Abstract
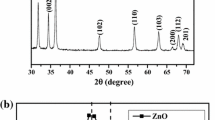
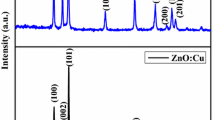
Metal chalcogenide ZnS nanoparticles have been synthesized using simple capped precipitation method using Potassium pyrosulfate as capping agent. The synthesized nano powder was characterized by X-ray diffraction (XRD), UV–Vis absorption, transmission electron microscopy, infrared spectroscopy, thermogravimetric analysis and fluorescence emission spectroscopy. The XRD pattern exhibits three major peaks at d-values 3.12, 1.904, 1.626 Å belonging to 2θ = 28.60°, 47.76° and 56.60° respectively showing Wurtzite crystal phase of ZnS. UV–Vis spectrum exhibits quantum confinement effect by a very sharp peak around 290 nm remarkably blue shifted compared to its bulk counterpart. The room temperature photoluminescence spectrum of the nanoparticles showed two strong emission bands at 421 and 447 nm. This first blue emission peak at 421 nm is due to sulphur vacancy and other peak at 447 nm belongs to native defect states.








Similar content being viewed by others
References
R. John, S. Florence, Chalcogenide Lett. 7(4), 269 (1910)
N.K. Abbas, K.T. Al-Rasoul, Z.J. Shanan, Int. J. Electrochem. Sci. 8, 3049 (2013)
S.K. Mehta, S. Kumar, M. Gradzielski, J. Colloid Interface Sci. 360, 497 (2011)
Y. Cheng, Z. Lin, H. Lu, L. Zhang, B. Yang, Nanotechnology 25(11), 115601 (2014)
S. Kumar, N.K. Verma, J. Mater. Sci. Mater. Electron. 25, 1132 (2014)
J.K. Salem, T.M. Hammad, S. Kuhn, I. Nahal, M.A. Draaz, N.K. Hejazy, R. Hempelmann, J. Mater. Sci. Mater. Electron. 25, 5188 (2014)
J.K. Salem, T.M. Hammad, S. Kuhn, M.A. Draaz, N.K. Hejazy, R. Hempelmann, J. Mater. Sci Mater. Electron. 25, 2177 (2014)
B. Bhattacharjee, D. Ganguli, K. Iakoubovskii, A. Stesmans, S. Chaudhuri, Bull. Mater. Sci. 25(3), 175 (2002)
T. Charinpanitkul, A. Chanagul, J. Dutta, U. Rungsardthong, W. Tanthapanichakoon, Sci. Technol. Adv. Mater. 6, 266 (2005)
A. Rahdar, J. Nanostruct. Chem. 3, 10 (2013)
F.A. La Porta, M.M. Ferrer, Y.V.B. de Santana, C.W. Raubach, V.M. Longo, J.R. Sambrano, E. Longo, J. Andrés, M.S. Li, J.A. Varela, J. Alloys. Compd. 556, 153 (2013)
H. Yang, C. Huang, X. Su, A. Tang, J. Alloys Compd. 402(1–2), 274 (2005)
O.A. Korotchenkov, A. Cantarero, A.P. Shpak, YuA Kunitskii, A.I. Senkevich, M.O. Borovoy, A.B. Nadtochii, Nanotechnology 16, 2033 (2005)
G. Ghosh, M.K. Naskar, A. Patra, M. Chatterjee, Opt. Mater. 28, 1047 (2006)
H.C. Warad, S.C. Ghosh, B. Hemtanon, C. Thanachayanont, J. Dutta, Sci. Technol. Adv. Mater. 6, 296 (2005)
R.K. Upadhyay, M. Sharma, D.K. Singh, S.S. Amritphale, N. Chandra, Sep. Purif. Technol. 88, 39 (2012)
B.D. Cullity, Elements of X-ray Diffraction, 2nd edn. (Addison Wesley Company, USA, 1956), p. 102
B. Geng, J. Ma, F. Zhan, Mater. Chem. Phys. 534, 113 (2009)
M. Nell, J. Marohn, G. Mclendon, J. Chem. Phys. 4359, 94 (1990)
L. Spanhel, M. Haase, H. Weller, A. Henglein, J. Am. Chem. Soc. 5649, 109 (1987)
W.Q. Peng, G.W. Cong, S.C. Qu, Z.G. Wang, Opt. Mater. 29, 313 (2006)
N. Soltani, E. Saion, M.Z. Hussein, M. Erfani, A. Abedini, G. Bahmanrokh, M. Navasery, P. Vaziri, Int. J. Mol. Sci. 13, 12242 (2012)
Acknowledgments
Authors would like to thank Dr. Navin Chandra, AMPRI, Bhopal for fruitful discussions and experimental support. Financial assistance from Madhya Pradesh Council of Science and Technology through project funding (1044/CST/R&D/2012) to AK is gratefully acknowledged.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kumar, A., Upadhyay, R.K. Optical, structural and morphological studies of ZnS nanoparticles synthesized using inorganic capping agent. J Mater Sci: Mater Electron 26, 2430–2435 (2015). https://doi.org/10.1007/s10854-015-2702-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10854-015-2702-3