Abstract
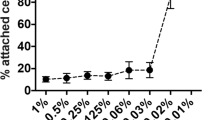
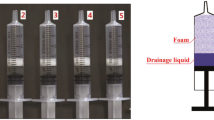
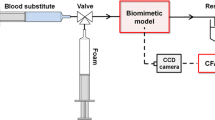
A novel analysis system for the quantification of sclerosing foam properties under clinically relevant conditions was developed with the purpose of establishing a robust methodology for comparative characterisation of different foam formulations and production strategies. The developed biomimetic-inspired model comprised of 4 or 10 mm inner diameter polytetrafluoroethylene tubing, filled with a blood substitute and fixed to a platform with an adjustable inclination angle. Sclerosing foams were produced by mixing polidocanol with either atmospheric air or 100 % CO2, using a double-syringe system method. Individual foams were injected into the tube, while videos were captured simultaneously. Videos were then transferred to an in-house computational foam analysis system (CFAS) which performed a sequence of semi-automated operations, allowing quantitative characterisation of sclerosing foam dynamic behaviour. Using CFAS, degradation rates of different foams were measured and the effect of gas composition, liquid sclerosant concentration and time delay between foam production and injection were evaluated.







Similar content being viewed by others
References
Pannier F, Rabe E. Endovenous laser therapy and radiofrequency ablation of saphenous varicose veins. J Cardiovasc Surg. 2006;47(1):3.
Sarin S, Scurr J, Smith P. Assessment of stripping the long saphenous vein in the treatment of primary varicose veins. Br J Surg. 1992;79(9):889–93.
Rivlin S. The surgical cure of primary varicose veins. Br J Surg. 1975;62(11):913–7.
Jia X, Mowatt G, Burr J, Cassar K, Cook J, Fraser C. Systematic review of foam sclerotherapy for varicose veins. Br J Surg. 2007;94(8):925–36.
Hamel-Desnos C, Desnos P, Wollmann JC, Ouvry P, Mako S, Allaert FA. Evaluation of the efficacy of polidocanol in the form of foam compared with liquid form in sclerotherapy of the greater saphenous vein: initial results. Dermatol Surg. 2003;29(12):1170–5.
Cabrera J, Cabrera J Jr, Garcia-Olmedo MA, Redondo P. Treatment of venous malformations with sclerosant in microfoam form. Arch Dermatol. 2003;139(11):1409.
Cavezzi A, Frullini A, Ricci S, Tessari L. Treatment of varicose veins by foam sclerotherapy: two clinical series. Phlebology. 2002;17(1):13–8.
Darke S, Baker S. Ultrasound-guided foam sclerotherapy for the treatment of varicose veins. Br J Surg. 2006;93(8):969–74.
Frullini A, Cavezzi A. Sclerosing foam in the treatment of varicose veins and telangiectases: history and analysis of safety and complications. Dermatol Surg. 2002;28(1):11–5.
Tessari L, Cavezzi A, Frullini A. Preliminary experience with a new sclerosing foam in the treatment of varicose veins. Dermatol Surg. 2001;27(1):58–60.
Smith PC. Foam and liquid sclerotherapy for varicose veins. Phlebology. 2009;24(1):62–72.
Myers K, Jolley D, Clough A, Kirwan J. Outcome of ultrasound-guided sclerotherapy for varicose veins: medium-term results assessed by ultrasound surveillance. Eur J Vasc Endovasc Surg. 2007;33(1):116–21.
Schramm LL. Emulsions, foams, and suspensions: fundamentals and applications. Weinheim: Wiley–VCH; 2005.
Simka M. Principles and technique of foam sclerotherapy and its specific use in the treatment of venous leg ulcers. Int J Low Extrem Wounds. 2011;10(3):138–45.
Watkins M. Deactivation of sodium tetradecyl sulphate injection by blood proteins. Eur J Vasc Endovasc Surg. 2011;41(4):521–5.
Wollmann J. Sclerosant foams. Phlébologie. 2010;39:208–17.
Höhler R, Cohen-Addad S. Rheology of liquid foam. J Phys Condens Matter. 2005;17:R1041.
Guex J. Complications and side-effects of foam sclerotherapy. Phlebology. 2009;24(6):270–4.
Weaire D, Hutzler S, Cox S, Kern N, Alonso MD, Drenckhan W. The fluid dynamics of foams. J Phys Condens Matter. 2003;15:S65.
Carugo D, Capretto L, Willis S, Lewis AL, Grey D, Hill M, et al. A microfluidic device for the characterisation of embolisation with polyvinyl alcohol beads through biomimetic bifurcations. Biomed Microdevices. 2012;14(1):153–63.
Segur JB, Oberstar HE. Viscosity of glycerol and its aqueous solutions. Ind Eng Chem. 1951;43(9):2117–20.
Pries A, Neuhaus D, Gaehtgens P. Blood viscosity in tube flow: dependence on diameter and hematocrit. Am J Physiol. 1992;263(6):H1770–8.
Fung Y. Biodynamics: circulation. New York: Springer; 1984.
Barrett JM, Allen B, Ockelford A, Goldman MP. Microfoam ultrasound-guided sclerotherapy of varicose veins in 100 legs. Dermatol Surg. 2004;30(1):6–12.
Gonzalez RC, Woods RE, Eddins SL. Digital image processing using MATLAB. USA: Prentice Hall Press; 2007.
Yang W, Cao W, Chung TS, Morris J. Applied numerical methods using MATLAB. USA: Wiley Online Library; 2005.
Eckmann DM. Polidocanol for endovenous microfoam sclerosant therapy. Expert Opin Investig Drugs. 2009;18(12):1919–27.
Morrison N. Foam sclerotherapy, how to improve results and reduce side effects. Phlébologie. 2009;62(1):23–34.
Morrison N, Neuhardt DL, Rogers CR, McEown J, Morrison T, Johnson E, et al. Comparisons of side effects using air and carbon dioxide foam for endovenous chemical ablation. J Vasc Surg. 2008;47(4):830–6.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Carugo, D., Ankrett, D.N., O’Byrne, V. et al. A novel biomimetic analysis system for quantitative characterisation of sclerosing foams used for the treatment of varicose veins. J Mater Sci: Mater Med 24, 1417–1423 (2013). https://doi.org/10.1007/s10856-013-4913-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10856-013-4913-6