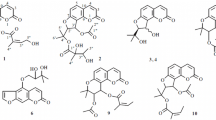
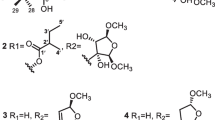
A new furanocoumarin oncosepalin A (1), together with two known compounds, ferulic acid and β-sitosterol, was isolated from the roots of Angelica oncosepala. The structure of the new compound was elucidated by spectroscopic methods, including extensive 1D and 2D NMR experiments, as well as high-resolution EI mass analysis. Compound 1 showed cytotoxic inhibition against HL-60 human tumor cell lines with an IC50 value of 2.04 μM.
Similar content being viewed by others
References
E. M. Murphy, L. Nahar, M. Siakalima, M. Rahman, M. Byres, A. I. Gray, and S. D. Sarker, Biochem. Syst. Ecol., 32, 203 (2003).
S. D. Sarker and L. Nahar, Curr. Med. Chem., 11, 1479 (2004).
J. T. Chen and C. R. Yang, Nat. Prod. Res. Dev., 16 (4), 359 (2004).
V. K. Ahluwalia, D. R. Boyd, A. K. Jain, C. H. Khanduri, and D. Sharma, Phytochemistry, 27, 1181 (1988).
S. K. Garg, S. R. Gupta, and N. D. Sharma, Phytochemistry, 18, 352 (1979).
K. Furumi, T. Fujioka, H. Fuji, H. Okabe, Y. Nakano, H. Matsunaga, M. Katano, M. Mori, and K. Mihashi, Bioorg. Med. Chem. Lett., 8, 93 (1998).
A. Monks, D. Scudiero, P. Skehan, R. Shoemaker, K. Paull, D. Vistica, C. Hose, P. Langley, and A. Vaigro-Wolff, J. Natl. Cancer Inst., 83, 757 (1991).
Acknowledgment
This work was financially supported by the Natural Science Foundation of Yunnan Province (No. 2008ZC110M) and the International Cooperation Foundation of Yunnan Province of China (No. 30811120009).
Author information
Authors and Affiliations
Corresponding author
Additional information
Published in Khimiya Prirodnykh Soedinenii, No. 5, September–October, 2012, pp. 672–673.
Rights and permissions
About this article
Cite this article
Zheng, B., Qing, C., Wang, JL. et al. A new furanocoumarin from the roots of Angelica oncosepala . Chem Nat Compd 48, 748–750 (2012). https://doi.org/10.1007/s10600-012-0373-3
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10600-012-0373-3