Abstract
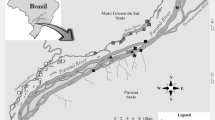
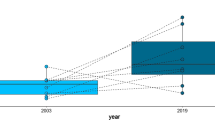
The processes that determine the coexistence of phylogenetically close species are very complex, particularly when species introduction leads to sympatry among species that did not co-evolved. We evaluated possible differences in δ13C and δ15N signatures between two piranha species (Serrasalmus marginatus and S. maculatus), in a system where S. marginatus invaded 30 years ago (floodplain ponds in the Upper Paraná River). We predicted that carbon and nitrogen stable isotope values would not differ between piranha species. Additionally, we evaluated the abundance (CPUE) of both piranha populations along the years (1986–2015). Native and non-native Serrasalmus species have different δ13C signatures, likely exploiting different energy pathways on the food web. Overall, native and non-native piranhas have similar δ15N values and occupy the third trophic level in the food web. Regarding the two piranha population fluctuations, there was an inversion of dominance after the non-native species establishment, where S. marginatus became dominant over S. maculatus (after 1988). Our results showed that trophic niche dimension (revealed by trophic segregation) is not the reason of the observed inversion in the dominant species, and this could be a primary factor driving the persistence of the native species in the ecosystem.




Similar content being viewed by others
References
Abell, R., M. L. Thieme, C. Revenga, M. Bryer, M. Kottelat, N. Bogutskaya, B. Coad, N. Mandrak, S. C. Balderas, W. Bussing, M. L. J. Stiassny, P. Skelton, G. R. Allen, P. Unmack, A. Naseka, R. Ng, N. Sindorf, J. Robertson, E. Armijo, J. V. Higgins, T. J. Heibel, E. Wikramanayake, D. Olson, H. L. López, R. E. Reis, J. G. Lundberg, M. H. S. Pérez & P. Petry, 2008. Freshwater ecoregions of the world: a new map of biogeographic units for freshwater biodiversity conservation. Bioscience 58: 403–414.
Agostinho, C. S. & H. F. Júlio Jr., 2002. Observation of an invasion of the piranha Serrasalmus marginatus, Serrasalmidae) into the Upper Paraná River. Brazil. Acta Scientiarium. Biological Sciences 24: 391–395.
Agostinho, C. S., N. S. Hahn & E. E. Marques, 2003. Patterns of food resource use by tow congeneric species of piranhas (Serrasalmus) on the Upper Paraná River floodplain. Brazilian Journal of Biology 63: 177–182.
Agostinho, A. A., H. F. Júlio Jr. & M. Petrere-Junior, 1994. Itaipu reservoir: impacts of the impoundment on fish fauna and fisheries. In Cowx, I. G. (ed.), Rehabilitation of Freshwaters Fisheries. Blackwell Scientific Publications, Oxford: 171–184.
Agostinho, A. A., H. I. Suzuki, R. Fugi, D. C. Alves, L. H. Tonella & L. A. Espindola, 2015. Ecological and life history traits of Hemiodus orthonops in the invasion process: looking for clues at home. Hydrobiologia 746: 415–430.
Almeida, V. L. L., N. S. Hahn & C. S. Agostinho, 1998. Stomach content of juvenile and adult of piranhas (Serrasalmus marginatus) in the Paraná floodplains, Brazil. Studies in Neotropical Fauna and Environment 33: 100–105.
Alves, G. H. Z., D. J. Hoeinghaus, G. I. Manetta & E. Benedito, in press. Dry season limnological conditions and basin geology exhibit complex relationships with δ13C and δ15N of carbon sources in four Neotropical floodplains. PLoS ONE.
Bøhn, T., P. A. Amundsen & A. Sparrow, 2008. Competitive exclusion after invasion? Biological Invasions 10: 359–368.
Bonetto, A. A., 1986. The Paraná river system. In Davies, B. R. & K. F. Walker (eds), The ecology of river systems. Junk Publishers, Dordrecht, Dr. W: 541–555.
Bowen, G. J., 2010. Isoscapes: spatial pattern in isotopic biogeochemistry. Annual Review of Earth and Planetary Sciences 38: 161–187.
Buchheister, A. & R. Latour, 2010. Turnover and fractionation of carbon and nitrogen stable isotopes in tissues of a migratory coastal predator, summer flounder (Paralichthys dentatus). Canadian Journal of Fisheries and Aquatic Sciences 67: 445–461.
Chesson, P., 2000. Mechanisms of maintenance of species diversity. Annual Review of Ecology, Evolution, and Systematics 31: 343–366.
Córdova-Tapia, F., M. Contreras & L. Zambrano, 2015. Trophic niche overlap between native and non-native fishes. Hydrobiologia 746: 291–301.
Correa, S. C. & K. O. Winemiller, 2014. Niche partitioning among frugivorous fishes in response to fluctuating resources in the Amazonian floodplain forest. Ecology 95: 210–224.
Daga, V. S., F. Skóra, A. A. Padial, V. Abilhoa, E. A. Gubiani & J. R. S. Vitule, 2015. Homogenization dynamics of the fish assemblages in Neotropical reservoirs: comparing the roles of introduced species and their vectors. Hydrobiologia 746: 327–347.
Dayan, T. & D. Simberloff, 2005. Ecological and community-wide character displacement the next generation. Ecology Letters 8: 875–894.
Dudgeon, D., A. H. Arthington, M. O. Gessner, Z. I. Kawabata, D. J. Knowler, C. Lévêque, R. J. Naiman, A. Prieur-Richard, D. Soto, M. L. J. Stiassny & C. A. Sullivan, 2006. Freshwater biodiversity: importance, threats, status and conservation challenges. Biological Reviews Cambridge Philosophical Society 81: 163–182.
Eloranta, A. P., P. Nieminen & K. K. Kahilainen, 2015. Trophic interactions between introduced lake trout (Salvelinus namaycush) and native Arctic charr (S. alpinus) in a large Fennoscandian subarctic lake. Ecology of Freshwater Fish 24: 181–192.
Ernandes-Silva, J., F. H. Ragonha, L. C. Rodrigues & R. P. Mormul, 2016. Freshwater invasibility level depends on the population age structure of the invading mussel species. Biological Invasions 18: 1421–1430.
Faye, D., L. T. Morais, J. Raffray, O. Sadio, O. T. Thiaw & F. Le Loc’h, 2011. Structure and seasonal variability of fish food webs in an estuarine tropical marine protected area (Senegal): evidence from stable isotope analysis. Estuarine, Coastal and Shelf Science 92: 607–617.
Ferreira, F. S., W. Vicentin, F. E. S. Costa & Y. R. Súarez, 2014. Trophic ecology of two piranha species, Pygocentrus nattereri and Serrasalmus marginatus (Characiformes, Characidae), in the floodplain of the Negro River, Pantanal. Acta Limnologica Brasiliensia 26: 381–391.
Forsberg, B. R., C. A. R. M. Araujo-Lima, L. A. Martinelli, R. L. Victoria & J. A. Bonassi, 1993. Autotrophic carbon sources for fish of the central Amazon. Ecology 74: 643–652.
Fry, B., 2006. Stable Isotope Ecology. Springer, New York.
Gause, G. F., 1934. The Struggle for Existence. Williams & Wilkins, Baltimore.
Graça, W. J. & C. S. Pavanelli, 2007. Peixes da planície de inundação do alto rio Paraná e áreas adjacentes. Eduem, Maringá.
Guzzo, M. M., G. D. Haffner, N. D. Legler, S. A. Rush & A. T. Fisk, 2013. Fifty years later: trophic ecology and niche overlap of a native and non-indigenous fish species in the western basin of Lake Erie. Biological Invasions 15: 1695–1711.
Hill, J. M., R. W. Jones, M. P. Hill & O. L. F. Weyl, 2015. Comparisons of isotopic niche widths of some invasive and indigenous fauna in a South African river. Freshwater Biology 60: 893–902.
Hobson, K. A. & R. G. Clark, 1992. Assessing avian diets using stable isotopes I: turnover of 13C in tissues. The Condor 94: 181–188.
Hobson, K. A., R. Barnett-Johnson & T. Cerling, 2010. Using isoscapes to track animal migration. In West, J. B., G. J. Bowen, T. E. Dawson & K. P. Tu (eds), Isoscapes. Springer, Dordrecht: 273–298.
Hoeinghaus, D. J., K. O. Winemiller & A. A. Agostinho, 2007. Landscape-scale hydrologic characteristics differentiate patterns of carbon flow in large-river food webs. Ecosystems 10: 1019–1033.
Júlio Jr., H. F., C. D. Tós, A. A. Agostinho & C. S. Pavanelli, 2009. A massive invasion of fish species after eliminating a natural barrier in the upper rio Paraná basin. Neotropical Ichthyology 7: 709–718.
Juncos, R., D. Milano, P. J. Macchi & P. H. Vigliano, 2015. Niche segregation facilitates coexistence between native and introduced fishes in a deep Patagonian lake. Hydrobiologia 747: 53–67.
Keppeler, F. W., L. E. K. Lanés, A. S. Rolon, C. Stenert, P. Lehmann, M. Reichard & L. Maltchik, 2014. The morphology-diet relationship and its role in the coexistence of two species of annual fishes. Ecology of Freshwater Fish 24: 77–90.
Latini, A. O. & M. Petrere Jr., 2004. Reduction of a native fish fauna by alien species: an example from Brazilian freshwater tropical lakes. Fisheries Management and Ecology 11: 71–79.
Layman, C. G., D. A. Arrington, C. G. Montaña & D. M. Post, 2007. Can stable isotope ratios provide for community-wide measures of trophic structure? Ecology 88: 42–48.
Le Loc’h, F., J. Durand, K. Diop & J. Panfili, 2015. Spatio-temporal isotopic signatures (δ13C and δ15N) reveal that two sympatric West African mullet species do not feed. Journal of Fish Biology 86: 1444–1453.
Li, S. P., M. W. Cadotte, S. J. Meiners, Z. S. Hua, H. Y. Shu, J. T. Li & W. S. Shu, 2015. The effects of phylogenetic relatedness on invasion success and impact: deconstructing Darwin’s naturalisation conundrum. Ecology Letters 18: 1285–1292.
Linde, A. R., J. I. Izquierdo, J. Costa-Moreira & E. Garcia-Vazquez, 2008. Invasive tilapia juveniles are associated with degraded river habitats. Aquatic Conservation 18: 891–895.
Martinelli, L. A., A. H. Devol, R. L. Victoria & J. E. Richey, 1991. Stable carbon isotope variation in C3 and C4 plants along the Amazon River. Nature 353: 57–59.
Martínez del Rio, C., N. Wolf, S. A. Carleton & L. Z. Gannes, 2009. Isotopic ecology ten years after a call for more laboratory experiments. Biological Reviews 84: 91–111.
McCutchan Jr., H. J., W. M. Lewis Jr., C. Kendall & C. C. McGrath, 2003. Vatiation in trophic shift for stable isotope ratios of carbon, nitrogen, and sulphur. Oikos 102: 378–390.
Mercado-Silva, N., M. R. Helmus & M. J. Vander-Zanden, 2009. The effects of impoundment and non-native species on a river food web in Mexico’s Central plateau. River Research and Application 25: 1090–1108.
Monroy, M., A. Maceda-Veiga, N. Caiola & A. De Sostoa, 2014. Trophic interactions between native and introduced fish species in a littoral fish community. Journal of Fish Biology 85: 1693–1706.
Olsson, K., P. Stenroth, P. Nystrom & W. Graneli, 2009. Invasions and niche width: does niche width of an introduced crayfish differ from a native crayfish? Freshwater Biology 54: 1731–1740.
Parnell. A. C. & A. Jackson, 2013. Siar: Stable Isotope Analysis in R. R package version 4.2. http://CRAN.R-project.org/package=siar.
Pelicice, F. M., J. R. S. Vitule, D. P. Lima-Junior, M. L. Orsi & A. A. Agostinho, 2014. A serious new threat to Brazilian freshwater ecosystems: the naturalization of nonnative fish by decree. Conservation Letters 7: 55–60.
Peterson, B. J. & B. Fry, 1987. Stable isotopes in ecosystem studies. Annual Review of Ecology Evolution and Systematics 18: 293–320.
Philippsen, J. S., M. Hauser & E. Benedito, 2015. Isotopic niches of sympatric native and exotic fish species in a Neotropical floodplain. Anais da Academia Brasileira de Ciências 87: 825–833.
Polačik, M., C. Harrod, R. Blažek & M. Reichard, 2014. Trophic niche partitioning in communities of African annual fish: evidence from stable isotopes. Hydrobiologia 721: 99–106.
Post, D. M., 2002. Using stable isotopes to estimate trophic position: models, methods, and assumptions. Ecology 83: 703–718.
R Core Team, 2014. R: A language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria. URL http://www.R-project.org/.
Reid, D. J., G. P. Quinn, P. S. Lake & P. Reich, 2008. Terrestrial detritus supports the food webs in lowland intermittent streams of south-eastern Australia: a stable isotope study. Freshwater Biology 53: 2036–2050.
Ricciardi, A. & M. Mottiar, 2006. Does Darwin’s naturalization hypothesis explain fish invasions? Biological Invasions 8: 1403–1407.
Roberto, M. C., M. F. Santana & S. M. Thomaz, 2009. Limnology in the Upper Paraná River floodplain: large-scale spatial and temporal patterns, and the influence of reservoirs. Brasilian Journal of Biology 69: 717–725.
Ruokonen, T. J., J. Karjalainen, M. Kiljunen, M. Pursiainen & H. Hämäläinene, 2012. Do introduced crayfish affect benthic fish in stony littoral habitats of large boreal lakes. Biological Invasions 14: 813–825.
Sacramento, P. A., G. I. Manetta & E. Benedito, 2016. Diet-tissue discrimination factors (Δ13C and Δ15N) and turnover rate in somatic tissues of a neotropical detritivorous fish on C3 and C4 diets. Journal of Fish Biology 89: 213–219.
Sagouis, A., J. Cucherousset, S. Villéger, F. Santoul & S. Boulêtreau, 2015. Non-native species modify the isotopic structure of freshwater fish communities across the globe. Ecography 38: 979–985.
Sanches, F. H. C., C. A. Miyai, T. M. Costa, R. A. Christofoletti, G. L. Volpato & R. E. Barreto, 2012. Aggressiveness overcomes body-size effects in fights staged between invasive and native fish species with overlapping niches. Plos ONE 7: e29746.
Sazima, I. & F. A. Machado, 1990. Underwater observations of piranhas in western Brazil. Environmental Biology of Fish 28: 17–31.
Thomaz, S. M., T. A. Pagioro, L. M. Bini, M. C. Roberto & R. R. A. Rocha, 2004. Limnological characterization of the aquatic environments and the influence of hydrometrics levels. In Thomaz, S. M., A. A. Agostinho & N. S. Hahn (eds) The Upper Paraná River and Its Floodplain: Physical Aspects, Ecology and Conservation. Backhuys Publishers, Leiden 75:102.
Thomaz, S. M., T. A. Pagioro, L. M. Bini, M. C. Roberto & R. R. A. Rocha, 2004. Limnology of the Upper Paraná River Floodplain: Patterns of Spatio-temporal Variations and Influence of the Water Levels. In Agostinho, A. A., L. Rodrigues, L. C. Gomes, S. M. Thomaz & L. E. Miranda (eds) Structure and Functioning of the Paraná River and Its Floodplain: LTER – site 6. EDUEM, Maringá 37: 42.
Vander-Zanden, M. J. & J. B. Rasmussen, 1999. Primary consumer δ13C and δ15N and the trophic position of aquatic consumers. Ecology 80: 1395–1404.
Vander-Zanden, M. J., G. Cabana & J. B. Rasmussen, 1997. Comparing the trophic position of litoral fish estimated using stable nitrogen isotopes (δ15N) and dietary. Canadian Journal of Fisheries and Aquatic Sciences 54: 1142–1158.
Vazzoler, A. E. A. M., H. I. Suzuki, E. E. Marques & M. L. A. P. Lizama, 1997. Primeira maturação gonadal, períodos e áreas de reprodução. In Vazzoler, A. E. A. M., A. A. Agostinho & N. S. Hahn (eds), A planície de inundação do alto rio Paraná: Aspectos físicos, biológicos e socioeconômicos. EDUEM, Maringá: 248–265.
Vitule, J. R. S., F. Skóra & V. Abilhoa, 2012. Homogenization of freshwater fish faunas after the elimination of a natural barrier by a dam in Neotropics. Diversity and Distributions 18: 111–120.
Acknowledgements
The authors thank Nupélia/UEM, the CNPq/SISBIOTA project, the Graduate Program in Ecology of Continental Aquatic Environments (Programa de Pós-Graduação em Ecologia de Ambientes Aquáticos Continentais - PEA); CNPq/PELD for financial support and infrastructure to develop the study; and CNPq and CAPES for the scholarship granted to Gustavo H. Z. Alves, Bruno R. S. Figueiredo, Raffael M. Tófoli, and Patrícia A. Sacramento. We are also grateful to three anonymous referees for providing valuable suggestions. This work was partially supported by CAPES, an organ of the Brazilian Government for the training of human resources.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Handling editor: Michael Power
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Alves, G.H.Z., Figueiredo, B.R.S., Manetta, G.I. et al. Trophic segregation underlies the coexistence of two piranha species after the removal of a geographic barrier. Hydrobiologia 797, 57–68 (2017). https://doi.org/10.1007/s10750-017-3159-6
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10750-017-3159-6