Abstract
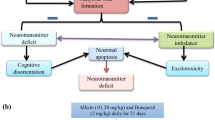
Alzheimer's disease (AD) is the most prevalent type of dementia, characterized by a gradual decline in cognitive and memory functions of the aged peoples. Long-term exposure to heavy metals (aluminium and iron) cause neurotoxicity by amyloid plaques accumulation, tau phosphorylation, increased oxidative stress, neuroinflammation, and cholinergic neurons degeneration, contributes to the development of AD-like symptoms. The present research work is designed to investigate the neuroprotective effect of spermine in aluminium chloride (AlCl3), and iron (Fe) induced AD-like symptoms in rats. Rats were administered of AlCl3 (100 mg/kg p.o.) alone and in combination with iron (120 μg/g, p.o.) for 28 days. Spermine (5 and 10 mg/kg) through intraperitoneal (i.p.) route was given for 14 days. The recognition and spatial memory impairment were tasted using Morris water maze (MWM), actophotometer, and Novel Object Recognition test (NORT). All the rats were sacrificed on day 29, brains were isolated, and tissue homogenate was used for neuroinflammatory, biochemical, neurotransmitters, metals concentration, and nuclear factor-kappa B (NF-κB) analysis. In the present study, AlCl3 and iron administration elevated oxidative stress, cytokines release, dysbalanced neurotransmitters concentration, and biochemical changes. Rats treated with spermine dose-dependently improved the recognition and spatial memory, attenuated proinflammatory cytokine release, and restored neurotransmitters concentration and antioxidant enzymes. Spermine also mitigated the increased beta-amyloid (Aβ42), with downregulation of tau phosphorylation. Furthermore, spermine augmented the hippocampal levels of B cell leukaemia/lymphoma-2 (Bcl-2), diminished nuclear factor-kappa B (NF-κB) and caspase-3 (casp-3) expression. Moreover, spermine exhibited the neuroprotective effect through anti-inflammatory, antioxidant, neurotransmitters restoration, anti-apoptotic Aβ42 concentration.













Similar content being viewed by others
Abbreviations
- AD:
-
Alzheimer’s disease
- Al:
-
Aluminium
- AlCl3:
-
Aluminium chloride
- Fe:
-
Iron
- MWM:
-
Morris water maze
- SOD:
-
Superoxide dismutase
- GR:
-
Glutathione reductase
- IL:
-
Interleukin
- NORT:
-
Novel object recognition test
- LPO:
-
Lipid peroxidation
- TNF-α:
-
Tumor necrosis factor-alpha
- MDA:
-
Malondialdehyde
- DA:
-
Dopamine
- AChE:
-
Acetylcholinesterase
- GABA:
-
Gamma-aminobutyric acid
- Aβ:
-
Amyloid-β peptide
- NFTs:
-
Neurofibrillary tangles
- CNS:
-
Central nervous system
- Ach:
-
Acetylcholine
- ELISA:
-
Enzyme-linked immunosorbent assay
- ECD:
-
Electrochemical detection
- HPLC:
-
High-performance liquid chromatography
- AAS:
-
Atomic absorption spectrophotometer
- CPCSEA:
-
Committee for the Purpose of Control and Supervision of Experiments on Animals
References
Aebi H (1984) [13] Catalase in vitro. Methods Enzymol 105:121–126
Alawdi SH, El-Denshary ES, Safar MM, Eidi H, David MO, Abdel-Wahhab MA (2017) Neuroprotective effect of nanodiamond in Alzheimer’s disease rat model: a pivotal role for modulating NF-κB and STAT3 signaling. Mol Neurobiol 54(3):1906–1918
Alexandrov PN, Zhao Y, Pogue AI, Tarr MA, Kruck TP, Percy ME (2005) Synergistic effects of iron and aluminum on stress-related gene expression in primary human neural cells. J Alzheimers Dis. 8:117–127 (discussion 209–215)
Ali H, Khan E, Ilahi I (2019) Environmental chemistry and ecotoxicology of hazardous heavy metals: environmental persistence, toxicity, and bioaccumulation. J Chem 2019:1–14
Bharathi, (2006) A new insight on Al-maltolate-treated aged rabbit as Alzheimer’s animal model. Brain Res Rev 52:275–292
Bhattacharjee S, Zhao Y, Hill JM, Percy ME, Lukiw WJ (2014) Aluminum and its potential contribution to Alzheimer’s disease (AD). Front Aging Neurosci 6:62
Bondy SC (2013) Prolonged exposure to low levels of aluminum leads to changes associated with brain aging and neurodegeneration. Toxicology 315:1–7. https://doi.org/10.1016/j.tox.2013.10.008
Brown RC, Lockwood AH, Sonawane BR (2005) Neurodegenerative diseases: an overview of environmental risk factors. Environ Health Perspect 113(9):1250–1256
Chen J, Wang M, Ruan D, She J (2002) Early chronic aluminium exposure impairs long-term potentiation and depression to the rat dentate gyrus in vivo. Neuroscience 112(4):879–887
Chipuk JE, Green DR (2008) How do BCL-2 proteins induce mitochondrial outer membrane permeabilization? Trends Cell Biol 18(4):157–164
Cho HH, Cahill CM, Vanderburg CR, Scherzer CR, Wang B, Huang X, Rogers JT (2010) Selective translational control of the alzheimer amyloid precursor protein transcript by iron regulatory protein-1. J Biol Chem 285(41):31217–31232
Cummings Cole JLG (2002) Alzheimer disease. JAMA 287(18):2335–2338
Del Maestro RF, McDonald W, Anderson R (1983) Superoxide dismutase, catalase and glutathione peroxidase in experimental and human brain tumours. Oxyg Radic Scav Syst 2:28
Dickstein DL, Walsh J, Brautigam H, Stockton SD, Gandy S, Hof PR (2010) Role of vascular risk factors and vascular dysfunction in Alzheimer’s disease. Mt Sinai J Med 77(1):82–102
Donzanti BA& Yamamoto BK, (1988) An improved and rapid HPLC-EC method for the isocratic separation of amino acid neurotransmitters from brain tissue and microdialysis perfusates. Life Sci 43(11):913–922
Ellman GL (1959) Tissue sulfhydryl groups. Arch Biochem Biophys 82(1):70–77
Ellman GL, Courtney KD, Andres V Jr, Featherstone RM (1961) A new and rapid colorimetric determination of acetylcholinesterase activity. Biochem Pharmacol 7(2):88–95
Enas AK (2010) Study of possible protective and therapeutic influence of coriander (Coriandrum sativum L.) against neurodegenerative disorders and Alzheimer’s disease induced by aluminum chloride in cerebral cortex of male albino rats. Nat Sci 8:202–213
Ennaceur A, Delacour J (1988) A new one-trial test for neurobiological studies of memory in rats. 1: behavioral data. Behav Brain Res. 31(1):47–59
Exley C, Esiri MM (2006) Severe cerebral congophilic angiopathy coincident with increased brain aluminium in a resident of Camelford, Cornwall, UK. J Neurol Neurosurg Psychiatry 77(7):877–879
Galva C, Artigas P, Gatto C (2012) Nuclear Na+/K+-ATPase plays an active role in nucleoplasmic Ca2+ homeostasis. J Cell Sci 125(24):6137–6147
Green LC, Wagner DA, Glogowski J, Skipper PL, Wishnok JS, Tannenbaum SR (1982) Analysis of nitrate, nitrite, and [15N] nitrate in biological fluids. Anal Biochem 126(1):131–138
Huang HC, Jiang ZF (2009) Accumulated amyloid-β peptide and hyperphosphorylated tau protein: relationship and links in Alzheimer’s disease. J Alzheimers Dis 16(1):15–27
Hussain T, Tan B, Ren W, Rahu N, Dad R, Kalhoro DH, Yin Y (2017) Polyamines: therapeutic perspectives in oxidative stress and inflammatory diseases. Amino Acids 49(9):1457–1468
Karantzoulis S, Galvin JE (2011) Distinguishing Alzheimer’s disease from other major forms of dementia. Expert Rev Neurother 11(11):1579–1591
Kaur S, Raj K, Gupta YK, Singh S (2021) Allicin ameliorates aluminium-and copper-induced cognitive dysfunction in Wistar rats: relevance to neuro-inflammation, neurotransmitters and Aβ (1–42) analysis. J Biol Inorg Chem 26(4):495–510
Kawahara M (2005) Effects of aluminum on the nervous system and its possible link with neurodegenerative diseases. J Alzheimers Dis. 8:171–82
Kim W, Ma L, Lomoio S, Willen R, Lombardo S, Dong J, Tesco G (2018) BACE1 elevation engendered by GGA3 deletion increases β-amyloid pathology in association with APP elevation and decreased CHL1 processing in 5XFAD mice. Mol Neurodegener 13(1):1–22
Kocahan S, Doğan Z (2017) Mechanisms of Alzheimer’s disease pathogenesis and prevention: the brain, neural pathology, N-methyl-d-aspartate receptors, tau protein and other risk factors. Clin Psychopharmacol Neurosci 15(1):1
Li B, Xu P, Wu S, Jiang Z, Huang Z, Li Q, Chen D (2018) Diosgenin attenuates lipopolysaccharide-induced Parkinson’s disease by inhibiting the TLR/NF-κB pathway. J Alzheimers Dis 64(3):943–955
Liang RF, Li WQ, Wang XH, Zhang HF, Wang H, Wang JX, Niu Q (2012) Aluminium-maltolate-induced impairment of learning, memory and hippocampal long-term potentiation in rats. Ind Health. 50:428–436
Limón ID, Angulo-Cruz I, Sánchez-Abdon L, Patricio-Martínez A (2021) Disturbance of the glutamate-glutamine cycle, secondary to hepatic damage, compromises memory function. Front Neurosci 15:578922
Lin WT, Chen RC, Lu WW, Liu SH, Yang FY (2015) Protective effects of low-intensity pulsed ultrasound on aluminum-induced cerebral damage in Alzheimer’s disease rat model. Sci Rep 5(1):1–7
Liu Y, Zhou W, Chen FF, Xiao F, Zhu HY, Zhou Y, Guo GC (2020) Overexpression of LMP-1 decreases apoptosis in human nucleus Pulposus cells via suppressing the NF-κB signaling pathway. Oxid Med Cell Longev 2020:1–13
Lowry OH, Rosebrough NJ, Farr AL, Randall RJ (1951) Protein measurement with the Folin phenol reagent. J Biol Chem 193:265–275
Mold MJ, O’Farrell A, Morris B, Exley C (2021) Aluminum and Tau in neurofibrillary tangles in familial alzheimer’s disease. J Alzheimer’s Dis Rep (Preprint). 5:283–294
Moris RM (1984) Developments of a water-maze procedure for studing spatial learing in the rats. J Neurosci Method 11(1):47–60
Nampoothiri M, John J, Kumar N, Mudgal J, Nampurath GK, Chamallamudi MR (2015) Modulatory role of simvastatin against aluminium chloride-induced behavioural and biochemical changes in rats. Behav Neurol. 2015:210169
Oeckinghaus A, Ghosh S (2009) The NF-κB family of transcription factors and its regulation. Cold Spring Harb Perspect Biol 1(4):a000034
Pratico D, Uryu K, Sung S, Tang S, Trojanowski JQ, Lee VMY (2002) Aluminum modulates brain amyloidosis through oxidative stress in APP transgenic mice. FASEB J 16(9):1138–1140
Reitz C, Brayne C, Mayeux R (2011) Epidemiology of Alzheimer disease. Nat Rev Neurol 7(3):137–152
Rotruck JT, Pope AL, Ganther HE, Swanson AB, Hafeman DG, Hoekstra W (1973) Selenium: biochemical role as a component of glutathione peroxidase. Science 179(4073):588–590
Saha S, Buttari B, Panieri E, Profumo E, Saso L (2020) An overview of Nrf2 signaling pathway and its role in inflammation. Molecules 25(22):5474
Saunders NR, Dziegielewska KM, Møllgård K, Habgood MD (2015) Markers for blood-brain barrier integrity: how appropriate is Evans blue in the twenty-first century and what are the alternatives? Front Neurosci 9:385
Sharma N, Nehru B (2018) Curcumin affords neuroprotection and inhibits α-synuclein aggregation in lipopolysaccharide-induced Parkinson’s disease model. Inflammopharmacology 26(2):349–360
Singh S, Kumar P (2018) Piperine in combination with quercetin halt 6-OHDA induced neurodegeneration in experimental rats: biochemical and neurochemical evidences. Neurosci Res 133:38–47
Singh S, Jamwal S, Kumar P (2015) Piperine enhances the protective effect of curcumin against 3-NP induced neurotoxicity: possible neurotransmitters modulation mechanism. Neurochem Res 40(8):1758–1766
Soda K, Dobashi Y, Kano Y, Tsujinaka S, Konishi F (2009) Polyamine-rich food decreases age-associated pathology and mortality in aged mice. Exp Gerontol 44(11):727–732
Sottocasa GL, Kuylenstierna BO, Ernster L, Bergstrand A (1967) An electron-transport system associated with the outer membrane of liver mitochondria: a biochemical and morphological study. J Cell Biol 32(2):415–438
Tan BL, Norhaizan ME, Liew WPP, Sulaiman Rahman H (2018) Antioxidant and oxidative stress: a mutual interplay in age-related diseases. Front Pharmacol 9:1162
Uttara B, Singh AV, Zamboni P, Mahajan RT (2009) Oxidative stress and neurodegenerative diseases: a review of upstream and downstream antioxidant therapeutic options. Curr Neuropharmacol 7(1):65–74
Wang HL, Kuo EY, Lai TW (2018) Vascular delivery of intraperitoneal Evans blue dye into the blood–brain barrier-intact and disrupted rat brains. NeuroReport 29(11):924–931
Wang L, Wang Y, Zhou S, Yang L, Shi Q, Li Y, Zhang K, Yang L, Zhao M, Yang Q (2016) Imbalance between glutamate and GABA in Fmr1 knockout astrocytes influences neuronal development. Genes 7(8):45–69
Wills E (1966) Mechanisms of lipid peroxide formation in animal tissues. Biochem J 99(3):667–676
Wu Z, Du Y, Xue H, Wu Y, Zhou B (2012) Aluminum induces neurodegeneration and its toxicity arises from increased iron accumulation and reactive oxygen species (ROS) production. Neurobiol Aging 33(1):199–201
Xu YZ, Deng XH, Bentivoglio M (2007) Differential response of apoptosis-regulatory Bcl-2 and Bax proteins to an inflammatory challenge in the cerebral cortex and hippocampus of aging mice. Brain Res Bull 74(5):329–335
Xu TT, Li H, Dai Z, Lau GK, Li BY, Zhu WL, Zhan SJ (2020) Spermidine and spermine delay brain aging by inducing autophagy in SAMP8 mice. Aging (Albany NY) 12(7):6401
Yatin SM, Yatin M, Varadarajan S, Ain B, Butterfield DA (2001) Role of spermine in amyloid β-peptide-associated free radical-induced neurotoxicity. J Neurosci Res 63(5):395–401
Yiannopoulou KG, Papageorgiou SG (2013) Current and future treatments for Alzheimer’s disease. Ther Adv Neurol Disord 6(1):19–33
Yu CA, Yu L (1980) Resolution and reconstitution of succinate-cytochrome c reductase. Preparations and properties of high purity succinate dehydrogenase and ubiquinol—cytochrome c reductase. Biochim Biophys Acta. 591(2):409–420
Yuzwa SA, Shan X, Jones BA, Zhao G, Woodward ML, Li X, Vocadlo DJ (2014) Pharmacological inhibition of O-GlcNAcase (OGA) prevents cognitive decline and amyloid plaque formation in bigenic tau/APP mutant mice. Mol Neurodegener 9(1):1–14
Zhang M, Caragine T, Wang H, Cohen PS, Botchkina G, Soda K, Tracey KJ (1997) Spermine inhibits proinflammatory cytokine synthesis in human mononuclear cells: a counterregulatory mechanism that restrains the immune response. J Exp Med 185(10):1759–1768
Zhang T, Han Y, Wang J, Hou D, Deng H, Deng YL, Song Z (2018) Comparative epidemiological investigation of Alzheimer’s disease and colorectal cancer: the possible role of gastrointestinal conditions in the pathogenesis of AD. Front Aging Neurosci 10:176
Zhao Y, Hill JM, Bhattacharjee S, Percy ME, Pogue AI, Lukiw WJ (2014) Aluminum-induced amyloidogenesis and impairment in the clearance of amyloid peptides from the central nervous system in Alzheimer’s disease. Front Neurol 5:167
Acknowledgements
All authors are thankful to Shri Praveen Garg, chairman ISF College of Pharmacy, Moga, Punjab for providing research platform.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Raj, K., Gupta, G.D. & Singh, S. Spermine protects aluminium chloride and iron-induced neurotoxicity in rat model of Alzheimer's disease via attenuation of tau phosphorylation, Amyloid-β (1–42) and NF-κB pathway. Inflammopharmacol 29, 1777–1793 (2021). https://doi.org/10.1007/s10787-021-00883-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10787-021-00883-y