakulev, V. A. Tetrahedron 2015, 71, 6189.
Kuang, C. X.; Yang, Q.; Senboku, H.; Tokuda, M. Tetrahedron 2005, 61, 4043.
Kuang, C. X.; Senboku, H.; Tokuda, M. Chem. Lett. 2005, 34, 28.
Zhang, W. S.; Kuang, C. X.; Yang, Q. Z. Naturforsch., B: J. Chem. Sci. 2009, 64, 292.
Jiang, Y. B.; Kuang, C. X. Prog. Chem. 2012, 24, 1983.
Zhang, W. S.; Xu, W. J.; Zhang, F.; Qu, G. R. Chin. Chem. Lett. 2013, 24, 407.
Zhang, W.; Xu, W.; Kuang, C. Chin. J. Org. Chem. 2014, 34, 605.
Zhang, W.; Kuang, C.; Yang, Q. Chin. J. Chem. 2009, 27, 1727.
Xu, W.; Zhang, W.; Zhang, F.; Kuang, C. J. Chem. Res. 2014, 38, 115.
Zhang, W.; Su, C.; Kuang, C.; Yang, Q. Synth. Commun. 2011, 41, 1267.
Zhang, W.; Xu, W.; Zhang, F.; Jin, H.; Wang, Y.; Li, J. Res. Chem. Intermed. 2014, 40, 1337.
This work was supported by Foundation of Henan Scientific and Technological Committee (No. 162300410270).
Author information
Authors and Affiliations
Corresponding author
Additional information
Published in Khimiya Geterotsiklicheskikh Soedinenii, 2016, 52(3), 192–195
Rights and permissions
About this article
Cite this article
Zhang, W., Xu, W. Synthesis of 1-arylsulfonyl-1,2,3-triazoles from (Z)-arylvinyl bromides by sequential elimination–cycloaddition reaction. Chem Heterocycl Comp 52, 192–195 (2016). https://doi.org/10.1007/s10593-016-1859-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10593-016-1859-x