Abstract
Purpose
Fibroids are the most common smooth muscle overgrowth in women. This study determined the expression and the effect of hypoxia on two potent antioxidant enzymes, superoxide dismutase (SOD) and catalase (CAT) on human fibroid cells.
Methods
Immortalized human leiomyoma (fibroid) and myometrial cells were subjected to hypoxia (2 % O2, 24 h). Total RNA and cell homogenate were obtained from control and treated cells; CAT and SOD mRNA and activity levels were determined by real-time RT-PCR and ELISA, respectively.
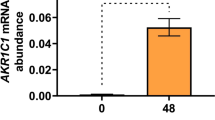
Results
Fibroid cells have significantly lower antioxidant enzymes, SOD and CAT mRNA and activity levels than normal myometrial cells (p < 0.05). Hypoxia treatment significantly increased SOD activity in myometrial cells while significantly decreasing CAT activity in fibroid cells (p < 0.05). There was no significant difference in CAT mRNA levels or activity in response to hypoxia in myometrial cells. Also, there was no significant difference in SOD mRNA levels in response to hypoxia in myometrial cells.
Conclusion
This is the first report to show that uterine fibroids are characterized by an impaired antioxidant cellular enzymatic system. More importantly, our results indicate a role for hypoxia in the modulation of the balance of those enzymes in fibroid and myometrial cells. Collectively, these results shed light on the pathophysiology of fibroids thereby providing potential targets for novel fibroid treatment.


Similar content being viewed by others
References
Agarwal A, Allamaneni SS. Role of free radicals in female reproductive diseases and assisted reproduction. Reprod Biomed Online. 2004;9(3):338–47.
Al-Hendy A, Lee EJ, et al. Gene therapy of uterine leiomyomas: adenovirus-mediated expression of dominant negative estrogen receptor inhibits tumor growth in nude mice. Am J Obstet Gynecol. 2004;191(5):1621–31.
Alpay Z, Saed GM, et al. Female infertility and free radicals: potential role in adhesions and endometriosis. J Soc Gynecol Investig. 2006;13(6):390–8.
Ara C, Kirimlioglu H, et al. Protective effect of melatonin against oxidative stress on adhesion formation in the rat cecum and uterine horn model. Life Sci. 2005;77(12):1341–50.
Augoulea A, Mastorakos G, et al. The role of the oxidative-stress in the endometriosis-related infertility. Gynecol Endocrinol. 2009;25(2):75–81.
Baird DD, Dunson DB, et al. High cumulative incidence of uterine leiomyoma in black and white women: ultrasound evidence. Am J Obstet Gynecol. 2003;188(1):100–7.
Binda MM, Molinas CR, et al. Reactive oxygen species and adhesion formation: clinical implications in adhesion prevention. Hum Reprod. 2003;18(12):2503–7.
Bulman JC, Ascher SM, et al. Current concepts in uterine fibroid embolization. Radiographics. 2012;32(6):1735–50.
Carney SA, Tahara H, et al. Immortalization of human uterine leiomyoma and myometrial cell lines after induction of telomerase activity: molecular and phenotypic characteristics. Lab Invest. 2002;82(6):719–28.
Chiaffarino F, Parazzini F, et al. Diet and uterine myomas. Obstet Gynecol. 1999;94(3):395–8.
Chiou JF, Hu ML. Elevated lipid peroxidation and disturbed antioxidant enzyme activities in plasma and erythrocytes of patients with uterine cervicitis and myoma. Clin Biochem. 1999;32(3):189–92.
Evans P, Brunsell S. Uterine fibroid tumors: diagnosis and treatment. Am Fam Physician. 2007;75(10):1503–8.
Foksinski M, Kotzbach R, et al. The level of typical biomarker of oxidative stress 8-hydroxy-2′-deoxyguanosine is higher in uterine myomas than in control tissues and correlates with the size of the tumor. Free Radic Biol Med. 2000;29(7):597–601.
Fruehauf JP, Meyskens Jr FL. Reactive oxygen species: a breath of life or death? Clin Cancer Res. 2007;13(3):789–94.
Galijasevic S, Saed GM, et al. High dissociation rate constant of ferrous-dioxy complex linked to the catalase-like activity in lactoperoxidase. J Biol Chem. 2004;279(38):39465–70.
Guo Y, Xiao P, et al. How is mRNA expression predictive for protein expression? A correlation study on human circulating monocytes. Acta Biochim Biophys Sin (Shanghai). 2008;40(5):426–36.
Halliwell B. Reactive oxygen species in living systems: source, biochemistry, and role in human disease. Am J Med. 1991;91(3C):14S–22S.
Harris AL. Hypoxia–a key regulatory factor in tumour growth. Nat Rev Cancer. 2002;2(1):38–47.
Hassan MH, Salama SA, et al. Adenovirus-mediated delivery of a dominant-negative estrogen receptor gene in uterine leiomyoma cells abrogates estrogen- and progesterone-regulated gene expression. J Clin Endocrinol Metab. 2007;92(10):3949–57.
Inoue M, Sato EF, et al. Mitochondrial generation of reactive oxygen species and its role in aerobic life. Curr Med Chem. 2003;10(23):2495–505.
Kamata H, Hirata H. Redox regulation of cellular signalling. Cell Signal. 1999;11(1):1–14.
Kieran MW, Folkman J, et al. Angiogenesis inhibitors and hypoxia. Nat Med. 2003;9(9):1104. author reply 1104–1105.
Kirkman HN, Gaetani GF. Catalase: a tetrameric enzyme with four tightly bound molecules of NADPH. Proc Natl Acad Sci U S A. 1984;81(14):4343–7.
Linnane AW, Eastwood H. Cellular redox regulation and prooxidant signaling systems: a new perspective on the free radical theory of aging. Ann N Y Acad Sci. 2006;1067:47–55.
Marklund SL. Extracellular superoxide dismutase in human tissues and human cell lines. J Clin Invest. 1984;74(4):1398–403.
Marklund SL, Holme E, et al. Superoxide dismutase in extracellular fluids. Clin Chim Acta. 1982;126(1):41–51.
Nair PM, Park SY, et al. Expression of catalase and glutathione S-transferase genes in Chironomus riparius on exposure to cadmium and nonylphenol. Comp Biochem Physiol C Toxicol Pharmacol. 2011;154(4):399–408.
NIH. 2011. Dietary Supplement Fact Sheet–National Institutes of Health. 2012, From http://ods.od.nih.gov/factsheets/VitaminC-HealthProfessional/.
Pejic S, Todorovic A, et al. Superoxide dismutase and lipid hydroperoxides in blood and endometrial tissue of patients with benign, hyperplastic and malignant endometrium. An Acad Bras Cienc. 2008;80(3):515–22.
Rahman K. Studies on free radicals, antioxidants, and co-factors. Clin Interv Aging. 2007;2(2):219–36.
Sabry M, Al-Hendy A. Innovative oral treatments of uterine leiomyoma. Obstet Gynecol Int. 2012;2012:943635.
Saed GM, Diamond MP. Hypoxia-induced irreversible up-regulation of type I collagen and transforming growth factor-beta1 in human peritoneal fibroblasts. Fertil Steril. 2002;78(1):144–7.
Saed GM, Diamond MP. Modulation of the expression of tissue plasminogen activator and its inhibitor by hypoxia in human peritoneal and adhesion fibroblasts. Fertil Steril. 2003;79(1):164–8.
Saed GM, Zhang W, et al. Molecular characterization of fibroblasts isolated from human peritoneum and adhesions. Fertil Steril. 2001;75(4):763–8.
Sahin K, Ozercan R, et al. Lycopene supplementation prevents the development of spontaneous smooth muscle tumors of the oviduct in Japanese quail. Nutr Cancer. 2004;50(2):181–9.
Sahin K, Ozercan R, et al. Dietary tomato powder supplementation in the prevention of leiomyoma of the oviduct in the Japanese quail. Nutr Cancer. 2007;59(1):70–5.
Sharan C, Halder SK, et al. Vitamin D inhibits proliferation of human uterine leiomyoma cells via catechol-O-methyltransferase. Fertil Steril. 2011;95(1):247–53.
Singh B, Bhat HK. Superoxide dismutase 3 is induced by antioxidants, inhibits oxidative DNA damage and is associated with inhibition of estrogen-induced breast cancer. Carcinogenesis. 2012;33(12):2601–10.
Stewart EA. Epidemiology, clinical manifestations, diagnosis, and natural history of uterine leiomyomas. 2009. From www.uptodate.com.
Trachootham D, Lu W, et al. Redox regulation of cell survival. Antioxid Redox Signal. 2008;10(8):1343–74.
Tuzcu M, Sahin N, et al. The effects of selenium supplementation on the spontaneously occurring fibroid tumors of oviduct, 8-hydroxy-2′-deoxyguanosine levels, and heat shock protein 70 response in Japanese quail. Nutr Cancer. 2010;62(4):495–500.
Ueda S, Masutani H, et al. Redox control of cell death. Antioxid Redox Signal. 2002;4(3):405–14.
Viswanathan M, Hartmann K, et al. Management of uterine fibroids: an update of the evidence. Evid Rep Technol Assess. 2007; (154):1–122 (Full Rep).
Vural M, Camuzcuoglu H, et al. Oxidative stress and prolidase activity in women with uterine fibroids. J Obstet Gynaecol. 2012;32(1):68–72.
Wight TN, Potter-Perigo S. The extracellular matrix: an active or passive player in fibrosis? Am J Physiol Gastrointest Liver Physiol. 2011;301(6):G950–955.
Wise LA, Radin RG, et al. Intake of fruit, vegetables, and carotenoids in relation to risk of uterine leiomyomata. Am J Clin Nutr. 2011;94(6):1620–31.
Yoshino O, Hayashi T, et al. Decreased pregnancy rate is linked to abnormal uterine peristalsis caused by intramural fibroids. Hum Reprod. 2010;25(10):2475–9.
Zhou S, Yi T, et al. Hypoxia: the driving force of uterine myometrial stem cells differentiation into leiomyoma cells. Med Hypotheses. 2011;77(6):985–6.
Author information
Authors and Affiliations
Corresponding author
Additional information
Capsule Uterine fibroids have an impaired antioxidant system.
Rights and permissions
About this article
Cite this article
Fletcher, N.M., Saed, M.G., Abu-Soud, H.M. et al. Uterine fibroids are characterized by an impaired antioxidant cellular system: potential role of hypoxia in the pathophysiology of uterine fibroids. J Assist Reprod Genet 30, 969–974 (2013). https://doi.org/10.1007/s10815-013-0029-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10815-013-0029-7