Abstract
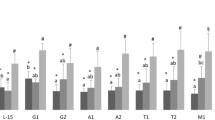
The shortage of the n-3 fatty acids eicosapentaenoic acid (EPA) and docosahexaenoic acid (DHA) on the international markets has led to increasing substitution of fish oil by plant oils in Atlantic salmon (Salmo salar) feed and thereby reducing the EPA and DHA content in salmon. However, the minimum required levels of these fatty acids in fish diets for securing fish health are unknown. Fish were fed with 0, 1 or 2% EPA or DHA alone or in combination of both over a period, growing from 50 to 400 g. Primary head kidney leucocytes were isolated and stimulated with Toll-like receptor (TLR) ligands to determine if EPA and DHA deficiency can affect expression of important immune genes and eicosanoid production. Several genes related to viral immune response did not vary between groups. However, there was a tendency that the high-level EPA and DHA groups expressed lower levels of IL-1β in non-stimulated leucocytes. These leucocytes were also more responsive to the TLR ligands, inducing higher expression levels of IL-1β and Mx1 after stimulation. The levels of prostaglandin E2 and leukotriene B4 in serum and media from stimulated leucocytes were lower in both low and high EPA and DHA groups. In conclusion, leucocytes from low EPA and DHA groups seemed to be less responsive towards immunostimulants, like TLR ligands, indicating that low levels or absence of dietary EPA and DHA may have immunosuppressive effects.







Similar content being viewed by others
Abbreviations
- EPA:
-
Eicosapentaenoic acid
- DHA:
-
Docosahexaenoic acid
- TLR:
-
Toll-like receptor
- PRR:
-
Pattern recognition receptors
- PAMPs:
-
Pathogen-associated molecular patterns
- PLA2:
-
Phospholipase A2
- COX-2:
-
Cyclooxygenase-2
References
Alexopoulou L, Holt AC, Medzhitov R, Flavell RA (2001) Recognition of double-stranded RNA and activation of NF-kappaB by Toll-like receptor 3. Nature 413:732–738. doi:10.1038/35099560
Anderson M, Fritsche KL (2002) (n-3) Fatty acids and infectious disease resistance. J Nutr 132:3566–3576
Arnemo M, Kavaliauskis A, Gjoen T (2014) Effects of TLR agonists and viral infection on cytokine and TLR expression in Atlantic salmon (Salmo salar). Developmental & Comparative Immunology 46:139–145. doi:10.1016/j.dci.2014.03.023
Awate S, Babiuk LA, Mutwiri G (2013) Mechanisms of action of adjuvants. Front Immunol 4. doi:10.3389/fimmu.2013.00114
Bauer S et al (2001) Human TLR9 confers responsiveness to bacterial DNA via species-specific CpG motif recognition. Proc Natl Acad Sci U S A 98:9237–9242. doi:10.1073/pnas.161293498
Bell JG, Dick JR, McVicar AH, Sargent JR, Thompson KD (1993) Dietary sunflower, linseed and fish oils affect phospholipid fatty acid composition, development of cardiac lesions, phospholipase activity and eicosanoid production in Atlantic salmon (Salmo salar). Prostaglandins Leukot Essent Fat Acids 49:665–673. doi:10.1016/0952-3278(93)90075-8
Bell JG, Farndale B, Dick J, Sargent J (1996) Modification of membrane fatty acid composition, eicosanoid production, and phospholipase A activity in Atlantic Salmon (Salmo salar) gill and kidney by dietary lipid. Lipids 31:1163–1171. doi:10.1007/BF02524291
Bell JG, Tocher DR, Farndale BM, Sargent JR (1998) Growth, mortality, tissue histopathology and fatty acid compositions, eicosanoid production and response to stress, in juvenile turbot fed diets rich in γ-linolenic acid in combination with eicosapentaenoic acid or docosahexaenoic acid. Prostaglandins Leukot Essent Fat Acids 58:353–364. doi:10.1016/S0952-3278(98)90071-0
Bransden MP, Carter CG, Nichols PD (2003) Replacement of fish oil with sunflower oil in feeds for Atlantic salmon (Salmo salar L.): effect on growth performance, tissue fatty acid composition and disease resistance. Comp Biochem Physiol B: Biochem Mol Biol 135:611–625. doi:10.1016/S1096-4959(03)00143-X
Buczynski MW, Stephens DL, Bowers-Gentry RC, Grkovich A, Deems RA, Dennis EA (2007) TLR-4 and sustained calcium agonists synergistically produce eicosanoids independent of protein synthesis in RAW264.7 cells. J Biol Chem 282:22834–22847. doi:10.1074/jbc.M701831200
Calder PC (2006) n-3 Polyunsaturated fatty acids, inflammation, and inflammatory diseases. Am J Clin Nutr 83:S1505–1519S
Calder PC (2013) n-3 Fatty acids, inflammation and immunity: new mechanisms to explain old actions. Proc Nutr Soc 72:326–336. doi:10.1017/s0029665113001031
Caughey GE, Mantzioris E, Gibson RA, Cleland LG, James MJ (1996) The effect on human tumor necrosis factor alpha and interleukin 1 beta production of diets enriched in n-3 fatty acids from vegetable oil or fish oil. Am J Clin Nutr 63:116–122
Chang HR, Dulloo AG, Vladoianu IR, Piguet PF, Arsenijevic D, Girardier L, Pechere JC (1992) Fish oil decreases natural resistance of mice to infection with Salmonella typhimurium. Metab Clin Exp 41:1–2
Cláudio N, Dalet A, Gatti E, Pierre P (2013) Mapping the crossroads of immune activation and cellular stress response pathways. EMBO J 32:1214–1224. doi:10.1038/emboj.2013.80
Das S, Mohapatra A, Sahoo PK (2015) Expression analysis of heat shock protein genes during Aeromonas hydrophila infection in rohu, Labeo rohita, with special reference to molecular characterization of Grp78. Cell Stress and Chaperones 20:73–84. doi:10.1007/s12192-014-0527-2
Fagone P, Jackowski S (2009) Membrane phospholipid synthesis and endoplasmic reticulum function. J Lipid Res 50(Suppl):S311–S316. doi:10.1194/jlr.R800049-JLR200
Ferreira MW, Costa DV, Leal CAG, Figueiredo HCP, Rosa PV (2015) Dietary oil sources on the innate immunity and resistance of Nile tilapia, Oreochromis niloticus, to Streptococcus agalactiae challenge. J World Aquacult Soc 46:252–262. doi:10.1111/jwas.12192
Folch J, Lees M, Stanley GHS (1957) A simple method for the isolation and purification of total lipides from animal tissues. J Biol Chem 226:497–509
Ganga R, Bell JG, Montero D, Robaina L, Caballero MJ, Izquierdo MS (2005) Effect of dietary lipids on plasma fatty acid profiles and prostaglandin and leptin production in gilthead seabream (Sparus aurata). Comp Biochem Physiol B: Biochem Mol Biol 142:410–418. doi:10.1016/j.cbpb.2005.09.010
Gjøen T, Obach A, Røsjø C, Helland BG, Rosenlund G, Hvattum E, Ruyter B (2004) Effect of dietary lipids on macrophage function, stress susceptibility and disease resistance in Atlantic salmon (Salmo salar), Fish Physiol Biochem 30:149–161 doi:10.1007/s10695-005-4318-7
Gjøen T et al (2007) Effects of dietary thia fatty acids on lipid composition, morphology and macrophage function of Atlantic salmon (Salmo salar L.) kidney. Comp Biochem Physiol B: Biochem Mol Biol 148:103–111. doi:10.1016/j.cbpb.2007.04.021
Hayashi F et al (2001) The innate immune response to bacterial flagellin is mediated by Toll-like receptor 5. Nature 410:1099–1103. doi:10.1038/35074106
Helland SJ, Grisdale-Helland B, Nerland S (1996) A simple method for the measurement of daily feed intake of groups of fish in tanks. Aquaculture 139:157–163. doi:10.1016/0044-8486(95)01145-5
Hemmi H et al (2000) A Toll-like receptor recognizes bacterial DNA. Nature 408:740–745
Hemmi H et al (2002) Small anti-viral compounds activate immune cells via the TLR7 MyD88-dependent signaling pathway. Nat Immunol 3:196–200
Hoshi M, Williams M, Kishimoto Y (1973) Esterification of fatty acids at room temperature by chloroform-methanolic HCl–cupric acetate. J Lipid Res 14:599–601
Hotamisligil GS (2010) Endoplasmic reticulum stress and the inflammatory basis of metabolic disease. Cell 140:900–917. doi:10.1016/j.cell.2010.02.034
Hummell DS (1993) Dietary lipids and immune function. Progress in food & nutrition science 17:287–329
Hvattum E, Røsjø C, Gjøen T, Rosenlund G, Ruyter B (2000) Effect of soybean oil and fish oil on individual molecular species of Atlantic salmon head kidney phospholipids determined by normal-phase liquid chromatography coupled to negative ion electrospray tandem mass spectrometry. J Chromatogr B Biomed Sci Appl 748:137–149. doi:10.1016/S0378-4347(00)00359-5
Jørgensen SM, Hetland DL, Press CM, Grimholt U, Gjøen T (2007) Effect of early infectious salmon anaemia virus (ISAV) infection on expression of MHC pathway genes and type I and II interferon in Atlantic salmon (Salmo salar L.) tissues. Fish & Shellfish Immunology 23:576–588. doi:10.1016/j.fsi.2007.01.005
Kato H et al (2006) Differential roles of MDA5 and RIG-I helicases in the recognition of RNA viruses. Nature 441:101–105 http://www.nature.com/nature/journal/v441/n7089/suppinfo/nature04734_S1.html
Kelley DS et al (1999) Docosahexaenoic acid ingestion inhibits natural killer cell activity and production of inflammatory mediators in young healthy men. Lipids 34:317–324. doi:10.1007/s11745-999-0369-5
Kiron V, Fukuda H, Takeuchi T, Watanabe T (1995) Essential fatty acid nutrition and defence mechanisms in rainbow trout Oncorhynchus mykiss. Comp Biochem Physiol A Physiol 111:361–367. doi:10.1016/0300-9629(95)00042-6
Lee AS (2005) The ER chaperone and signaling regulator GRP78/BiP as a monitor of endoplasmic reticulum stress. Methods (San Diego, Calif) 35:373–381. doi:10.1016/j.ymeth.2004.10.010
Lee J et al (2003a) Molecular basis for the immunostimulatory activity of guanine nucleoside analogs: activation of Toll-like receptor 7. Proc Natl Acad Sci U S A 100:6646–6651. doi:10.1073/pnas.0631696100
Lee JY, Plakidas A, Lee WH, Heikkinen A, Chanmugam P, Bray G, Hwang DH (2003b) Differential modulation of Toll-like receptors by fatty acids: preferential inhibition by n-3 polyunsaturated fatty acids. J Lipid Res 44:479–486. doi:10.1194/jlr.M200361-JLR200
Lee PT, Zou J, Holland JW, Martin SAM, Kanellos T, Secombes C J (2013) Identification and characterization of TLR7, TLR8a2, TLR8b1 and TLR8b2 genes in Atlantic salmon (Salmo salar). Dev Comp Immunol 41(2):295-305. doi:10.1016/j.dci.2013.05.013
Lødemel JB, Mayhew TM, Myklebust R, Olsen RE, Espelid S, Ringø E (2001) Effect of three dietary oils on disease susceptibility in Arctic charr (Salvelinus alpinus L.) during cohabitant challenge with Aeromonas salmonicida ssp. salmonicida. Aquac Res 32:935–945. doi:10.1046/j.1365-2109.2001.00621.x
Martinez-Rubio L et al (2012) Functional feeds reduce heart inflammation and pathology in Atlantic Salmon (Salmo salar L.) following experimental challenge with Atlantic salmon reovirus (ASRV). PLoS One 7:e40266. doi:10.1371/journal.pone.0040266
Martinez-Rubio L et al (2013) Effect of functional feeds on fatty acid and eicosanoid metabolism in liver and head kidney of Atlantic salmon (Salmo salar L.) with experimentally induced heart and skeletal muscle inflammation. Fish & Shellfish Immunology 34:1533–1545. doi:10.1016/j.fsi.2013.03.363
Martinez-Rubio L et al (2014) Effects of functional feeds on the lipid composition, transcriptomic responses and pathology in heart of Atlantic salmon (Salmo salar L.) before and after experimental challenge with Piscine Myocarditis Virus (PMCV). BMC Genomics 15:462. doi:10.1186/1471-2164-15-462
Mason ME, Waller GR (1964) Dimethoxypropane induced transesterification of fats and oils in preparation of methyl esters for gas chromatographic analysis. Anal Chem 36:583–586. doi:10.1021/ac60209a008
Matsuo A et al (2008) Teleost TLR22 recognizes RNA duplex to induce IFN and protect cells from birnaviruses. J Immunol 181:3474–3485
McMurray DN, Bonilla DL, Chapkin RS (2011) n-3 Fatty acids uniquely affect anti-microbial resistance and immune cell plasma membrane organization. Chem Phys Lipids 164:626–635. doi:10.1016/j.chemphyslip.2011.07.003
Medzhitov R, Janeway C Jr (2000) Innate immune recognition: mechanisms and pathways. Immunol Rev 173:89–97. doi:10.1034/j.1600-065X.2000.917309.x
Montero D, Izquierdo M (2010) Welfare and health of fish fed vegetable oils as alternative lipid sources to fish oil. In: Turchini G, Ng W-K, Tocher D (eds) Fish oil replacement and alternative lipid sources in aquaculture feeds. CRC Press
Montero D, Kalinowski T, Obach A, Robaina L, Tort L, Caballero MJ, Izquierdo MS (2003) Vegetable lipid sources for gilthead seabream (Sparus aurata): effects on fish health. Aquaculture 225:353–370. doi:10.1016/S0044-8486(03)00301-6
Montero D et al (2008) Total substitution of fish oil by vegetable oils in gilthead sea bream (Sparus aurata) diets: effects on hepatic Mx expression and some immune parameters. Fish & Shellfish Immunology 24:147–155. doi:10.1016/j.fsi.2007.08.002
Mourente G, Good JE, Bell JG (2005) Partial substitution of fish oil with rapeseed, linseed and olive oils in diets for European sea bass (Dicentrarchus labrax L.): effects on flesh fatty acid composition, plasma prostaglandins E2 and F2α, immune function and effectiveness of a fish oil finishing diet. Aquac Nutr 11:25–40. doi:10.1111/j.1365-2095.2004.00320.x
Norris PC, Dennis EA (2012) Omega-3 fatty acids cause dramatic changes in TLR4 and purinergic eicosanoid signaling. Proc Natl Acad Sci U S A 109:8517–8522. doi:10.1073/pnas.1200189109
Oarada M, Tsuduki T, Suzuki T, Miyazawa T, Nikawa T, Hong-quan G, Kurita N (2003) Dietary supplementation with docosahexaenoic acid, but not with eicosapentaenoic acid, reduces host resistance to fungal infection in mice. Biochim Biophys Acta Gen Subj 1622:151–160
O’Neill LAJ, Golenbock D, Bowie AG (2013) The history of Toll-like receptors - redefining innate immunity. Nat Rev Immunol 13:453–460. doi:10.1038/nri3446
Pfaffl MW (2001) A new mathematical model for relative quantification in real-time RT–PCR. Nucleic Acids Res 29:e45. doi:10.1093/nar/29.9.e45
Rauta PR, Nayak B, Das S (2012) Immune system and immune responses in fish and their role in comparative immunity study: a model for higher organisms. Immunol Lett 148:23–33. doi:10.1016/j.imlet.2012.08.003
Robertsen B, Trobridge G, Leongt J-A (1997) Molecular cloning of double-stranded RNA inducible MX genes from Atlantic salmon (Salmo salar l.). Developmental & Comparative Immunology 21:397–412. doi:10.1016/S0145-305X(97)00019-0
Rockett BD, Salameh M, Carraway K, Morrison K, Shaikh SR (2010) n-3 PUFA improves fatty acid composition, prevents palmitate-induced apoptosis, and differentially modifies B cell cytokine secretion in vitro and ex vivo. J Lipid Res 51:1284–1297. doi:10.1194/jlr.M000851
Ruyter B, Røsjø C, Einen O, Thomassen MS (2000) Essential fatty acids in Atlantic salmon: effects of increasing dietary doses of n-6 and n-3 fatty acids on growth, survival and fatty acid composition of liver, blood and carcass. Aquac Nutr 6(2):119–127. doi:10.1046/j.1365-2095.2000.00137.x
Schiotz B, Baekkevold E, Poulsen L, Mjaaland S, Gjøen T (2009) Analysis of host- and strain-dependent cell death responses during infectious salmon anemia virus infection in vitro. Virol J 6(1):91. doi:10.1186/1743-422X-6-91
Seierstad SL, Haugland Ø, Larsen S, Waagbø R, Evensen Ø (2009) Pro-inflammatory cytokine expression and respiratory burst activity following replacement of fish oil with rapeseed oil in the feed for Atlantic salmon (Salmo salar L.). Aquaculture 289:212–218. doi:10.1016/j.aquaculture.2008.12.004
Sheldon WM, Blazer VS (1991) Influence of dietary lipid and temperature on bactericidal activity of channel catfish macrophages. J Aquat Anim Health 3:87–93. doi:10.1577/1548-8667(1991)003<0087:IODLAT>2.3.CO;2
Svingerud T, Solstad T, Sun B, Nyrud M L J, Kileng Ø, Greiner-Tollersrud L, Robertsen B (2012) Atlantic Salmon Type I IFN Subtypes Show Differences in Antiviral Activity and Cell-Dependent Expression: Evidence for High IFNb/IFNc–Producing Cells in Fish Lymphoid Tissues. J Immunol 189(12): 5912-5923. doi:10.4049/jimmunol.1201188
Thompson KD, Tatner MF, Henderson RJ (1996) Effects of dietary (n-3) and (n-6) polyunsaturated fatty acid ratio on the immune response of Atlantic salmon, Salmo salar L. Aquac Nutr 2:21–31. doi:10.1111/j.1365-2095.1996.tb00004.x
Tomai MA, Vasilakos JP (2012) TLR agonists as vaccine adjuvants. In: Baschieri S (ed) Innovation in vaccinology. Springer, Dordrecht, pp 205–228. doi:10.1007/978-94-007-4543-8_9
Torstensen BE et al (2005) Tailoring of Atlantic salmon (Salmo salar L.) flesh lipid composition and sensory quality by replacing fish oil with a vegetable oil blend. J Agric Food Chem 53:10166–10178. doi:10.1021/jf051308i
Waagbø R (1994) The impact of nutritional factors on the immune system in Atlantic salmon, Salmo salar L.: a review. Aquac Res 25:175–197. doi:10.1111/j.1365-2109.1994.tb00573.x
Wei D et al (2016) The molecular chaperone GRP78 contributes to toll-like receptor 3-mediated innate immune response to hepatitis C virus in hepatocytes. J Biol Chem 291:12294–12309. doi:10.1074/jbc.M115.711598
Zhang J, Kong X, Zhou C, Li L, Nie G, Li X (2014) Toll-like receptor recognition of bacteria in fish: ligand specificity and signal pathways. Fish & Shellfish Immunology 41:380–388. doi:10.1016/j.fsi.2014.09.022
Zheng X, Tocher D, Dickson C, Bell JG, Teale A (2005) Highly unsaturated fatty acid synthesis in vertebrates: new insights with the cloning and characterization of a Δ6 desaturase of Atlantic salmon. Lipids 40:13–24. doi:10.1007/s11745-005-1355-7
Acknowledgements
The work was supported by Norwegian Research Council (NRC 224913) and The Norwegian Seafood Research Fund (FHF 900957). The authors wish to thank Anne-Lise Rishovd for excellent technical assistance
Author information
Authors and Affiliations
Corresponding author
Additional information
Statement of relevance
The shortage of n-3 unsaturated fatty acids on the international markets has led to increasing substitution of fish oil by plant oils in Atlantic salmon feed and thereby reducing the EPA and DHA content of salmon. However, the effects of very low dietary levels of these fatty acids on fish health are unknown and therefore important to examine.
Highlights
• Salmon head kidney EPA/DHA composition correlated with dietary EPA/DHA.
• Head kidney EPA correlated negatively with IL-1β expression in salmon leucocytes.
• Induction of immune transcripts was reduced in leucocytes from low EPA/DHA groups.
• Prostaglandin levels were affected by dietary EPA/DHA.
• Dietary EPA/DHA may be important in resistance to infection and vaccine effect.
Electronic supplementary material
Supplementary Figure 1
(PDF 206 kb)
Rights and permissions
About this article
Cite this article
Arnemo, M., Kavaliauskis, A., Andresen, A.M.S. et al. Effects of dietary n-3 fatty acids on Toll-like receptor activation in primary leucocytes from Atlantic salmon (Salmo salar). Fish Physiol Biochem 43, 1065–1080 (2017). https://doi.org/10.1007/s10695-017-0353-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10695-017-0353-4